 Back
BackChapter 19: Amines – Structure, Properties, and Reactions
Study Guide - Smart Notes

Chapter 19: Amines
Introduction to Amines
Amines are organic compounds derived from ammonia (NH3) by replacement of one or more hydrogen atoms with alkyl or aryl groups. They are important in biological systems and industrial applications, and are characterized by their basicity and nucleophilicity.
Odor: Many amines, especially lower molecular weight ones, have a characteristic fishy smell due to the presence of volatile amines such as trimethylamine.
Occurrence: Amines are found in proteins, vitamins, alkaloids, and neurotransmitters.

Classification and Structure of Amines
Classes of Amines
Amines are classified based on the number of organic substituents attached to the nitrogen atom:
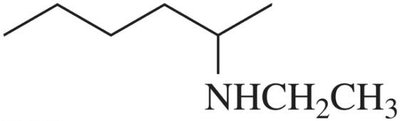
Primary (1°) amine: One alkyl or aryl group attached (RNH2).
Secondary (2°) amine: Two alkyl or aryl groups attached (R2NH).
Tertiary (3°) amine: Three alkyl or aryl groups attached (R3N).
Quaternary (4°) ammonium salt: Four organic groups attached; nitrogen bears a positive charge (R4N+).
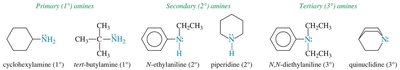
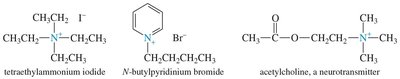
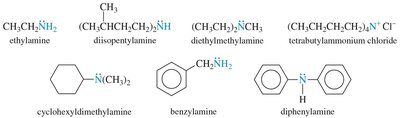
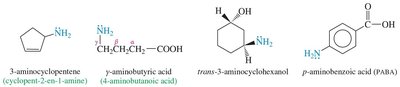
Nomenclature of Amines
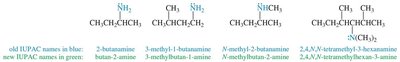
Amines can be named using common or IUPAC nomenclature. The common name is derived from the names of the alkyl groups followed by the suffix '-amine'. For IUPAC names, the longest carbon chain is chosen as the parent, and the '-e' of the alkane is replaced with '-amine'.
As a substituent: When a higher-priority functional group is present, the amine is named as an 'amino-' substituent.
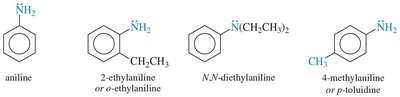
Aromatic amines: Amines attached to aromatic rings are named as derivatives of aniline.
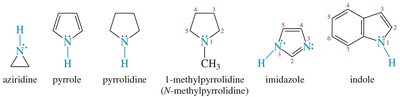
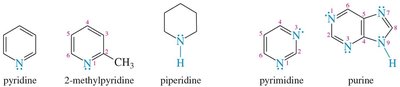
Heterocyclic amines: For cyclic amines, the nitrogen is assigned position 1.
Structure and Stereochemistry of Amines
Hybridization and Geometry
The nitrogen atom in amines is typically sp3 hybridized, resulting in a trigonal pyramidal geometry with a lone pair of electrons. The bond angle is slightly less than 109.5° due to the lone pair repulsion.
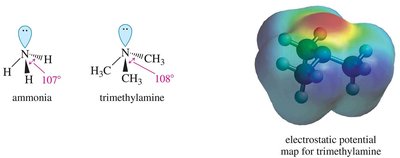
Chirality and Inversion
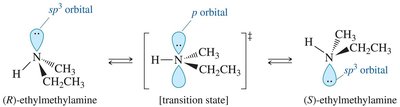
Amines with three different substituents and a lone pair are technically chiral, but rapid inversion at nitrogen (umbrella-like motion) prevents isolation of enantiomers. However, quaternary ammonium salts with four different groups can be chiral and resolved into enantiomers, as inversion is not possible without a lone pair.
Cyclic Amines and Chirality
In small rings, nitrogen inversion is hindered, allowing for the resolution of chiral amines.
Physical Properties of Amines
Boiling Points and Hydrogen Bonding
Amines can form hydrogen bonds, but these are weaker than those in alcohols due to the lower electronegativity of nitrogen compared to oxygen. As a result, amines have lower boiling points than corresponding alcohols. Tertiary amines, lacking N–H bonds, cannot hydrogen bond with themselves and thus have the lowest boiling points among amines of similar molecular weight.
