 Back
BackChapter 9: Alkynes – Structure, Properties, Synthesis, and Reactions
Study Guide - Smart Notes

Alkynes: Structure, Properties, Synthesis, and Reactions
Introduction to Alkynes
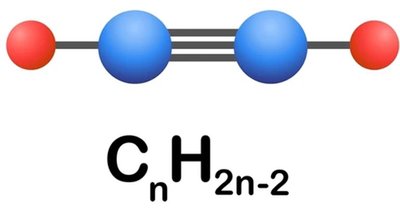
Alkynes are hydrocarbons that contain at least one carbon–carbon triple bond. They are an important class of unsaturated organic compounds, exhibiting unique chemical and physical properties due to the presence of the triple bond.
General Formula: Noncyclic alkynes have the molecular formula .
Simplest Alkyne: Acetylene (ethyne, ) is the simplest member of this family.
Functional Group: The carbon–carbon triple bond is the defining functional group of alkynes, and it reacts with many of the same reagents as alkenes.
Classification of Alkynes
Alkynes can be classified based on the position of the triple bond and the symmetry of the molecule.
Terminal Alkynes: The triple bond is at the end of the carbon chain (e.g., HC≡C–R).
Internal Alkynes: The triple bond is between two carbon atoms within the chain (e.g., R–C≡C–R').
Symmetric Alkynes: Both groups attached to the triple bond are the same.
Asymmetric Alkynes: The groups attached to the triple bond are different.
Cyclic Alkynes: The triple bond is part of a ring structure (rare due to ring strain).
Molecular Structure and Bonding in Alkynes
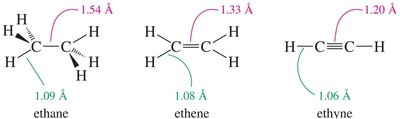
The carbon atoms in a triple bond are sp-hybridized, resulting in a linear geometry and unique bonding characteristics.
Bonding: The triple bond consists of one sigma () bond and two pi () bonds.
Geometry: The bond angle around the triple bond is 180°, making alkynes linear.
Bond Length and Strength: The C≡C bond is shorter and stronger than C=C and C–C bonds due to increased s-character and orbital overlap.

Nomenclature of Alkynes
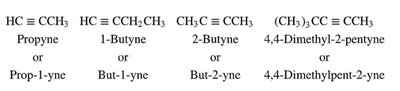
Alkynes are named according to IUPAC rules, similar to alkanes and alkenes, with the suffix -yne indicating the presence of a triple bond.
Number the longest carbon chain containing the triple bond to give the triple bond the lowest possible number.
For multiple triple bonds, use suffixes such as -diyne, -triyne, etc.
If both double and triple bonds are present, number to give the lowest set of locants to the multiple bonds.
The ethynyl group (–C≡CH) is used as a substituent name.
Physical Properties of Alkynes
Alkynes share many physical properties with other hydrocarbons but have some unique features due to their triple bond.
Polarity: Nonpolar and insoluble in water; soluble in most organic solvents.
Boiling Points: Similar to alkanes of comparable molecular weight.
Density: Less dense than water.
State: Alkynes with up to four carbons are gases at room temperature.
Acidity of Alkynes
Terminal alkynes are significantly more acidic than alkenes and alkanes due to the high s-character of the sp-hybridized carbon.
pKa Values: Terminal alkynes (pKa ≈ 25) < Alkenes (pKa ≈ 44) < Alkanes (pKa ≈ 50).
Reason: Greater s-character in sp orbitals holds electrons closer to the nucleus, increasing acidity.
Acetylide Ions: Deprotonation of terminal alkynes forms acetylide ions, which are strong nucleophiles and bases.
Bases Required: Only very strong bases (e.g., NaNH2) can deprotonate terminal alkynes.
Synthesis of Alkynes
Alkylation of Acetylide Ions
Alkynes can be synthesized by alkylation of acetylide ions, which involves the nucleophilic substitution (SN2) of alkyl halides by acetylide anions.
Mechanism: The acetylide ion attacks a primary alkyl halide, displacing the halide and forming a new C–C bond.
Limitation: Only methyl and primary alkyl halides give good yields; secondary and tertiary halides undergo elimination.
Preparation by Elimination Reactions
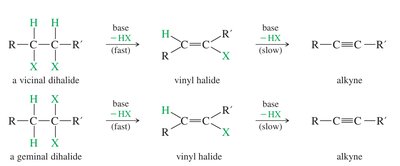
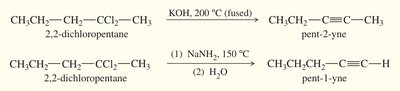
Alkynes can also be prepared by double dehydrohalogenation of dihaloalkanes (geminal or vicinal dihalides).
Geminal Dihalide: Both halogens on the same carbon.
Vicinal Dihalide: Halogens on adjacent carbons.
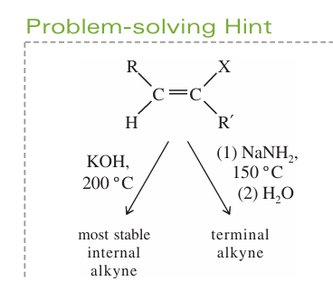
Reagents: Strong bases such as KOH (at high temperature) or NaNH2 (at lower temperature) are used.
Product: KOH tends to give internal alkynes; NaNH2 can yield terminal alkynes.
Reactions of Alkynes
Hydrogenation of Alkynes
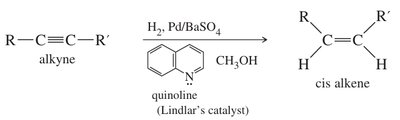
Alkynes undergo catalytic hydrogenation to form alkenes or alkanes, depending on the catalyst and conditions.
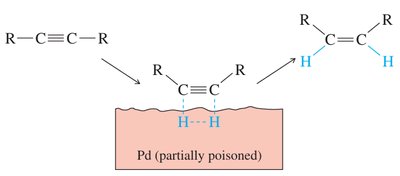
Partial Hydrogenation (cis-alkenes): Lindlar's catalyst (Pd/BaSO4 with quinoline) or Ni2B selectively reduces alkynes to cis-alkenes.
Complete Hydrogenation: Excess H2 with Pd/C or Pt yields alkanes.

