 Back
BackCarboxylic Acids and Carboxylic Acid Derivatives: Structure, Properties, Synthesis, and Reactions
Study Guide - Smart Notes

Carboxylic Acids: Structure, Properties, and Nomenclature
Introduction to Carboxylic Acids
Carboxylic acids are a fundamental class of organic compounds characterized by the presence of a carboxyl group (-COOH). They are widespread in nature and play essential roles in biological and industrial processes. Common examples include citric acid (found in citrus fruits), butyric acid (in dairy products), acetic acid (in vinegar), aspirin (acetylsalicylic acid), and amino acids (building blocks of proteins).



Nomenclature of Carboxylic Acids
Carboxylic acids are named according to IUPAC rules. The parent chain includes the carboxyl group, and the suffix "-oic acid" is used. For example, ethanoic acid (acetic acid) and propanoic acid.
Structure and Physical Properties
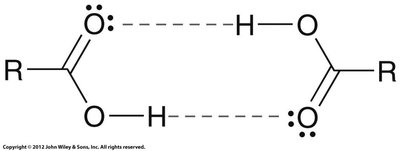
The carbon atom in the carboxyl group exhibits trigonal planar geometry due to sp2 hybridization. Carboxylic acids are capable of strong hydrogen bonding, including dimer formation, which leads to relatively high boiling points compared to similar-sized alcohols.
Hydrogen bonding: Carboxylic acids can form dimers via two hydrogen bonds, increasing their boiling points.
Boiling points: For example, acetic acid boils at 118°C, while isopropanol boils at 82°C.
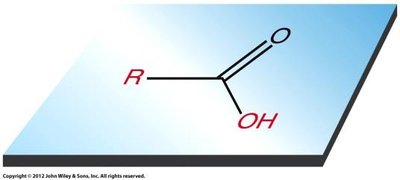
Acidity of Carboxylic Acids
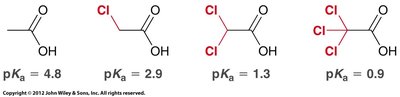
Carboxylic acids are weak acids, typically with pKa values between 4 and 5. Their acidity is due to resonance stabilization of the conjugate base (carboxylate ion) and is further influenced by inductive effects from substituents.
Resonance stabilization: The negative charge in the carboxylate ion is delocalized over two oxygen atoms.
Inductive effects: Electron-withdrawing groups (e.g., halogens) increase acidity by stabilizing the negative charge.
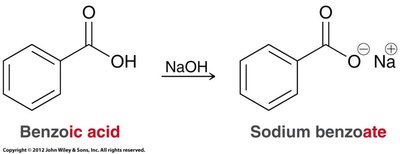
Nomenclature: The conjugate base is named with the suffix "-oate" (e.g., acetate, benzoate).
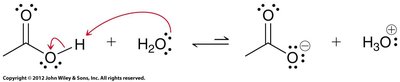
Preparation of Carboxylic Acids
Review of Methods
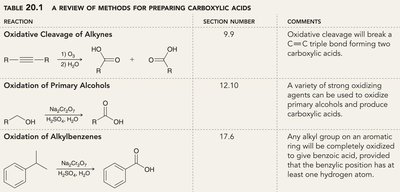
Carboxylic acids can be synthesized by several oxidation reactions:
Reaction | Section Number | Comments |
|---|---|---|
Oxidative Cleavage of Alkynes | 9.9 | Breaks C≡C triple bond, forming two carboxylic acids. |
Oxidation of Primary Alcohols | 12.10 | Strong oxidizing agents convert primary alcohols to acids. |
Oxidation of Alkylbenzenes | 17.6 | Any alkyl group on an aromatic ring is oxidized to benzoic acid. |
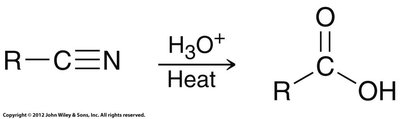
Hydrolysis of Nitriles
Nitriles can be hydrolyzed to carboxylic acids via acid or base catalysis. This method is useful for converting alkyl halides to carboxylic acids in two steps: SN2 substitution with cyanide, followed by hydrolysis.
