 Back
BackChain-Growth Polymerisation: Anionic, Coordination, and Living Polymerisation
Study Guide - Smart Notes

Chain-Growth Polymerisation
Anionic Chain Polymerisation
Anionic chain polymerisation is a type of chain-growth polymerisation where the active center of the growing polymer chain is a carbanion. This process is widely used for the synthesis of polymers with well-defined structures and properties.
Initiation: Initiated by basic or nucleophilic species, such as strong bases (e.g., n-butyllithium), weak bases, or organometallic compounds. The choice of initiator and solvent is crucial for controlling the polymerisation.
Propagation: The carbanion adds to the monomer, typically one with electron-withdrawing groups (EWG) that stabilize the negative charge via resonance or inductive effects. Examples include cyano, carboxyl, vinyl, or phenyl groups.
Transfer Reactions: Can occur via intermolecular or intramolecular nucleophilic substitution, potentially leading to cyclisation or chain re-initiation.
Termination: Usually occurs by reaction with protic impurities (e.g., water or alcohols) or by spontaneous processes such as hydride elimination. The stability of the carbanion and the counter-cation (e.g., Li+, Na+, K+) influences the termination pathway.
Applications: Used to synthesize thermoplastic elastomers such as styrene-butadiene-styrene (SBS).


Effect of Solvent in Anionic Polymerisation
The solvent plays a significant role in anionic polymerisation by affecting ion-pair dissociation, reaction rate, and carbanion stability. Solvents with higher dielectric constants promote free ion-pair formation, increasing the reaction rate. Ethers and hydrocarbons are commonly used, but solvents must be non-protic and non-electrophilic.
Solvent | Dielectric Constant (ε) | Relative Rate (kp) |
|---|---|---|
Benzene | 2.2 | 1 |
Dioxane | 2.2 | 2.5 |
Tetrahydrofuran (THF) | 7.6 | 250 |
1,2-dimethoxyethane | 5.5 | 1900 |
Additional info: The choice of counter-cation (Li+ to Cs+) also affects the polymerisation rate and selectivity.
Coordination Polymerisation and Polyolefins
Coordination Polymerisation
Coordination polymerisation involves the use of metal catalysts (e.g., Ziegler-Natta, Phillips, metallocenes) to control the polymerisation of olefins. The polymer chains are coordinated to a cationic metal center, allowing for high activity, controlled side reactions, and precise control over polymer properties such as tacticity and molar mass.
Ziegler-Natta Catalysts: Typically Ti/Al systems used for the production of high-density polyethylene (HDPE) and polypropylene.
Phillips Catalysts: Chromium oxide/silica systems used for ethylene polymerisation.
Metallocenes: Group 4 metal complexes offering fine control over polymer microstructure.
Polyolefins, such as polyethylene and polypropylene, are among the most produced polymers globally, accounting for nearly half of all plastic production.
Types of Polyethylene
Low-Density Polyethylene (LDPE): Branched structure, made by radical polymerisation, moderate crystallinity, Tg ≈ -120°C, Tm ≈ 105-115°C.
High-Density Polyethylene (HDPE): Linear structure, made using coordination catalysts, high crystallinity, Tm ≈ 135°C.
Linear Low-Density Polyethylene (LLDPE): Linear copolymer with α-olefins, made using coordination catalysts, properties similar to LDPE but with controlled comonomer incorporation.
Living Polymerisation
Principles of Living Polymerisation
Living polymerisation is a chain-growth process where chain-breaking reactions (termination and irreversible transfer) are absent or reversible and fast. This allows for precise control over polymer molar mass, narrow dispersity, and the ability to synthesize block copolymers.
Predictable Molar Mass: The degree of polymerisation (DP) is determined by the ratio of consumed monomer to the number of chains:
Narrow Dispersity: Achieved by fast initiation so all chains start growing simultaneously.
End-Group Fidelity: Enables functionalisation and block copolymer synthesis.
Types: Includes radical (ATRP, RAFT), anionic, and metal-catalysed ring-opening polymerisations.
Anionic Living Polymerisation
Anionic living polymerisation is easier to achieve than radical or cationic living polymerisation due to the absence of termination by coupling. Careful control of solvent, counter-cation, temperature, and purity is required. Industrially, it is used for the production of thermoplastic elastomers such as SBS.
Ring-Opening Polymerisation (ROP)
Overview of ROP
Ring-opening polymerisation is a chain-growth process that converts cyclic monomers (ethers, esters, carbonates, etc.) into linear polymers. ROP can be catalysed by anionic, cationic, radical, metal, or even enzymatic systems. It is important for the synthesis of sustainable and biodegradable polymers, as many monomers can be bio-based.



Case Study: Poly(lactic acid) (PLA)
PLA is a bio-based, industrially compostable polymer obtained by the ROP of lactide (a dimer of lactic acid). It is widely used for sustainable packaging and biomedical applications.
Tg: ~60°C
Tm: ~160°C (up to 220°C for stereocomplex PLA)
Structure: Isotactic PLA (PLLA) is crystalline


Metal-Catalysed ROP
Industrial production of PLA uses Sn(Oct)2 as a catalyst, following a coordination-insertion mechanism. This allows for living polymerisation, resulting in well-defined polymers with controlled molar mass and dispersity.
Superabsorbent Polymers (SAPs)
Cross-Linking and Structure
Superabsorbent polymers, such as cross-linked polyacrylates, are capable of absorbing and retaining large amounts of water. They are synthesized using cross-linkers (e.g., N,N'-methylenebisacrylamide) and initiators (e.g., VA-044) in radical polymerisation processes.
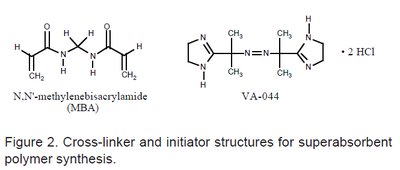
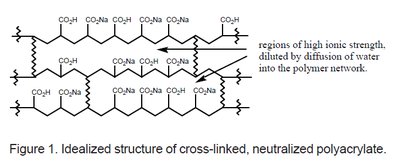
The cross-linked network structure allows for regions of high ionic strength, which are diluted by water diffusion into the polymer network, enabling high water absorption capacity.
Summary Table: Types of Chain-Growth Polymerisation
Type | Initiator/Catalyst | Monomer Type | Key Features |
|---|---|---|---|
Anionic | Strong/weak bases, organometallics | EWG-substituted alkenes | High control, living possible |
Radical | Radical initiators (e.g., AIBN) | Vinyl monomers | Versatile, less control |
Cationic | Acids, Lewis acids | Electron-rich alkenes | Less common, sensitive to impurities |
Coordination | Metal catalysts (e.g., ZN, metallocene) | Olefins | High molar mass, tacticity control |
ROP | Metal, anionic, cationic, radical | Cyclic monomers | Access to biodegradable polymers |
