 Back
BackChapter 6: An Overview of Organic Reactions – Electrophiles, Nucleophiles, Polar and Radical Mechanisms
Study Guide - Smart Notes

Organic Reaction Fundamentals
Electrophiles and Nucleophiles
Organic reactions often involve the interaction between electrophiles and nucleophiles. Electrophiles are "electron-loving" species with a positively polarized, electron-poor atom, capable of accepting a pair of electrons. Nucleophiles are electron-rich species that donate electron pairs to electrophiles. The distinction between these two is fundamental to understanding reaction mechanisms.
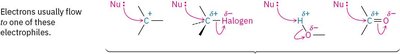
Electrophile: An atom or molecule that accepts an electron pair (Lewis acid).
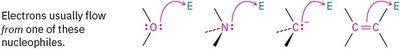
Nucleophile: An atom or molecule that donates an electron pair (Lewis base).
Charge: Electrophiles can be neutral or positively charged; nucleophiles can be neutral or negatively charged.
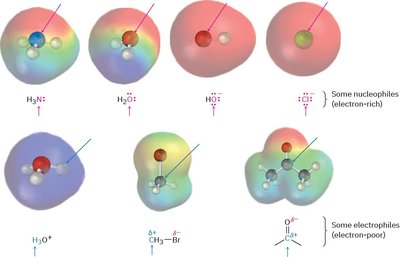
Example: Ammonia (NH3), water (H2O), and chloride ion (Cl-) are common nucleophiles, while hydronium ion (H3O+), methyl bromide (CH3Br), and carbonyl compounds are typical electrophiles.
Polar Reactions and Addition Mechanisms
Accessibility of π Electrons in Alkenes
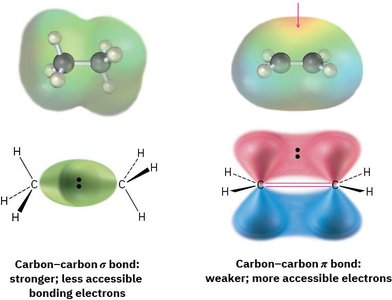
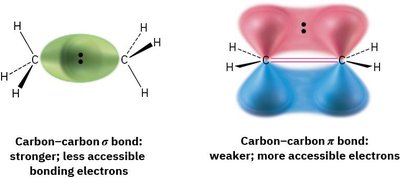
Alkenes contain π bonds whose electrons are located above and below the plane of the molecule, making them more accessible to reactants than σ bonds. This accessibility makes alkenes nucleophilic and prone to reactions with electrophiles.
σ Bond: Stronger, less accessible bonding electrons.
π Bond: Weaker, more accessible electrons, leading to higher reactivity.
Addition of HBr to Ethylene: Mechanism
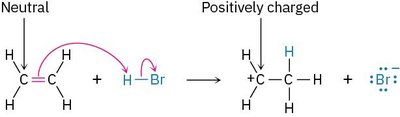
The addition of HBr to ethylene is a classic example of a polar reaction. The π electrons of ethylene attack the electrophilic hydrogen of HBr, forming a carbocation intermediate, which is then attacked by the bromide ion.
Step 1: π electrons from ethylene attack H+ from HBr, forming a carbocation and Br-.
Step 2: Br- donates an electron pair to the carbocation, forming bromoethane.
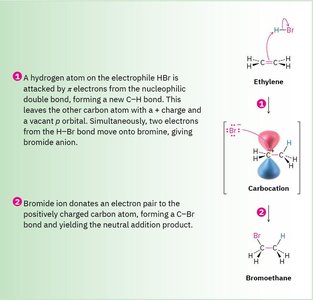
Example: The formation of bromoethane from ethylene and HBr demonstrates the typical electrophile–nucleophile interaction.
Curved Arrow Notation in Polar Reaction Mechanisms
Rules for Electron Movement
Curved arrows are used to depict the movement of electron pairs in reaction mechanisms. Understanding these rules is essential for writing and interpreting organic reaction mechanisms.
Rule 1: Electrons move from a nucleophilic source to an electrophilic sink. The nucleophile must have an available electron pair.
Rule 2: Nucleophiles can be neutral or negatively charged. If negatively charged, the donating atom becomes neutral after electron transfer.
Rule 3: Electrophiles can be neutral or positively charged. If positively charged, the accepting atom becomes neutral after electron transfer.
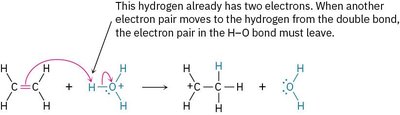
Rule 4: The octet rule must be followed; no second-row atom can have more than eight electrons. If an electron pair moves to an atom with an octet, another pair must leave to maintain the octet.
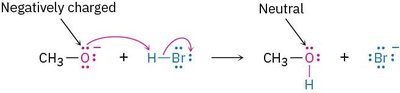
Example: The reaction of methoxide ion (CH3O-) with HBr illustrates the change from negatively charged to neutral species.
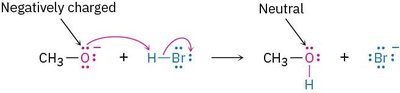
Example: The reaction of ethylene with HBr shows the formation of a carbocation intermediate.

