 Back
BackBonding and Hybridization in Organic Molecules: Valence Bond Theory and Molecular Structure
Study Guide - Smart Notes

Bonding and Molecular Structure
Introduction to Covalent Bonding and Valence Bond Theory
Covalent bonding is fundamental to organic chemistry, describing how atoms share electron pairs to achieve stable electron configurations. Valence Bond Theory explains covalent bond formation as the result of the overlap of atomic orbitals, with electrons paired and attracted to the nuclei of both atoms. This theory is essential for understanding the structure and reactivity of organic molecules.
Covalent Bond: A chemical bond formed by the sharing of one or more pairs of electrons between atoms.
Valence Bond Theory: Describes bonds as resulting from the overlap of atomic orbitals, with electron pairs localized between atoms.
Sigma (σ) Bond: Formed by head-on overlap of orbitals along the axis connecting two nuclei; cylindrically symmetrical.
Pi (π) Bond: Formed by sideways overlap of parallel p orbitals; electron density is above and below the plane of the nuclei.
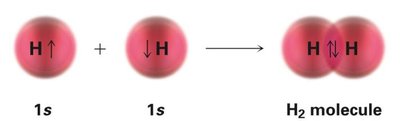
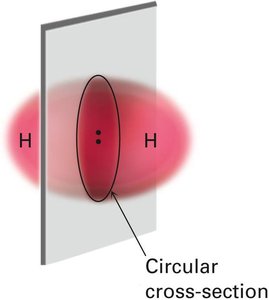
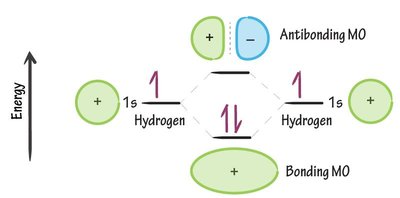
Molecular Orbitals: Bonding and Antibonding
When atomic orbitals combine, they form molecular orbitals that can be bonding or antibonding. In the hydrogen molecule (H2), two 1s orbitals combine to form a lower-energy bonding orbital (σ) and a higher-energy antibonding orbital (σ*). Electrons fill the bonding orbital, stabilizing the molecule.
Bonding Molecular Orbital (MO): Lower in energy, increases electron density between nuclei, stabilizing the molecule.
Antibonding Molecular Orbital (MO): Higher in energy, decreases electron density between nuclei, destabilizing the molecule.
p orbitals can combine to form π and π* molecular orbitals.
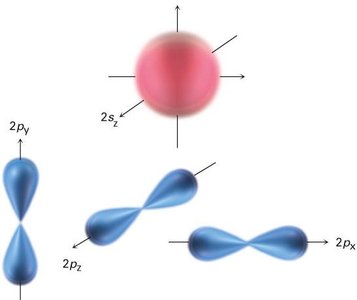
Hybridization and the Structure of Organic Molecules
sp3 Hybridization: Methane (CH4)
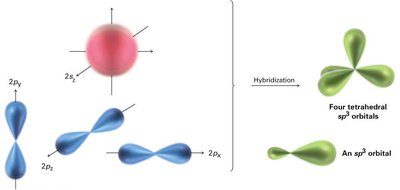
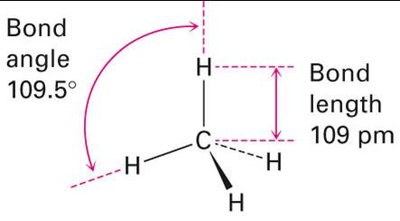
Carbon has four valence electrons and forms four equivalent bonds in methane, which are oriented toward the corners of a regular tetrahedron. This is explained by sp3 hybridization, where one 2s and three 2p orbitals mix to form four equivalent sp3 hybrid orbitals.
sp3 Hybrid Orbitals: Four orbitals arranged tetrahedrally, bond angles of 109.5°.
All C–H bonds in methane are identical due to hybridization.
Stronger bonds result from the effective overlap of sp3 orbitals with hydrogen 1s orbitals.
sp3 Hybridization in Ethane (C2H6)
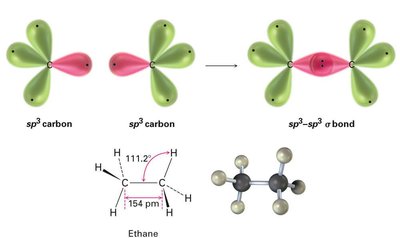
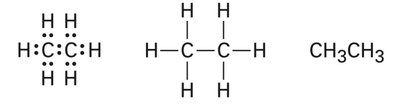
In ethane, each carbon is sp3 hybridized, forming a sigma bond between the two carbons and six C–H sigma bonds. The molecule is tetrahedral around each carbon, with bond angles close to 109.5°.
C–C Bond: Sigma bond formed by overlap of sp3 orbitals from each carbon.
C–H Bonds: Sigma bonds formed by overlap of sp3 orbitals with hydrogen 1s orbitals.
