 Back
BackCarboxylic Acids and Their Derivatives: Structure, Properties, and Reactions
Study Guide - Smart Notes

Carboxylic Acids and Derivatives
Introduction to Carboxylic Acids and Derivatives
Carboxylic acids and their derivatives are a central class of organic compounds characterized by the presence of a carbonyl group (C=O) bonded to an electronegative atom or group. These compounds are fundamental in both synthetic organic chemistry and biochemistry due to their versatile reactivity and prevalence in biological systems. The derivatives include esters, acyl chlorides, amides, and anhydrides, each with distinct reactivity profiles.
Carbonyl group: A functional group composed of a carbon atom double-bonded to an oxygen atom.
Acyl derivatives: Compounds in which the -OH group of a carboxylic acid is replaced by another group (e.g., -Cl, -OR, -NH2).
Nucleophilic acyl substitution: The primary reaction mechanism for these compounds, where a nucleophile replaces the leaving group attached to the acyl carbon.
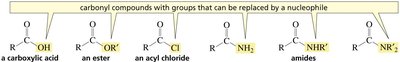
Structure and Properties
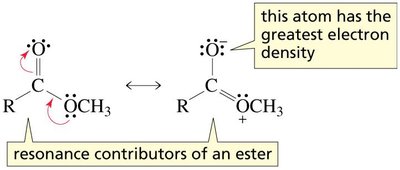
Resonance in Carboxylic Acid Derivatives
Resonance stabilization is a key feature of carboxylic acids and their derivatives. Resonance structures delocalize electron density, affecting both reactivity and physical properties. Amides exhibit the greatest resonance stabilization due to the ability of nitrogen to donate electron density efficiently.
Resonance contributors: Multiple Lewis structures can be drawn, showing delocalization of electrons between the carbonyl and adjacent atoms.
Amides: Show the greatest resonance stabilization, making them less reactive than other derivatives.
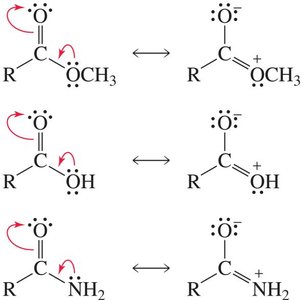
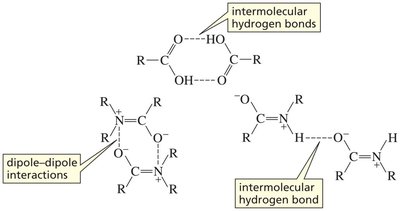
Boiling Points and Intermolecular Forces
The boiling points of carboxylic acids and their derivatives are influenced by their ability to form hydrogen bonds and dipole-dipole interactions. Amides and acids, in particular, have high boiling points due to extensive hydrogen bonding.
Hydrogen bonding: Carboxylic acids can form strong intermolecular hydrogen bonds, often dimerizing in the liquid phase.
Dipole-dipole interactions: Amides can engage in both hydrogen bonding and dipole-dipole interactions, further elevating their boiling points.
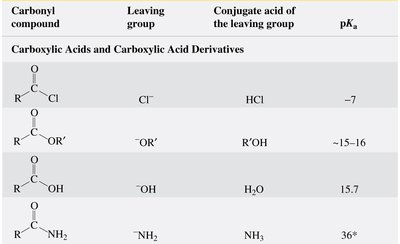
Leaving Groups and Reactivity
Leaving Group Ability and Reactivity Trends
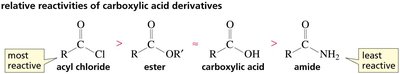
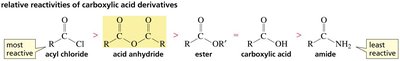
The reactivity of carboxylic acid derivatives is largely determined by the stability of the leaving group. The weaker the base (more stable the leaving group), the more reactive the derivative. This trend is reflected in the order: acyl chlorides > anhydrides > esters > carboxylic acids > amides.
Leaving group: The atom or group that departs with a pair of electrons during nucleophilic substitution.
pKa of conjugate acid: Lower pKa values indicate better leaving groups.