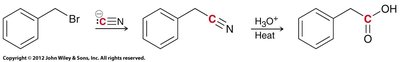
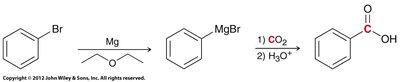
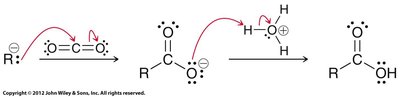
Carboxylation of Grignard Reagents
Grignard reagents react with carbon dioxide to form carboxylic acids after acidic workup. This is a valuable method for extending carbon chains.
Reactions of Carboxylic Acids
General Reactivity
Carboxylic acids undergo several important reactions:
Proton transfer: Formation of carboxylate salts by reaction with bases.
Reduction: Conversion to primary alcohols using strong reducing agents.
Reduction to Alcohols
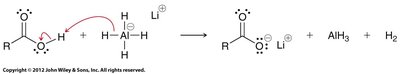
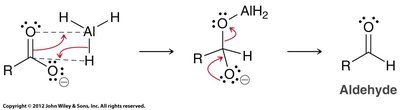
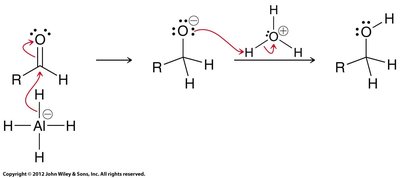
Carboxylic acids can be reduced to primary alcohols using lithium aluminum hydride (LiAlH4, LAH) or borane (BH3).
LAH reduction: Proceeds via aldehyde intermediate, but cannot be stopped at the aldehyde stage.
Borane reduction: Selective for carboxylic acids and milder than LAH.
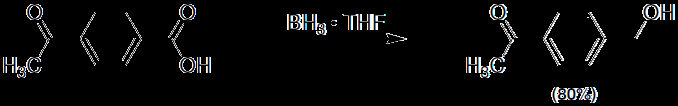
Carboxylic Acid Derivatives
Definition and Types
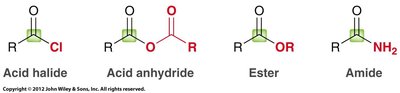
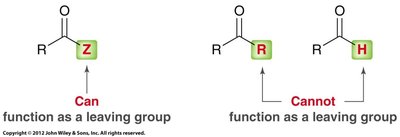
Carboxylic acid derivatives are compounds in which the hydroxyl group of the carboxylic acid is replaced by another group (Z). Common derivatives include acid halides, acid anhydrides, esters, amides, and nitriles.


Occurrence in Nature
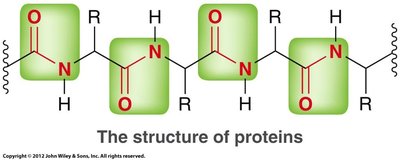
Esters are often responsible for pleasant odors, while amides are prevalent in biological molecules such as proteins. Acid halides and anhydrides are less common in nature due to their instability.
Naming Carboxylic Acid Derivatives
Acid halides: Replace "-ic acid" with "-yl halide" (e.g., acetyl chloride).
Acid anhydrides: Replace "acid" with "anhydride" (e.g., acetic anhydride).
Esters: Name the alkyl group attached to oxygen, then the acid with "-ate" (e.g., ethyl acetate).
Amides: Replace "-ic acid" or "-oic acid" with "amide" (e.g., acetamide).
Nitriles: Replace "-ic acid" or "-oic acid" with "onitrile" (e.g., acetonitrile).
