 Back
Backπ-Bonds as Nucleophiles and Electrophilic Addition Reactions of Alkenes
Study Guide - Smart Notes

π-Bonds as Nucleophiles
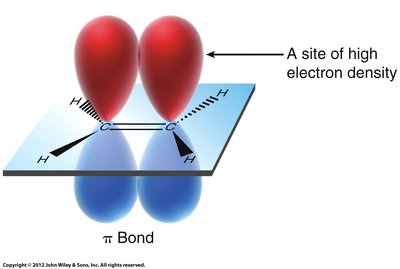
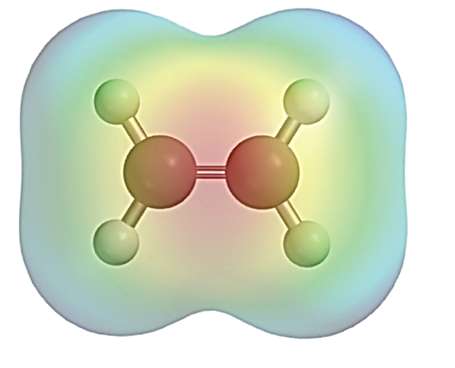
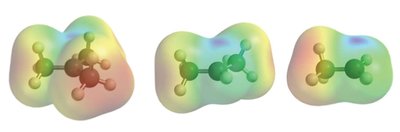
Structure and Electron Density of π Bonds
Alkenes contain a π bond formed by the sideways overlap of p orbitals, resulting in a region of high electron density above and below the plane of the carbon atoms. This electron-rich area makes π bonds act as nucleophiles, readily interacting with electrophilic reagents.
π Bond: A molecular orbital formed from the overlap of two p orbitals, found in double bonds (C=C).
Nucleophile: A species that donates an electron pair to an electrophile.
Electrostatic Potential Mapping: Shows regions of high electron density (often colored red) around the π bond.
Example: Ethene (C2H4) has a π bond that is susceptible to attack by electrophiles.
Electrophilic Addition Reactions of Alkenes
Overview of Addition Reactions
Alkenes undergo a variety of electrophilic addition reactions where the π bond acts as a nucleophile and reacts with electrophiles. These reactions are fundamental in organic synthesis and include:
Hydrohalogenation: Addition of HX (where X = Cl, Br, I) to alkenes.
Acid-Catalyzed Hydration: Addition of water to form alcohols.
Acid-Catalyzed Alcohol Addition: Addition of alcohols to alkenes.
Oxymercuration-Demercuration: Hydration without carbocation rearrangement.
Halogenation: Addition of halogens (Br2, Cl2).
Hydroboration-Oxidation: Alternative method for hydration.
Epoxidation: Formation of epoxides.
Hydrogenation: Addition of hydrogen (H2).
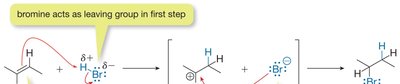
Hydrohalogenation Mechanism
Hydrohalogenation involves the addition of a hydrogen halide (HX) to an alkene. The reaction proceeds via a two-step mechanism:
The π bond attacks the hydrogen of HX, forming a carbocation intermediate and a halide ion.
The halide ion then attacks the carbocation, forming the final alkyl halide product.

Reaction Energy Diagrams
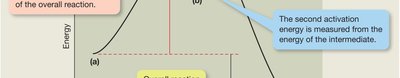
Energy Profile of Electrophilic Addition
The energy diagram for electrophilic addition reactions shows two transition states corresponding to the two steps of the mechanism. The first step, formation of the carbocation, is usually the rate-determining step and has a higher activation energy.
Activation Energy: The energy required to reach the transition state from the reactants.
Rate-Determining Step: The slowest step in a reaction mechanism, which controls the overall rate.

Regioselectivity and Carbocation Stability
Regioselectivity in Addition Reactions
Regioselectivity refers to the preference for forming one constitutional isomer over another in a reaction. In hydrohalogenation, regioselectivity is determined by the stability of the carbocation intermediate.
Constitutional Isomers: Compounds with the same molecular formula but different connectivity.
Carbocation Stability: More substituted carbocations are more stable due to hyperconjugation and resonance.
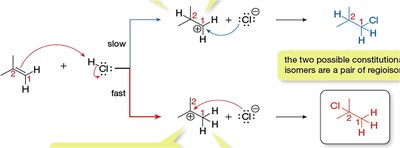
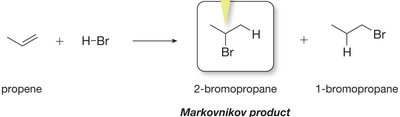
Markovnikov’s Rule
Markovnikov’s Rule states that in the addition of HX to an alkene, the hydrogen atom attaches to the carbon with more hydrogens, and the halide attaches to the more substituted carbon. This is because the most stable carbocation intermediate determines the major product.
Hyperconjugation: Stabilization by electron donation from adjacent σ bonds.
Resonance Stabilization: Delocalization of charge in conjugated systems.
Hammond's Postulate
Transition State Theory
Hammond's Postulate states that the structure of the transition state resembles the species (reactants or products) closest in energy. In electrophilic addition, a more stable carbocation has a lower activation energy, making its formation more favorable.
Transition State: A high-energy, unstable state during a chemical reaction.
Activation Energy: Lower for more stable carbocations.