Carbonyl Compound | Leaving Group | Conjugate Acid of Leaving Group | pKa |
|---|---|---|---|
Acyl chloride | Cl- | HCl | -7 |
Ester | OR'- | R'OH | ~15-16 |
Carboxylic acid | OH- | H2O | 15.7 |
Amide | NH2- | NH3 | 36* |
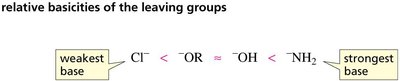
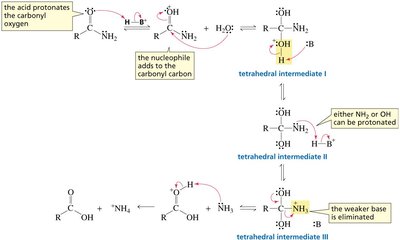
Nucleophilic Acyl Substitution Mechanism
General Mechanism
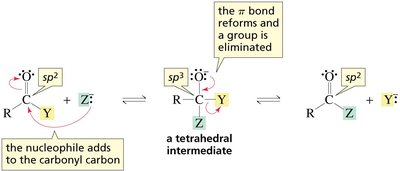
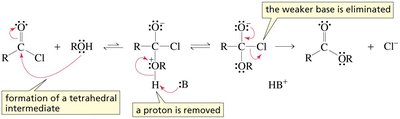
Nucleophilic acyl substitution involves two main steps: nucleophilic addition to the carbonyl carbon, forming a tetrahedral intermediate, followed by elimination of the leaving group. This mechanism is distinct from SN1 and SN2 mechanisms seen in alkyl halides.
Tetrahedral intermediate: A transient species formed when the nucleophile adds to the sp2 carbonyl carbon, converting it to sp3.
Elimination: The leaving group departs, restoring the carbonyl functionality.
Reactions of Acyl Chlorides
Reactivity and Mechanisms
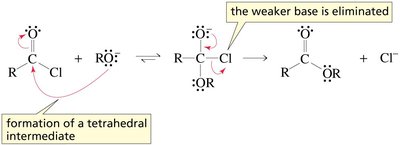
Acyl chlorides are the most reactive carboxylic acid derivatives due to the excellent leaving ability of chloride. They react readily with nucleophiles such as alcohols, water, and amines to form esters, acids, and amides, respectively.
Negatively charged nucleophile: Direct attack forms a tetrahedral intermediate, followed by elimination of Cl-.
Neutral nucleophile: Proton transfer is required to facilitate the elimination step.
Reactions of Esters
Hydrolysis and Transesterification
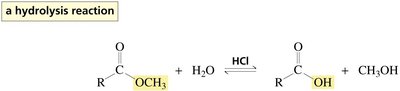
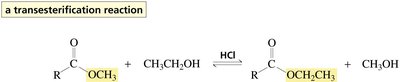
Esters undergo hydrolysis in the presence of acid or base to yield carboxylic acids and alcohols. Acid-catalyzed hydrolysis and transesterification require protonation of the carbonyl oxygen to increase electrophilicity. Aminolysis of esters with amines yields amides.
Hydrolysis: Ester + H2O (acid catalyst) → carboxylic acid + alcohol
Transesterification: Ester + alcohol (acid catalyst) → new ester + alcohol
Aminolysis: Ester + amine → amide + alcohol
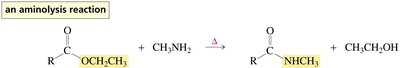
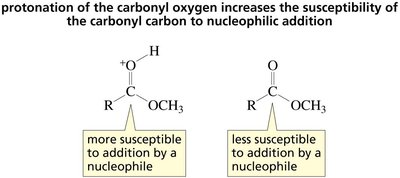
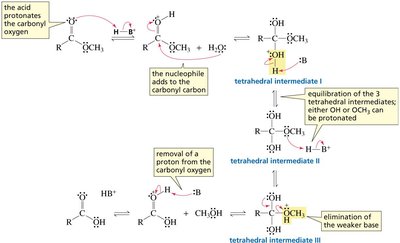
Mechanistic Details of Acid-Catalyzed Ester Hydrolysis
Protonation of the carbonyl oxygen increases the susceptibility of the carbonyl carbon to nucleophilic attack. The mechanism involves formation of tetrahedral intermediates and elimination of the weaker base as the leaving group.
Saponification: Base-Catalyzed Ester Hydrolysis
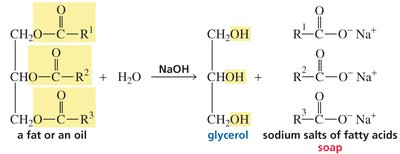
Soap Formation and Micelles
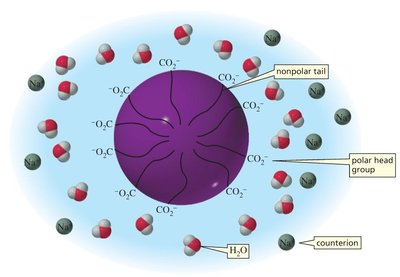
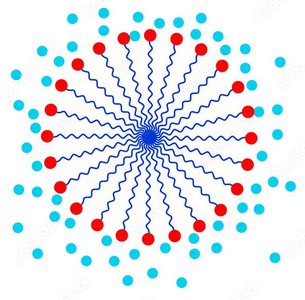
Saponification is the base-catalyzed hydrolysis of esters (typically fats or oils) to produce glycerol and the sodium salts of fatty acids (soap). Soap molecules aggregate into micelles, which encapsulate nonpolar dirt and grease in their hydrophobic interiors.