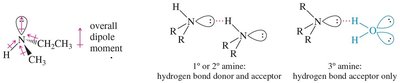
Solubility and Odor
Small amines (fewer than six carbons) are soluble in water due to their ability to accept hydrogen bonds. Branching increases solubility. Most amines have a characteristic fishy odor, especially those with low molecular weight.
Chemical Properties of Amines
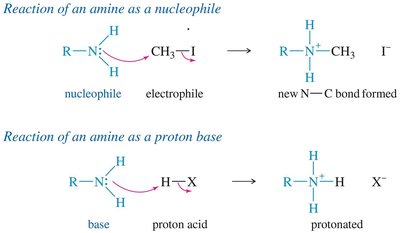
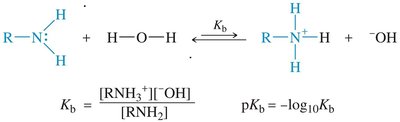
Basicity of Amines
The lone pair of electrons on nitrogen makes amines basic; they can accept a proton from acids. Alkyl amines are generally stronger bases than ammonia due to electron-donating alkyl groups. The basicity is measured by the base-dissociation constant (Kb):
Equation:
Base-dissociation constant:
pKb:
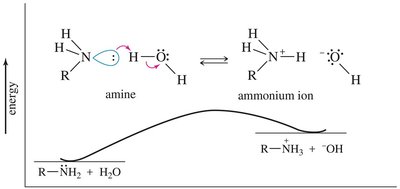
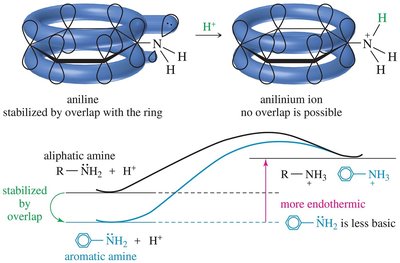
Resonance and Hybridization Effects
Resonance delocalization of the nitrogen lone pair (as in aniline or pyrrole) decreases basicity. Hybridization also affects basicity: sp3 hybridized nitrogens (as in aliphatic amines) are more basic than sp2 (as in pyridine) or sp (as in nitriles).
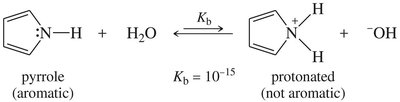
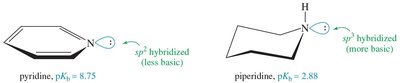
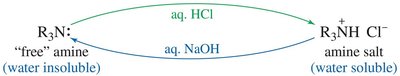
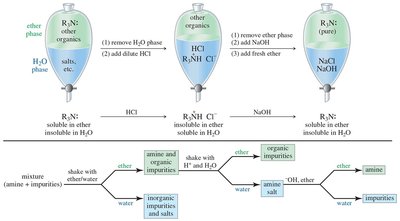
Ammonium Salts and Separation
Quaternary ammonium salts are ionic, have high melting points, and are water-soluble. Amines can be separated and purified based on their acid-base properties by converting them to water-soluble salts and back to free amines.
Spectroscopic Properties of Amines
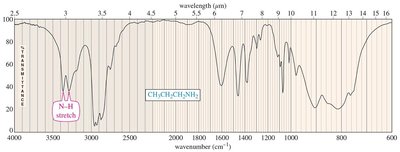
Infrared (IR) Spectroscopy
Amines show N–H stretching vibrations between 3200 and 3500 cm–1. Primary amines display two peaks, while secondary amines show one.
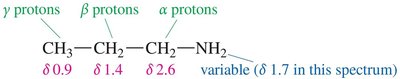
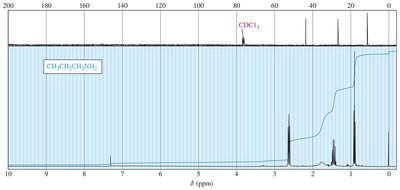
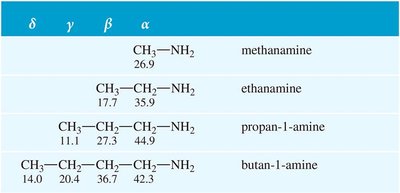
Nuclear Magnetic Resonance (NMR) Spectroscopy
Protons on the α-carbon of amines are less deshielded than those adjacent to oxygen. The chemical shifts for α, β, and γ protons can be used to identify amines.