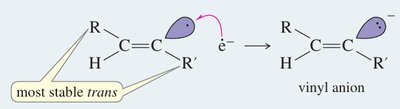
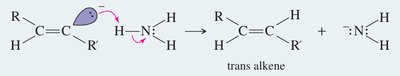
Metal-Ammonia Reduction (trans-alkenes)
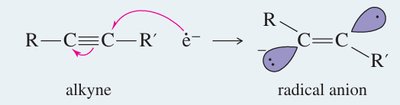
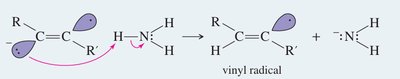
Sodium in liquid ammonia reduces alkynes to trans-alkenes via anti addition of hydrogen atoms.
Mechanism: Involves formation of a radical anion, protonation, and further reduction to yield the trans-alkene.
Conditions: Na/NH3 at low temperature (–33°C).
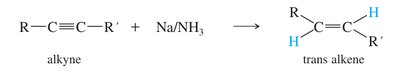
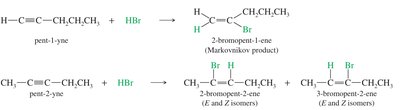
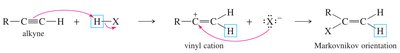
Addition of Hydrogen Halides (HX) to Alkynes
Alkynes react with hydrogen halides to form haloalkenes and, with excess HX, geminal dihalides. The regioselectivity follows Markovnikov's rule for terminal alkynes.
Markovnikov Addition: The halide adds to the more substituted carbon.
Anti-Markovnikov Addition: In the presence of peroxides (ROOR), the addition can proceed with anti-Markovnikov orientation.
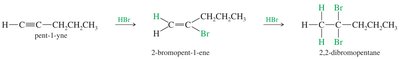

Addition of Halogens to Alkynes
Bromine and chlorine add to alkynes to form dihaloalkenes and, with excess halogen, tetrahaloalkanes. The addition can be syn or anti, often yielding mixtures of isomers.
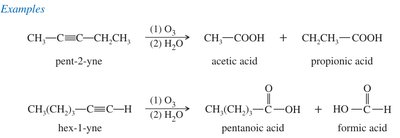
Oxidative Cleavage of Alkynes
Alkynes can be cleaved by ozonolysis or permanganate oxidation to yield carboxylic acids, which is useful for locating the position of the triple bond.
Ozonolysis: cleaves the triple bond, forming two carboxylic acids.
Permanganate Oxidation: Cold, neutral gives diketones; hot, basic $KMnO_4$ cleaves to carboxylic acids.
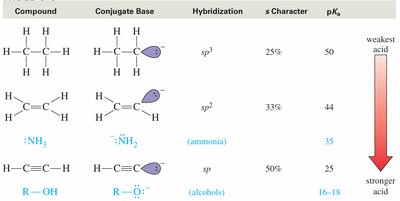
Summary Table: Acidity of Hydrocarbons and Related Compounds
Compound | Conjugate Base | Hybridization | s Character | pKa |
|---|---|---|---|---|
Alkane (R–CH3) | :CH2R | sp3 | 25% | ~50 |
Alkene (R–CH=CH2) | :CH=CHR | sp2 | 33% | ~44 |
Alkyne (HC≡CH) | :C≡CH | sp | 50% | ~25 |
Ammonia (NH3) | :NH2 | sp3 | 25% | ~35 |
Alcohol (R–OH) | R–O– | sp3 | 25% | 16–18 |
Key Concepts and Applications
Alkynes are versatile intermediates in organic synthesis, allowing for chain extension and functional group transformations.
Terminal alkynes are more acidic than other hydrocarbons, enabling the formation of nucleophilic acetylide ions for C–C bond formation.
Hydrogenation and reduction reactions allow for stereoselective synthesis of alkenes from alkynes.
Addition reactions (HX, X2, hydration, hydroboration) provide routes to a variety of functionalized products.
Oxidative cleavage is a diagnostic tool for locating triple bonds in unknown compounds.
Additional info: This guide covers the core concepts, mechanisms, and synthetic applications of alkynes as presented in a typical college-level organic chemistry course.
