 Back
BackBonding and Polarity: Electronegativity, Inductive Effect, and Dipole Moments
Study Guide - Smart Notes

Bonding and Polarity
Introduction to Bonding and Polarity
Chemical bonds in organic molecules can be classified based on how electrons are distributed between atoms. The nature of these bonds—ionic, nonpolar covalent, or polar covalent—determines many physical and chemical properties of molecules. Understanding bond polarity, electronegativity, and the inductive effect is essential for predicting molecular behavior and reactivity.
Types of Chemical Bonds
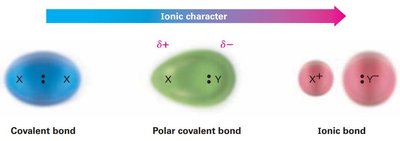
Ionic Bonds: Formed when electrons are transferred from one atom to another, resulting in oppositely charged ions held together by electrostatic attraction (e.g., NaCl).
Nonpolar Covalent Bonds: Electrons are shared equally between two atoms, leading to a symmetrical electron distribution (e.g., C–C bond in ethane).
Polar Covalent Bonds: Electrons are shared unequally due to differences in electronegativity, resulting in partial charges (δ+ and δ–) on the bonded atoms.
Electronegativity
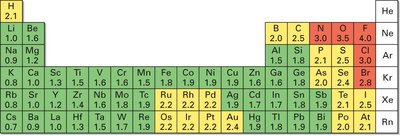
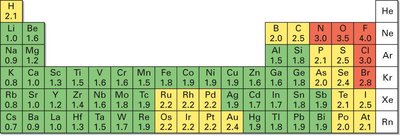
Electronegativity (EN) is the intrinsic ability of an atom to attract shared electrons in a covalent bond. It generally increases across a period (left to right) and up a group (bottom to top) in the periodic table. Reactive nonmetals have high EN, while metals have low EN.
Trends: Highest in the upper right (e.g., F, O, N), lowest in the lower left (e.g., Cs, Fr).
Classification of Bonds by EN Difference:
EN difference < 0.5: Nonpolar covalent
EN difference 0.5–2.0: Polar covalent
EN difference > 2.0: Ionic
Bond Polarity and Direction
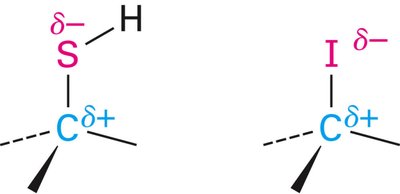
Bond polarity arises from differences in electronegativity. The more electronegative atom attracts electrons more strongly, acquiring a partial negative charge (δ–), while the less electronegative atom becomes partially positive (δ+). The direction of bond polarity is indicated by a cross arrow (→), with the tail at the electron-poor (δ+) end and the head at the electron-rich (δ–) end.
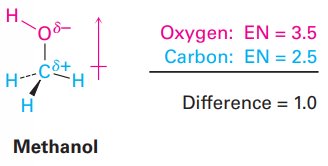
Example: In a C–O bond (EN difference = 1.0), oxygen is δ– and carbon is δ+.
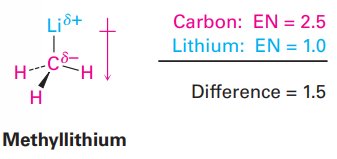
Example: In a C–Li bond (EN difference = 1.5), carbon is δ– and lithium is δ+.
Polarizability and the Inductive Effect
Polarizability refers to the ease with which the electron cloud around an atom can be distorted by an external electric field. Larger atoms with loosely held electrons are more polarizable, while smaller atoms with tightly held electrons are less polarizable.
The inductive effect is the electron-withdrawing or electron-donating effect transmitted through sigma (σ) bonds due to differences in electronegativity. Electronegative atoms withdraw electrons (–I effect), while metals can donate electrons (+I effect). The inductive effect influences chemical reactivity and stability of molecules.
Polarity in Functional Groups and Molecules
Polarity patterns are observed in common functional groups. Bonds between carbon and more electronegative atoms (N, O, Cl, F, Br) are polarized, making carbon partially positive. In contrast, carbon bonded to less electronegative metals (e.g., Mg, Li) is partially negative.
Polar molecules: Water (H2O), ethanol (CH3CH2OH), acetic acid (CH3COOH)
Nonpolar molecules: Hydrocarbons (ethane, benzene), CO2
Dipole Moment
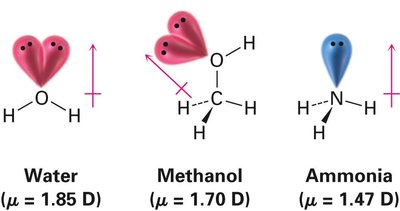
The dipole moment (μ) is a quantitative measure of molecular polarity, defined as the product of the magnitude of the charge and the distance between the charges. It is measured in Debye units (D). For molecules with only one covalent bond, the molecular dipole moment equals the bond dipole moment. For polyatomic molecules, the overall dipole moment is the vector sum of all bond dipoles and lone-pair contributions.
Symmetrical molecules (e.g., CO2, CH4, C2H6, benzene) have zero net dipole moment due to cancellation of individual bond dipoles.
Asymmetrical molecules (e.g., H2O, CH3OH, NH3) have nonzero dipole moments.
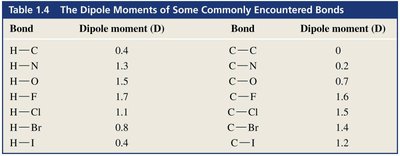
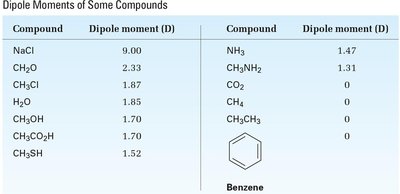
Tables: Dipole Moments of Bonds and Compounds
The following tables summarize the dipole moments of some commonly encountered bonds and compounds. These values help predict molecular polarity and solubility behavior.
Bond | Dipole moment (D) | Bond | Dipole moment (D) |
|---|---|---|---|
H—C | 0.4 | C—C | 0 |
H—N | 1.3 | C—N | 0.2 |
H—O | 1.5 | C—O | 0.7 |
H—F | 1.7 | C—F | 1.6 |
H—Cl | 1.1 | C—Cl | 1.5 |
H—Br | 0.8 | C—Br | 1.4 |
H—I | 0.4 | C—I | 1.2 |
Compound | Dipole moment (D) | Compound | Dipole moment (D) |
|---|---|---|---|
NaCl | 9.00 | NH3 | 1.47 |
CH2O | 2.33 | CH3NH2 | 1.31 |
CH3Cl | 1.87 | CO2 | 0 |
H2O | 1.85 | CH4 | 0 |
CH3OH | 1.70 | CH3CH3 | 0 |
CH3CO2H | 1.70 | Benzene | 0 |
CH3SH | 1.52 |
Summary
Bond polarity is determined by differences in electronegativity between atoms.
The inductive effect and polarizability influence electron distribution and molecular reactivity.
Molecular polarity is quantified by the dipole moment, which is the vector sum of all bond dipoles and lone-pair contributions.
Understanding these concepts is fundamental for predicting the behavior of organic molecules in various chemical contexts.
