 Back
BackChapter 1: The Basics of Organic Chemistry – Structure, Bonding, and Molecular Geometry
Study Guide - Smart Notes

Chapter 1: The Basics of Organic Chemistry
Introduction to Organic Chemistry
Organic chemistry is the study of carbon-containing compounds, which typically also include hydrogen and may contain other elements such as nitrogen, oxygen, phosphorus, sulfur, and halogens (collectively called heteroatoms). The presence and arrangement of these atoms define the structure, reactivity, and function of organic molecules.
Functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules (e.g., alcohol groups).
Organic compounds are classified and named based on their functional groups and molecular structure.

Atomic Structure and the Periodic Table
Atoms are the fundamental units of elements, distinguished by their atomic number (Z), which is the number of protons in the nucleus. Compounds are formed by the combination of elements in fixed proportions.
The periodic table organizes elements by increasing atomic number and similar chemical properties.
Groups (columns) indicate elements with similar valence electron configurations, while periods (rows) indicate increasing energy levels.
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons, resulting in different mass numbers but identical chemical properties.
Examples: 12C, 13C, 14C (all have 6 protons, but 6, 7, and 8 neutrons, respectively).
Hydrogen isotopes: 1H (protium), 2H (deuterium, D), 3H (tritium, T).
Some isotopes are radioactive (e.g., tritium, 14C).

Valence Electrons and Bonding
The valence shell is the outermost electron shell of an atom, and the electrons in this shell (valence electrons) are involved in chemical bonding. The number of valence electrons typically equals the group number for main-group elements.

The Structural Theory of Organic Chemistry
Atoms form covalent bonds to achieve stable electron configurations. The typical valency (number of bonds formed) for common elements in organic chemistry is:
Carbon (C): tetravalent (forms 4 bonds)
Oxygen (O): divalent (forms 2 bonds)
Hydrogen (H): monovalent (forms 1 bond)
Halogens (e.g., Br, Cl): monovalent
Never draw more than four bonds to carbon in neutral organic molecules.
Isomerism
Constitutional isomers are compounds with the same molecular formula but different connectivity of their atoms. For example, C4H10O can be 1-butanol or diethyl ether.


Chemical Bonds: Ionic and Covalent
Chemical bonds are the forces holding atoms together in compounds:
Ionic bonds: Formed by the transfer of electrons from one atom to another, resulting in oppositely charged ions (e.g., Na+Cl-).
Covalent bonds: Formed by the sharing of electrons between atoms, typically between atoms of similar electronegativity.

The Octet Rule and Exceptions
Atoms tend to form bonds to achieve a filled valence shell (usually 8 electrons for main-group elements, the "octet rule"). However, there are exceptions, especially for elements in the third period and beyond, which can have expanded octets due to available d orbitals (e.g., SiF62-).
Electronegativity
Electronegativity (EN) is the ability of an atom to attract electrons in a bond. Fluorine is the most electronegative element (EN = 4.0), while cesium is among the least (EN = 0.7). The difference in electronegativity between atoms influences bond polarity and reactivity.
How to Write Lewis Structures
Lewis structures represent molecules by showing all valence electrons as dots (lone pairs) or lines (bonding pairs). All atoms must have appropriate numbers of bonds and lone pairs, and formal charges should be indicated where necessary.
All electrons must be shown.
Formal charge calculation: Where F = formal charge, Z = number of valence electrons, S = number of shared electrons, U = number of unshared electrons.
Resonance Theory
Some molecules can be represented by two or more valid Lewis structures, called resonance structures. The actual molecule is a hybrid of these structures, with electrons delocalized over the molecule. Resonance increases stability, and all resonance structures must be valid Lewis structures with the same arrangement of atoms.
Quantum Mechanics and Atomic Structure
Electrons in atoms are described by wave functions (orbitals), which define the probability distribution of finding an electron in a given region of space. The main types of atomic orbitals are s, p, d, and f, each with characteristic shapes and energies.
Aufbau principle: Electrons fill the lowest energy orbitals first.
Pauli exclusion principle: Each orbital can hold a maximum of two electrons with opposite spins.
Hund’s rule: Electrons occupy degenerate orbitals singly before pairing up.

