 Back
BackCarboxylic Acids and Nitriles (Chapter 19)
Study Guide - Smart Notes

Carboxylic Acids and Nitriles
Introduction
This chapter explores the structure, nomenclature, physical properties, and chemical reactivity of carboxylic acids and nitriles. These functional groups are central to organic chemistry and biochemistry, playing key roles in metabolism, synthesis, and industrial applications.
Structure and Bonding
Carboxylic Acid Structure
Carboxylic acids contain the carboxy group (–COOH), which consists of a carbonyl (C=O) and a hydroxyl (–OH) group attached to the same carbon atom. The structure is often abbreviated as RCOOH or RCO2H.
The central carbon is sp2 hybridized, resulting in a planar structure.
The C=O bond is shorter and stronger than the C–O single bond.
Both the C–O and O–H bonds are polar due to the high electronegativity of oxygen.
Nitrile Structure
Nitriles are organic compounds containing a cyano group (–C≡N) bonded to an alkyl group. The carbon of the cyano group is sp hybridized, making the group linear with a bond angle of 180°.
The C≡N bond consists of one σ and two π bonds.
The carbon in the cyano group is electrophilic and susceptible to nucleophilic attack.

Nomenclature
Naming Carboxylic Acids
Carboxylic acids are named according to IUPAC rules:
If the COOH is bonded to a chain, the parent alkane's "e" is replaced with "oic acid" (e.g., pentanoic acid).
If the COOH is bonded to a ring, name the ring and add "carboxylic acid" (e.g., cyclohexanecarboxylic acid).
Number the chain to give the COOH group position 1, but omit the number in the name.
Diacids (with two COOH groups) use the suffix "-dioic acid" (e.g., succinic acid is butanedioic acid).
Naming Metal Salts of Carboxylate Anions
Metal salts of carboxylic acids are named by combining the metal cation, the parent acid name, and the suffix "-ate" (e.g., sodium acetate, potassium propanoate).

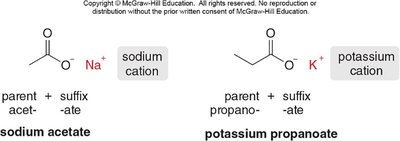
Naming Nitriles
Nitriles are named as alkane derivatives by adding "nitrile" to the parent chain containing the CN group. The chain is numbered to give the CN group position 1 (number omitted in the name). Common names are derived from the corresponding carboxylic acid by replacing "-ic acid" with "-onitrile". When the CN group is a substituent, it is called a cyano group.