Reactivity of Carboxylic Acid Derivatives
Factors Affecting Reactivity
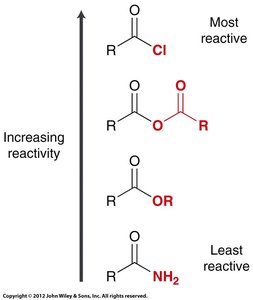
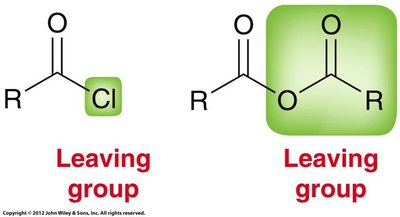
The reactivity of carboxylic acid derivatives as electrophiles depends on induction, resonance, steric effects, and the quality of the leaving group. Acid chlorides are the most reactive, while amides are the least reactive.
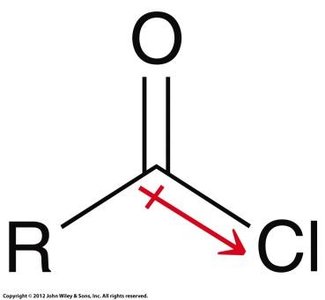
Acid Chlorides
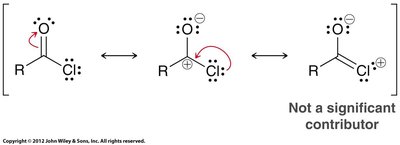
Acid chlorides are highly reactive due to the electron-withdrawing effect of chlorine and the quality of chloride as a leaving group. There are three resonance contributors, but the one with a positive charge on chlorine is not significant.
Amides
Amides are the least reactive due to resonance stabilization (partial double bond character between nitrogen and carbonyl carbon) and the poor leaving group ability of the amide ion.
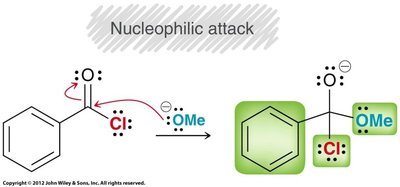
Nucleophilic Acyl Substitution
Mechanism and General Features
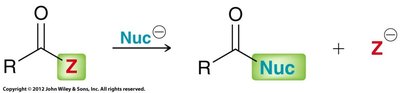
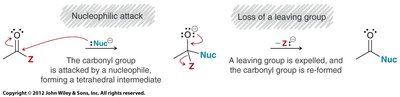
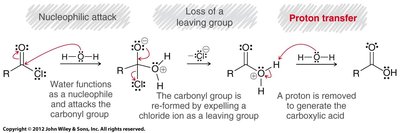
Carboxylic acid derivatives undergo nucleophilic acyl substitution, a two-step process involving nucleophilic attack on the carbonyl carbon followed by loss of a leaving group. This is distinct from aldehydes and ketones, which lack a suitable leaving group.

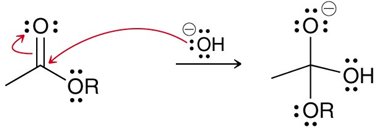
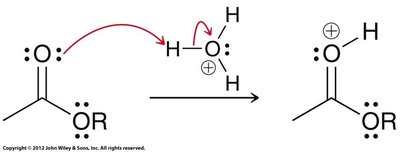
Acidic vs. Basic Conditions
The mechanism of nucleophilic acyl substitution varies with reaction conditions. Under acidic conditions, negative charges are rare; under basic conditions, positive charges are rare. Proton transfers may be required before, during, or after the main steps to maintain charge consistency.

Acid Halides and Acid Anhydrides
Preparation
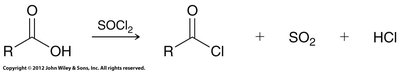
Acid chlorides: Prepared by treating carboxylic acids with thionyl chloride (SOCl2).
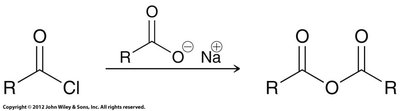
Acid anhydrides: Prepared by reacting an acid chloride with a carboxylate anion.