Saponification reaction: Fat/oil + NaOH → glycerol + soap (fatty acid salts)
Micelles: Spherical aggregates with hydrophobic tails inward and hydrophilic heads outward.
Detergents vs. Soaps
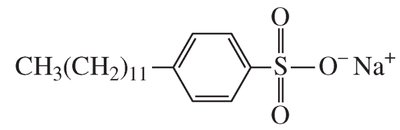
Detergents are synthetic analogs of soaps that contain sulfonate groups instead of carboxylates, preventing precipitation with divalent cations (soap scum).
Reactions of Carboxylic Acids
Fischer Esterification and Amide Formation
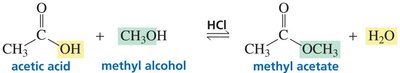
Carboxylic acids react with alcohols in the presence of acid to form esters (Fischer esterification). Direct formation of amides from carboxylic acids is not feasible due to the poor leaving ability of hydroxide; activation is required.
Fischer esterification: Carboxylic acid + alcohol + acid catalyst → ester + water
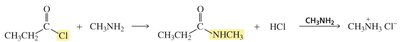
Amide formation: Requires activation of the acid (e.g., conversion to acyl chloride)
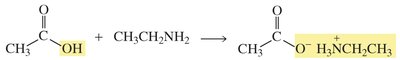
Reactions of Amides
Hydrolysis of Amides
Amides are the least reactive of the carboxylic acid derivatives and require strong acid or base for hydrolysis to occur, yielding carboxylic acids or their salts.
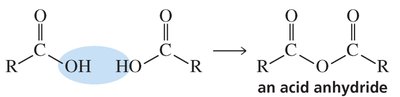
Anhydrides
Preparation and Reactivity
Anhydrides are formed by the condensation of two carboxylic acid molecules with the elimination of water. They are more reactive than esters but less reactive than acyl chlorides, and can react with alcohols, water, and amines to form esters, acids, and amides, respectively.
Activation of Carboxylic Acids
Laboratory Methods
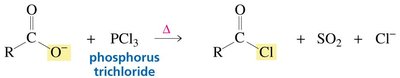
Carboxylic acids can be activated for nucleophilic substitution by conversion to acyl chlorides using reagents such as phosphorus trichloride (PCl3) or thionyl chloride (SOCl2). The resulting acyl chloride can then react with nucleophiles to form amides, esters, or other derivatives.
Biochemical Activation and Relevance
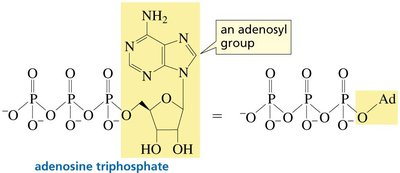
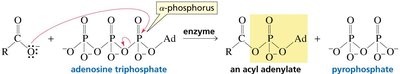
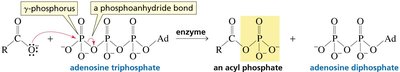
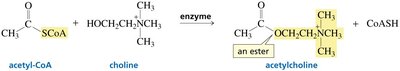
In biological systems, carboxylic acids are often activated as thioesters or mixed anhydrides (e.g., acyl phosphates) to facilitate biosynthetic reactions. Notably, ATP can activate carboxylic acids via formation of acyl adenylates or acyl phosphates. Thioesters, such as acetyl-CoA, are key intermediates in metabolism due to their high reactivity.
Thioesters: More reactive than esters; thiols are better leaving groups than alcohols.
Phosphoric anhydrides: Found in ATP, which can activate carboxylic acids for biosynthesis.

Summary Table: Reactivity of Carboxylic Acid Derivatives
Derivative | General Formula | Leaving Group | Relative Reactivity |
|---|---|---|---|
Acyl chloride | RCOCl | Cl- | Most reactive |
Acid anhydride | (RCO)2O | RCOO- | High |
Ester | RCOOR' | RO- | Moderate |
Carboxylic acid | RCOOH | OH- | Moderate |
Amide | RCONH2 | NH2- | Least reactive |
Additional info: The content above integrates mechanistic details, resonance effects, and biological relevance to provide a comprehensive overview suitable for college-level organic chemistry students.