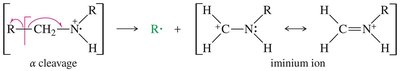
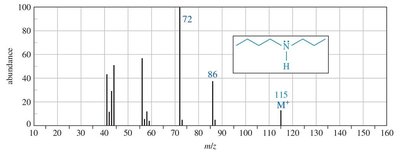
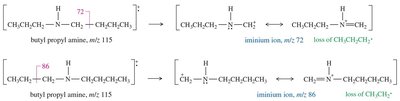
Mass Spectrometry
The presence of nitrogen in a molecule leads to an odd molecular ion mass. Amines commonly undergo α-cleavage, producing an iminium ion.
Chemical Reactions of Amines
Reaction with Ketones and Aldehydes
Amines react with carbonyl compounds to form imines, oximes, or hydrazones, depending on the nitrogen nucleophile. The reaction involves nucleophilic attack, formation of a tetrahedral intermediate, and elimination of water.

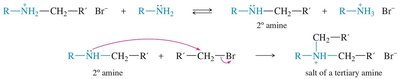
Alkylation of Amines
Amines can be alkylated by alkyl halides via the SN2 mechanism. Multiple alkylations can occur, leading to mixtures. Exhaustive alkylation with excess alkyl halide yields quaternary ammonium salts.
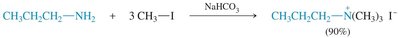
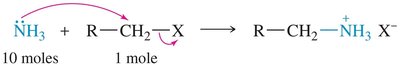
Acylation of Amines
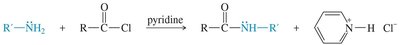
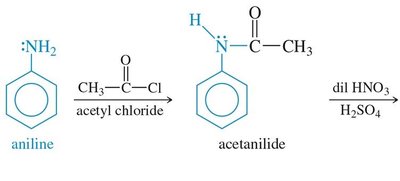
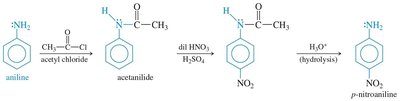
Primary and secondary amines react with acid chlorides to form amides via nucleophilic acyl substitution. This reaction is important for the synthesis of amides and for protecting amine groups in synthesis.
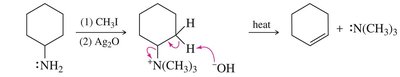
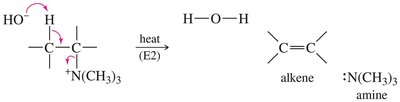
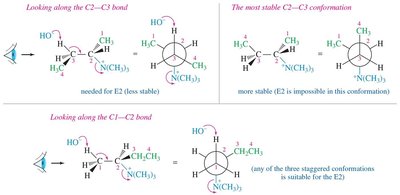
Hofmann Elimination
Quaternary ammonium salts undergo Hofmann elimination upon heating with base, yielding the least substituted alkene (Hofmann product) via an E2 mechanism.
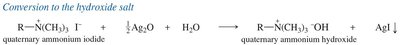
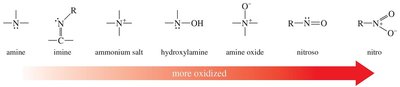
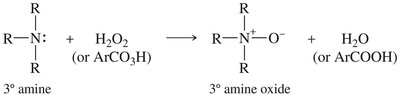
Oxidation of Amines
Amines are easily oxidized. Secondary amines yield hydroxylamines, tertiary amines yield amine oxides, and primary amines can be further oxidized to nitroso or nitro compounds. Amine oxides can undergo elimination (Cope elimination) to form alkenes.
Synthesis of Amines
Reductive Amination
Reductive amination is a general method for synthesizing primary, secondary, and tertiary amines by condensation of ammonia or an amine with a carbonyl compound, followed by reduction of the resulting imine or iminium ion.
Reducing agents: or
Acylation–Reduction
Acylation of an amine with an acid chloride forms an amide, which can be reduced to an amine using . This method prevents over-alkylation and allows for the stepwise synthesis of higher amines.
Gabriel Synthesis
The Gabriel synthesis is used to prepare primary amines by alkylation of phthalimide followed by hydrazinolysis.
Reduction of Azides, Nitriles, and Nitro Compounds
Azides: Reduced to amines by catalytic hydrogenation or .
Nitriles: Reduced to primary amines by or .
Nitro compounds: Reduced to amines by catalytic hydrogenation or active metals and acid.
Additional info: This guide covers the structure, nomenclature, physical and chemical properties, spectroscopic identification, and synthetic methods for amines, as well as their reactions and applications in organic chemistry.