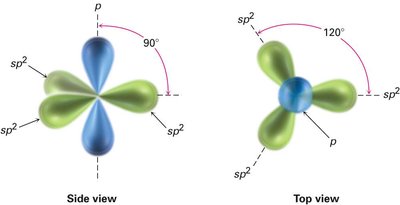
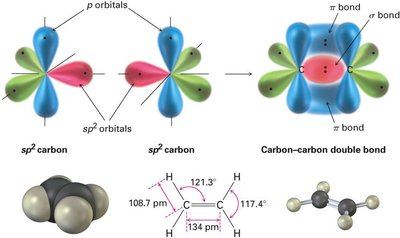
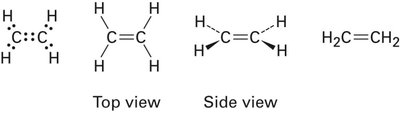
sp2 Hybridization: Ethylene (C2H4)
In ethylene, each carbon atom undergoes sp2 hybridization, mixing one s and two p orbitals to form three sp2 hybrid orbitals arranged in a plane at 120° angles. The remaining unhybridized p orbital on each carbon forms a π bond by sideways overlap, resulting in a double bond (one σ and one π bond).
sp2 Hybrid Orbitals: Three orbitals in a trigonal planar arrangement, bond angles of 120°.
Double Bond: Consists of one sigma (σ) bond and one pi (π) bond.
Planar structure, shorter and stronger C=C bond compared to C–C single bond.
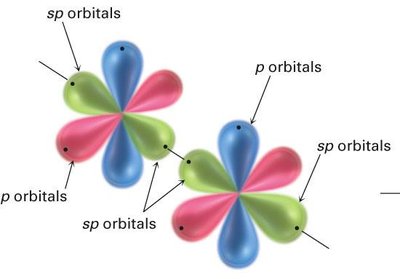
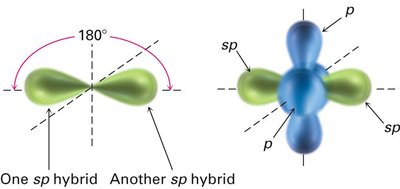
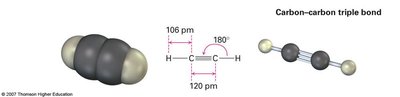
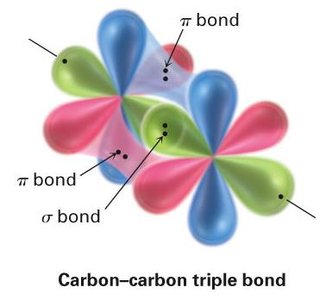
sp Hybridization: Acetylene (C2H2)
In acetylene, each carbon atom undergoes sp hybridization, mixing one s and one p orbital to form two sp hybrid orbitals oriented linearly at 180°. The two remaining unhybridized p orbitals on each carbon form two π bonds, resulting in a triple bond (one σ and two π bonds).
sp Hybrid Orbitals: Two orbitals arranged linearly, bond angles of 180°.
Triple Bond: Consists of one sigma (σ) bond and two pi (π) bonds.
Linear structure, shortest and strongest C≡C bond.
Hybridization in Oxygen and Nitrogen Compounds
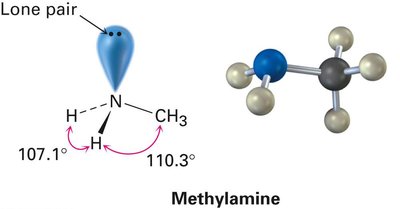
sp3 Hybridization in Water and Amines
Oxygen and nitrogen atoms also undergo hybridization. In water (H2O) and amines (e.g., methylamine), the central atom is sp3 hybridized, resulting in a tetrahedral arrangement of electron pairs. The presence of lone pairs affects bond angles, making them slightly less than the ideal tetrahedral angle.
Water (H2O): Oxygen is sp3 hybridized, with two lone pairs and two bonding pairs, bond angle ~105°.
Amines (e.g., methylamine): Nitrogen is sp3 hybridized, with one lone pair and three bonding pairs, bond angles ~107°.
sp2 Hybridization in Carbonyl and Imine Groups
Oxygen and nitrogen can also be sp2 hybridized, as seen in carbonyl (C=O) and imine (C=N) groups. Here, the double bond consists of one σ and one π bond, similar to the bonding in ethylene.
Carbonyl Group (C=O): Oxygen is sp2 hybridized, forming a planar structure with a double bond to carbon.
Imine Group (C=N): Nitrogen is sp2 hybridized, forming a planar structure with a double bond to carbon.
Summary Table: Types of Hybridization and Molecular Geometry
Hybridization | Number of Hybrid Orbitals | Geometry | Bond Angle | Example |
|---|---|---|---|---|
sp3 | 4 | Tetrahedral | 109.5° | Methane (CH4), Ethane (C2H6), Water (H2O) |
sp2 | 3 | Trigonal planar | 120° | Ethylene (C2H4), Carbonyl (C=O) |
sp | 2 | Linear | 180° | Acetylene (C2H2) |
Key Points and Summary
Covalent bonds are formed by sharing electron pairs between atoms.
Valence bond theory explains bonding as the overlap of atomic orbitals.
Sigma (σ) bonds are formed by head-on overlap; pi (π) bonds by sideways overlap.
Hybridization explains the observed geometries of organic molecules:
sp3: Tetrahedral (single bonds)
sp2: Trigonal planar (double bonds)
sp: Linear (triple bonds)
Oxygen and nitrogen also undergo hybridization, affecting the structure and reactivity of organic compounds.