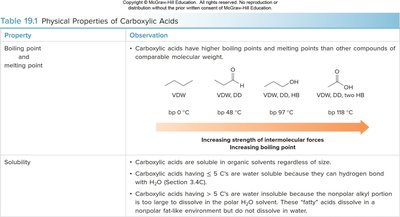
Physical Properties
Boiling and Melting Points
Carboxylic acids have higher boiling and melting points than other compounds of similar molecular weight due to strong hydrogen bonding and dipole-dipole interactions.
Boiling points increase with the strength of intermolecular forces.
Carboxylic acids with up to 5 carbons are water soluble; larger acids are less soluble due to the nonpolar alkyl group.
Property | Observation |
|---|---|
Boiling point and melting point | Carboxylic acids have higher boiling and melting points than other compounds of comparable molecular weight due to hydrogen bonding. |
Solubility | Carboxylic acids (≤5 C) are water soluble; larger acids are less soluble in water but soluble in organic solvents. |
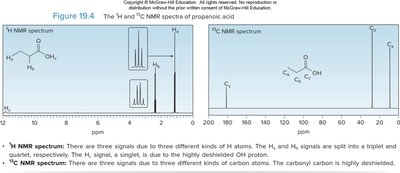
Spectroscopy of Carboxylic Acids
Infrared (IR) and NMR Spectroscopy
IR: C=O stretch at ~1710 cm-1; O–H stretch from 2500–3500 cm-1.
1H NMR: O–H proton at 10–12 ppm; α-protons at 2–2.5 ppm.
13C NMR: C=O carbon at 170–210 ppm.
Carboxylic Acids in Nature and Industry
Examples and Applications
Formic acid: Responsible for ant stings.
Acetic acid: Main component of vinegar; used in polymer synthesis.
Hexanoic acid: Has a strong odor; found in locker rooms and goat-derived products.
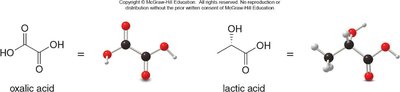
Oxalic acid: Found in spinach and rhubarb.
Lactic acid: Gives sour milk its taste.
Salts of Carboxylic Acids
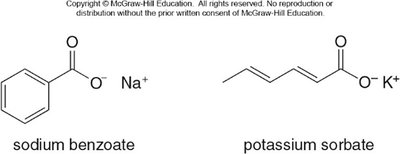
Sodium benzoate: Used as a preservative in soft drinks.
Potassium sorbate: Used to prolong shelf-life of foods.
Preparation of Carboxylic Acids
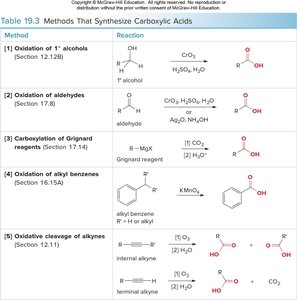
Methods of Synthesis
Oxidation of primary alcohols and aldehydes
Carboxylation of Grignard reagents
Oxidation of alkyl benzenes
Oxidative cleavage of alkynes
Method | Reaction |
|---|---|
Oxidation of 1° alcohols | RCH2OH → RCOOH |
Oxidation of aldehydes | RCHO → RCOOH |
Carboxylation of Grignard reagents | RMgX + CO2 → RCOOH |
Oxidation of alkyl benzenes | Ar-CH2R → Ar-COOH |
Oxidative cleavage of alkynes | RC≡CR' → RCOOH + R'COOH |
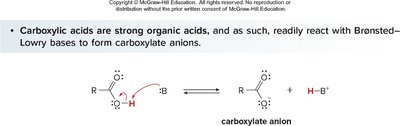
Acid–Base Reactions of Carboxylic Acids
Deprotonation and Conjugate Base Stability
Carboxylic acids are strong organic acids and react with bases to form carboxylate anions. The acidity is determined by the stability of the conjugate base, which is resonance stabilized.
Bases with conjugate acids having pKa > 5 can deprotonate carboxylic acids.
Resonance stabilization of the carboxylate anion is a key factor in acidity.
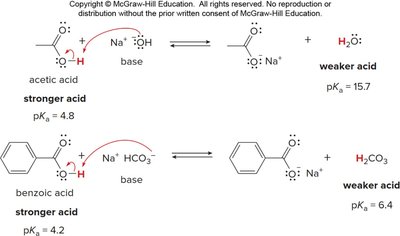
Base | Conjugate acid (pKa) |
|---|---|
Na+ HCO3- | H2CO3 (6.4) |
NH3 | NH4+ (9.4) |
Na2CO3 | HCO3- (10.2) |
Na+ OCH3- | CH3OH (15.5) |
Na+ OH- | H2O (15.7) |
Na+ OCH2CH3- | CH3CH2OH (16) |
H- | H2 (35) |

