 Back
BackCHEM 130: Introduction to Organic Chemistry – Structured Study Notes
Study Guide - Smart Notes

Introduction to Organic Chemistry
Overview and Importance
Organic chemistry is the branch of chemistry that studies carbon compounds and their reactions. Carbon compounds are central to all living things and are found in a wide variety of substances, including drugs, vitamins, plastics, fibers, carbohydrates, proteins, and fats. Organic chemistry has profoundly impacted modern life by improving natural materials and synthesizing new ones for health, comfort, and convenience. Key Points:
Catenation: Carbon atoms bond covalently to form chains and rings, a phenomenon called catenation.
Elements in Organic Molecules: Most organic molecules contain C and H, sometimes O, S, and N.
Applications: Knowledge of organic chemistry is essential for developing drugs, agricultural chemicals, and other life-impacting substances.
Electronic Configuration and Hybridization of Carbon
Electronic Configuration
Carbon has 6 electrons: 2 in the first shell, 4 in the second (valence) shell. The four valence electrons allow carbon to form four covalent bonds, giving it a valency of four. Electronic configuration: $1s^2 2s^2 2p^2$ Orbital shapes: The s orbital is spherical; p orbitals are dumbbell-shaped and oriented in x, y, and z planes.
Hybridization: sp3, sp2, sp
Hybridization is the mixing of atomic orbitals to form new hybrid orbitals suitable for bonding. Carbon can form single, double, or triple bonds depending on the hybridization.
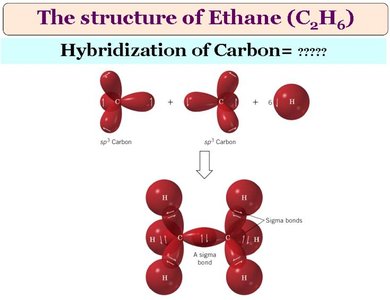
sp3 Hybridization: Four equivalent orbitals arranged tetrahedrally (bond angle ≈ 109.5°). Found in alkanes and saturated compounds.
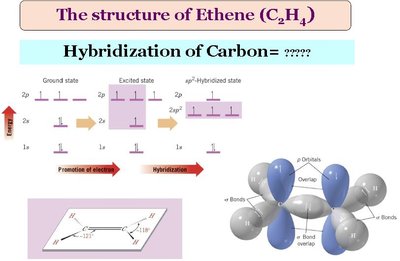
sp2 Hybridization: Three equivalent orbitals in a trigonal planar arrangement (bond angle ≈ 120°), with one unhybridized p orbital forming a π bond. Found in alkenes and aromatic compounds.
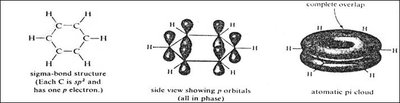
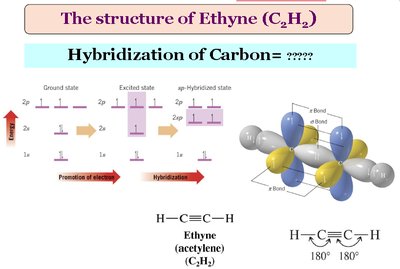
sp Hybridization: Two equivalent orbitals in a linear arrangement (bond angle ≈ 180°), with two unhybridized p orbitals forming two π bonds. Found in alkynes.
Bond Lengths and Angles
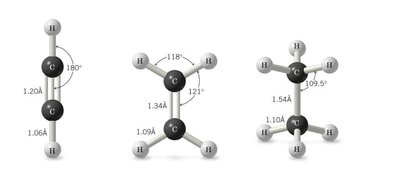
As the s character increases (sp > sp2 > sp3), bond length decreases and bond strength increases.
Typical bond angles: sp3 (109.5°), sp2 (120°), sp (180°).
Elemental Analysis (Qualitative)
Determining Molecular Formula
Empirical formula: Simplest whole-number ratio of atoms.
Molecular formula: Actual number of atoms, determined by molecular weight (often via mass spectrometry).
Example calculation: Given percentages and molecular weight, calculate moles, ratios, empirical formula, and molecular formula.
Hydrocarbons: Classification and Structure
Types of Hydrocarbons
Hydrocarbons are compounds containing only carbon and hydrogen. They are divided into:
Aliphatic hydrocarbons: Open-chain (acyclic) or cyclic structures, including alkanes, alkenes, and alkynes.
Aromatic hydrocarbons: Contain benzene rings; highly stable due to delocalized π electrons.
Alkanes
General Properties
Saturated hydrocarbons with general formula $C_nH_{2n+2}$ (acyclic) or $C_nH_{2n}$ (cyclic).
All carbons are sp3 hybridized.
Members differ by a constant amount (homologous series).
Homologous Series
Examples: Methane (CH4), Ethane (C2H6), Propane (C3H8), etc.