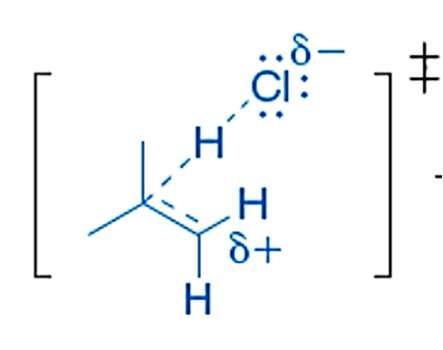
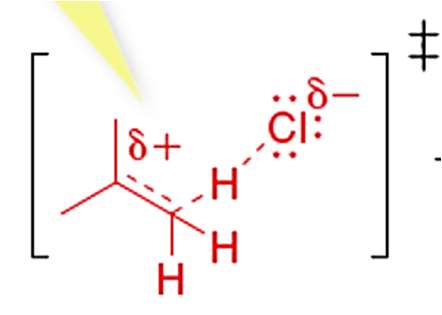
Hyperconjugation and Carbocation Stability
Mechanism of Hyperconjugation
Hyperconjugation is a stabilizing interaction where electrons from a σ bond (typically C-H or C-C) interact with a vacant p orbital on a carbocation. This electron donation reduces the positive charge on the carbocation, increasing its stability.
Proper Alignment: Hyperconjugation occurs only when the σ bond is aligned (eclipsed) with the vacant p orbital.
Carbocation Stability: Increases with the number of alkyl group substitutions.
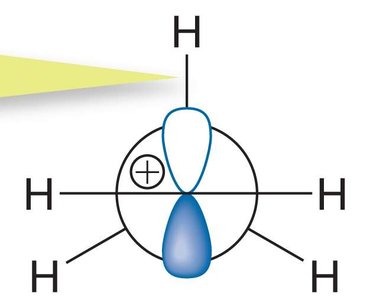
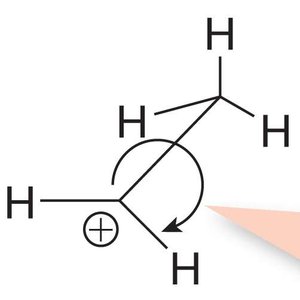
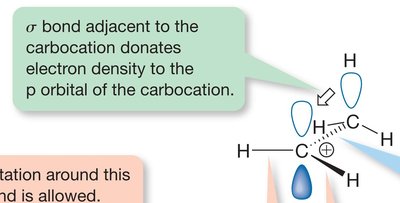
Relative Stability of Carbocations
Carbocations are classified by the number of alkyl groups attached to the positively charged carbon:
Tertiary Carbocation: Three alkyl groups (most stable).
Secondary Carbocation: Two alkyl groups.
Primary Carbocation: One alkyl group.
Methyl Carbocation: No alkyl groups (least stable).






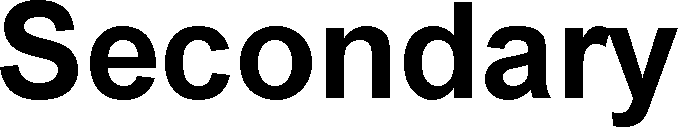

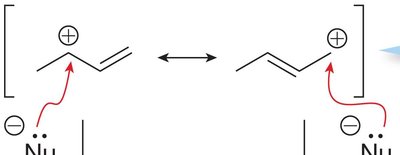
Resonance Stabilization of Carbocations
Charge Delocalization
Carbocations adjacent to π bonds or atoms with lone pairs (O, N, S) can undergo resonance, spreading the positive charge over multiple atoms. This increases the stability of the carbocation and can lead to multiple reactive sites, resulting in a mixture of products.
Resonance: Delocalization of electrons across adjacent atoms.
Example: Allylic and benzylic carbocations are stabilized by resonance.
Carbocation Stability Summary Table
The stability of carbocations increases with the degree of alkyl substitution and resonance stabilization. The following table summarizes the relative stability:
Carbocation Type | Stability |
|---|---|
Methyl | Least stable |
Primary alkyl | Low stability |
Secondary alkyl | Moderate stability |
Tertiary alkyl | High stability |
Allylic/benzylic (resonance) | Most stable |

Practice Problem
Mechanism Drawing and Major Product Identification
Draw the mechanism for the addition of HBr to a cyclic alkene and identify the major product based on carbocation stability and Markovnikov's rule.
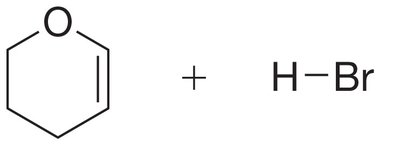 Additional info: The notes above expand on the mechanistic details, stability factors, and theoretical principles underlying electrophilic addition reactions of alkenes, providing context for exam preparation and practical application in organic synthesis.
Additional info: The notes above expand on the mechanistic details, stability factors, and theoretical principles underlying electrophilic addition reactions of alkenes, providing context for exam preparation and practical application in organic synthesis.