Resonance Stabilization
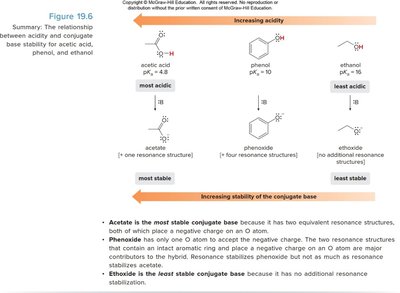
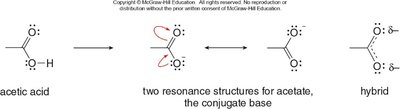
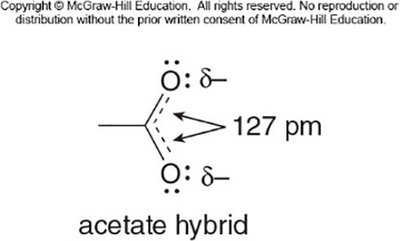
The carboxylate anion formed after deprotonation is stabilized by resonance, with the negative charge delocalized over two oxygen atoms. This makes carboxylic acids more acidic than alcohols or phenols.
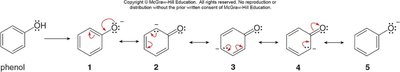
Comparison with Phenol and Ethanol
Acetate is the most stable conjugate base due to resonance. Phenoxide is less stable (only one oxygen for delocalization), and ethoxide is the least stable (no resonance stabilization).
Inductive and Substituent Effects
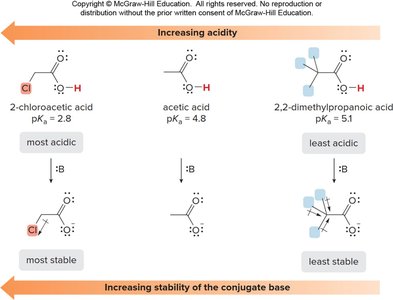
Inductive Effects
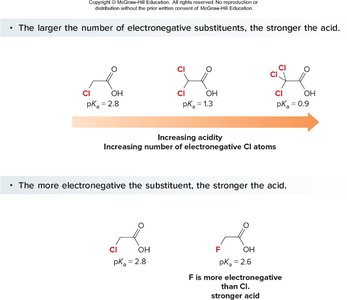
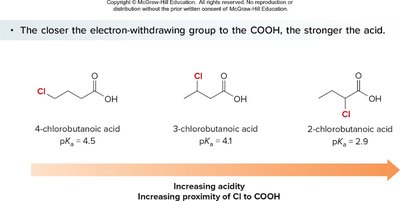
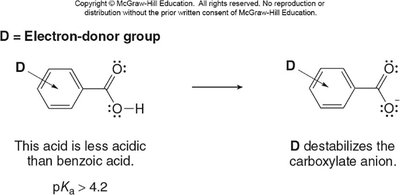
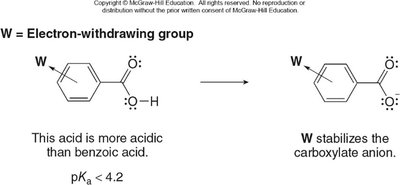
Electron-withdrawing groups (EWGs) stabilize the conjugate base, increasing acidity. Electron-donating groups (EDGs) destabilize the conjugate base, decreasing acidity. The effect is stronger when the substituent is closer to the carboxyl group.

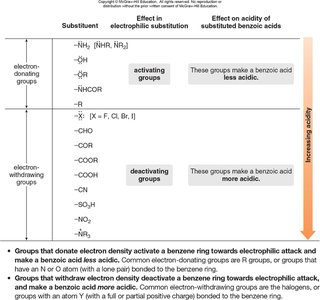
Substituent Effects in Aromatic Carboxylic Acids
Substituents on a benzene ring can either donate or withdraw electron density, affecting acidity:
Electron-donating groups (EDGs) destabilize the conjugate base, making the acid less acidic.
Electron-withdrawing groups (EWGs) stabilize the conjugate base, making the acid more acidic.
Substituent | Effect in Electrophilic Substitution | Effect on Acidity |
|---|---|---|
–NH2, –OH, –OR, –NHCOR | Activating group | Less acidic |
–X (halogens), –CHO, –COOR, –COOH, –CN, –NO2, –SO3H | Deactivating group | More acidic |
Amino Acids
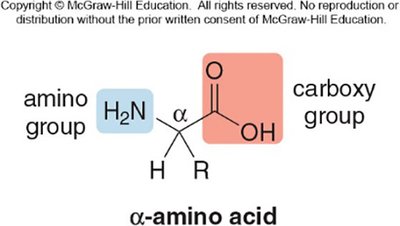
Structure and Stereochemistry
Amino acids contain both an amine (NH2) and a carboxy (COOH) group. The simplest amino acid is glycine (R = H). All other amino acids have a stereogenic α-carbon.
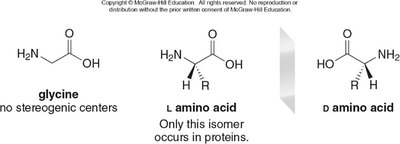
R group | Name | Three-letter abbreviation | One-letter abbreviation |
|---|---|---|---|
H | Glycine | Gly | G |
CH3 | Alanine | Ala | A |
CH2CH2Ph | Phenylalanine | Phe | F |
CH2OH | Serine | Ser | S |
CH2SH | Cysteine | Cys | C |
CH2CH2SCH3 | Methionine | Met | M |
CH2CH2COOH | Glutamic acid | Glu | E |
CH2CH2NH2 | Lysine | Lys | K |