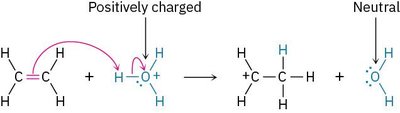
Example: The reaction of ethylene with H3O+ demonstrates the change from positively charged to neutral species.

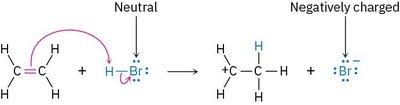
Example: The reaction of ethylene with HBr shows the change from neutral to negatively charged species.

Example: The reaction of ethylene with H3O+ illustrates the octet rule in action.

Radical Reactions
Definition and Properties of Radicals
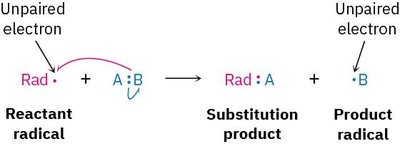
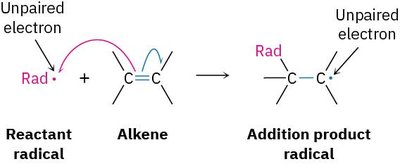
A radical is a highly reactive species containing an atom with an odd number of electrons (usually seven) in its valence shell, rather than a noble-gas octet. Radical reactions are important in both industrial and biological contexts.
Radical: An atom or molecule with an unpaired electron.
Reactivity: Radicals are highly reactive due to their electron deficiency.
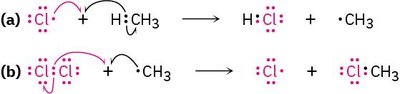
Example: The reaction of chlorine with methane produces chloromethane, a precursor for solvents like dichloromethane and chloroform.
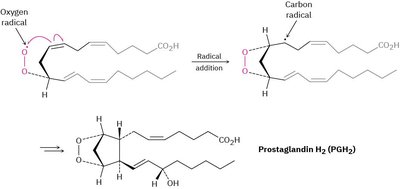
Example: Radical addition reactions are also involved in the biosynthesis of prostaglandins from arachidonic acid.
Describing a Reaction: Equilibria, Rates, and Energy Changes
Chemical Equilibrium
Chemical equilibrium is a state in which the concentrations of reactants and products remain constant over time. The equilibrium constant, Keq, quantifies the ratio of product to reactant concentrations at equilibrium.
Equilibrium Constant:
Interpretation: If is near 1, both reactants and products are present in appreciable amounts at equilibrium.
Example: The equilibrium constant is used to predict the direction and extent of chemical reactions.