Molecular Orbitals
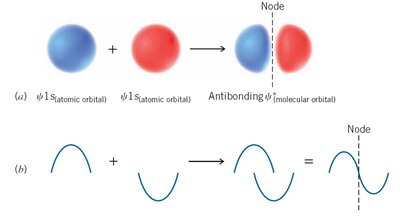
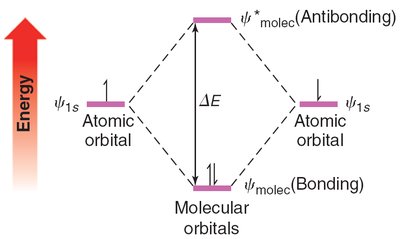
When atomic orbitals combine during bond formation, they create molecular orbitals. The number of molecular orbitals formed equals the number of atomic orbitals combined. There are two main types:
Bonding molecular orbitals (\psi_{molec}): Lower in energy, result from constructive interference of atomic orbitals.
Antibonding molecular orbitals (\psi^*_{molec}): Higher in energy, result from destructive interference.
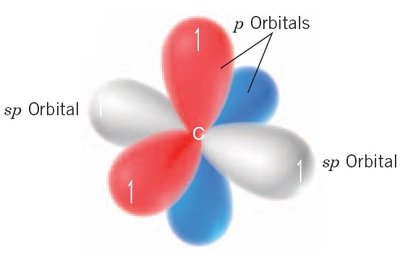
Hybridization
Atomic orbitals can mix to form hybrid orbitals that are used in bonding. The main types are:
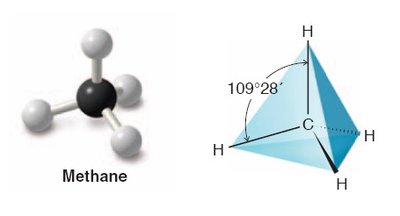
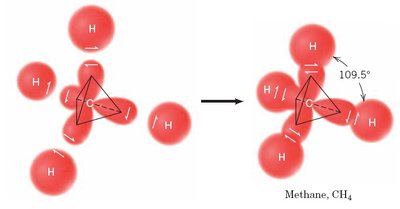
sp3 hybridization: Mixing one s and three p orbitals, forming four equivalent orbitals arranged tetrahedrally (109.5° bond angles), as in methane (CH4).
sp2 hybridization: Mixing one s and two p orbitals, forming three orbitals in a trigonal planar arrangement (120° bond angles), as in ethene (C2H4).
sp hybridization: Mixing one s and one p orbital, forming two orbitals in a linear arrangement (180° bond angles), as in acetylene (C2H2).

Sigma and Pi Bonds
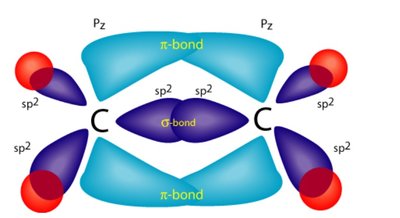
Sigma (\sigma) bonds are formed by head-on overlap of orbitals and are symmetrical around the bond axis. Pi (\pi) bonds are formed by side-on overlap of p orbitals and are not symmetrical around the bond axis. Double and triple bonds contain one sigma bond and one or two pi bonds, respectively.
Molecular Geometry and VSEPR Theory
The Valence Shell Electron Pair Repulsion (VSEPR) model predicts the shapes of molecules based on the repulsion between electron pairs (bonding and nonbonding) around a central atom. Nonbonding pairs repel more strongly than bonding pairs, affecting bond angles.
Methane (CH4): Tetrahedral, 109.5° bond angles.
Ammonia (NH3): Trigonal pyramidal, 107° bond angles due to one lone pair.
Boron trifluoride (BF3): Trigonal planar, 120° bond angles.
Beryllium hydride (BeH2): Linear, 180° bond angle.
Structural Formulas in Organic Chemistry
Organic molecules can be represented in several ways:
Dash structural formulas: Show all atoms and bonds explicitly.
Condensed structural formulas: Group atoms together to simplify the structure.
Bond-line (skeletal) formulas: Show the carbon backbone as lines, omitting hydrogen atoms bonded to carbon for simplicity.
Three-dimensional (wedge-dash) formulas: Indicate the spatial arrangement of atoms using wedges and dashes.
Summary Table: Types of Bonds and Hybridization
Bond Type | Orbitals Involved | Geometry | Example |
|---|---|---|---|
sp3 | 1 s + 3 p | Tetrahedral (109.5°) | Methane (CH4) |
sp2 | 1 s + 2 p | Trigonal planar (120°) | Ethene (C2H4) |
sp | 1 s + 1 p | Linear (180°) | Acetylene (C2H2) |
Additional info: This chapter provides foundational concepts for understanding organic molecules, their bonding, structure, and representation, which are essential for all subsequent topics in organic chemistry.