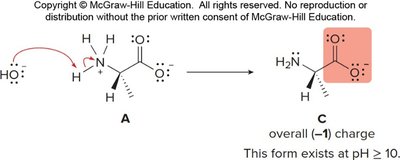
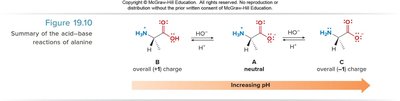
Zwitterion Character and pH Dependence
Amino acids exist as zwitterions (internal salts) at physiological pH, with both a positive (ammonium) and negative (carboxylate) charge. Their structure varies with pH:
At low pH: fully protonated, net positive charge.
At neutral pH: zwitterion, net zero charge.
At high pH: deprotonated, net negative charge.
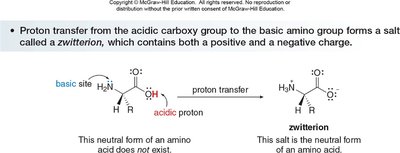
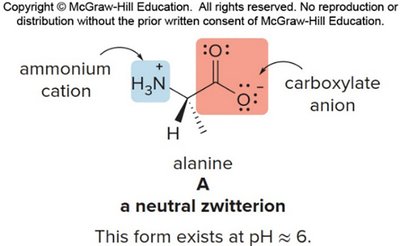
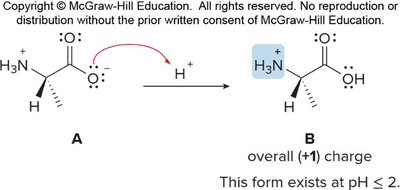

Isoelectric Point (pI)
The isoelectric point (pI) is the pH at which an amino acid exists primarily as a zwitterion (net neutral charge). It is calculated as the average of the two pKa values for the carboxyl and ammonium groups:
For alanine:

Reactions of Nitriles
Preparation of Nitriles
Nitriles are commonly prepared by SN2 substitution of alkyl halides with cyanide ion (–CN).

Hydrolysis of Nitriles
Nitriles are hydrolyzed with acid or base to yield carboxylic acids or carboxylate anions. The reaction proceeds via amide intermediates and involves the replacement of C–N bonds with C–O bonds.
*Additional info: The mechanism involves nucleophilic addition of water, followed by tautomerization and further hydrolysis.*
Reduction of Nitriles
Nitriles can be reduced to primary amines (with LiAlH4) or to aldehydes (with DIBAL-H). The mechanism involves sequential addition of hydride ions and protonation.
*Additional info: Grignard reagents and organolithium compounds react with nitriles to form ketones after hydrolysis.*
Summary
Carboxylic acids and nitriles are important functional groups with distinct structures, properties, and reactivity.
Nomenclature follows systematic IUPAC rules, with special conventions for salts and derivatives.
Physical properties are dominated by hydrogen bonding and polarity.
Acidity is influenced by resonance, inductive, and substituent effects.
Amino acids are biologically important carboxylic acids with both acidic and basic groups, existing as zwitterions at physiological pH.
Nitriles are versatile intermediates in organic synthesis, easily converted to carboxylic acids, amines, or ketones.