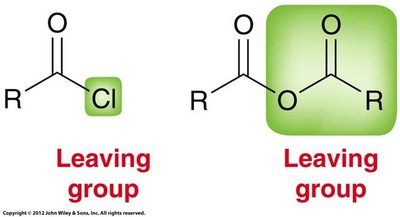
Reactions
Hydrolysis: Reacts with water to regenerate carboxylic acid.
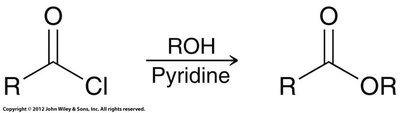
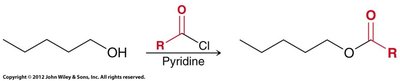
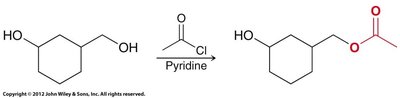
Alcoholysis: Reacts with alcohols to form esters.
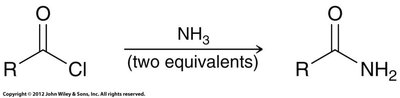
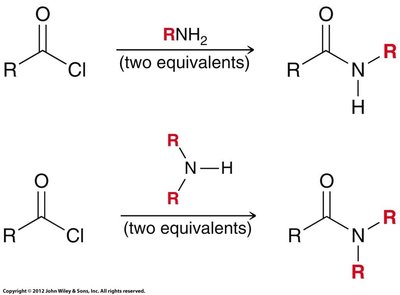
Aminolysis: Reacts with amines to form amides.
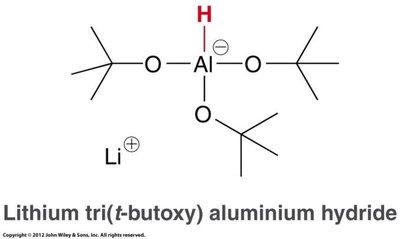
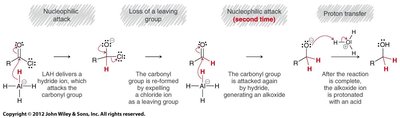
Reduction: Reduced to alcohols or aldehydes (with bulky LAH derivatives).
Organometallic reagents: Reacts with Grignard or Gilman reagents to form alcohols or ketones.
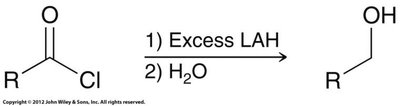
Esters
Preparation of Esters
SN2 process: Deprotonation of carboxylic acid followed by reaction with alkyl halide.
Alcoholysis: Reaction of acid chloride or anhydride with alcohol.
Fischer esterification: Acid-catalyzed reaction of carboxylic acid with alcohol; equilibrium process.
Reactions of Esters
Hydrolysis: Acid- or base-catalyzed (saponification) to yield carboxylic acid and alcohol.
Reduction: Excess LAH reduces esters to alcohols; DIBAH can stop at the aldehyde stage.
Reaction with Grignard reagents: Forms tertiary alcohols after two additions.
Amides
Preparation and Reactions
Preparation: Most efficiently made by reacting acid chlorides with amines.
Hydrolysis: Acid- or base-catalyzed hydrolysis to carboxylic acids; requires high heat and is slow.
Reduction: Reduced to amines via imine intermediate using LAH.
Nitriles
Preparation and Reactions
Preparation: SN2 reaction of alkyl halides with cyanide ion; dehydration of amides.
Hydrolysis: Acid- or base-catalyzed hydrolysis to carboxylic acids via amide intermediate.
Reaction with Grignard reagents: Forms imines, which are hydrolyzed to ketones.
Reduction: Reduced to amines using LAH.
Summary Table: Reactivity of Carboxylic Acid Derivatives
Derivative | Reactivity |
|---|---|
Acid chloride | Most reactive |
Acid anhydride | High reactivity |
Ester | Moderate reactivity |
Amide | Least reactive |