Sources of Alkanes
Petroleum: Primary source; complex mixture of alkanes and aromatics.
Natural gas: Mainly methane, produced by anaerobic oxidation of plant material.
Nomenclature of Alkanes
IUPAC rules: Longest chain is the parent; substituents are named and numbered for lowest possible locants; prefixes (di-, tri-) for multiple identical groups; alphabetical order for different substituents.
Alkyl groups: Formed by removing a hydrogen from an alkane; named by replacing -ane with -yl.
Structural Isomerism
Isomers have the same molecular formula but different atom arrangements.
Example: Butane (C4H10) has two isomers: n-butane and 2-methylpropane.
Physical Properties
C1-C4: Gases; C5-C17: Liquids; C18+: Solids.
Less dense than water; nonpolar; insoluble in water but soluble in nonpolar solvents.
Boiling and melting points increase with molecular size; branching lowers boiling point but can raise melting point if symmetry increases.
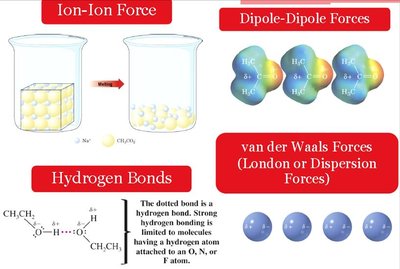
Intermolecular Forces
Van der Waals (London dispersion) forces dominate in alkanes.
Hydrogen bonding occurs in molecules with O, N, or F atoms.
Chemical Reactions of Alkanes
Combustion: $CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O + \text{energy}$
Halogenation: Reaction with halogens to form alkyl halides.
Cracking: Pyrolysis to produce smaller alkanes and alkenes.
Free Radical Substitution: Halogenation proceeds via initiation, propagation, and termination steps.
Cycloalkanes and Geometric Isomerism
Cycloalkanes: Ring structures, formula $C_nH_{2n}$.
Geometric isomerism: Cis/trans isomers based on substituent orientation.
Alkenes
General Properties
Unsaturated hydrocarbons with general formula $C_nH_{2n}$.
Contain C=C double bonds (one sigma, one pi bond); sp2 hybridized.
No free rotation about the double bond; rigid structure.
Nomenclature and Isomerism
Named by replacing -ane with -ene; longest chain with double bond is parent.
Geometric isomerism: Cis/trans and E/Z (Cahn-Ingold-Prelog convention) based on substituent priority.
Physical Properties
C2-C4: Gases; C5-C15: Liquids; >C15: Solids.
Burn with luminous flame; insoluble in water; boiling/melting points increase with size.
Chemical Reactions
Addition reactions: Hydrogenation, halogenation, hydration, hydroboration, polymerization.
Markovnikov's rule: In addition of HX, H adds to the carbon with more hydrogens.
Qualitative tests: Bromine water and Baeyer's test (KMnO4) for unsaturation.
Alkynes
General Properties
Unsaturated hydrocarbons with general formula $C_nH_{2n-2}$.
Contain C≡C triple bonds (one sigma, two pi bonds); sp hybridized.
Nomenclature and Isomerism
Named by replacing -ane with -yne; location of triple bond indicated by number.
Position, structural, and functional isomerism; no geometric isomerism.
Physical Properties
C2-C4: Gases; C5-C13: Liquids; >C13: Solids.
Colorless and odorless (except ethyne); boiling/melting points increase with size.
Chemical Reactions
Addition reactions: Hydrogenation, halogenation, hydration (keto-enol tautomerism).
Reactions with alkylmagnesium halide or alkyllithium: Only terminal alkynes react.
Qualitative tests: Bromine test, Baeyer's test, ammoniacal silver/copper nitrate for terminal alkynes.
Functional Groups in Organic Chemistry
Definition and Importance
A functional group is an atom or group of atoms responsible for the characteristic reactivity and physical properties of an organic compound. The structure of a carbon compound consists of a relatively unreactive hydrocarbon skeleton and a reactive functional group. 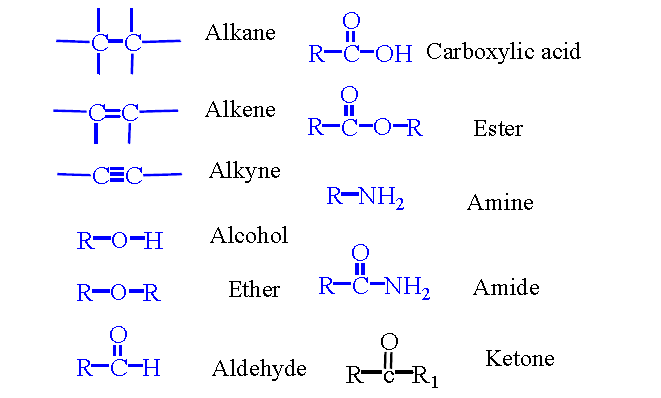
Major Functional Groups
Alcohols (ROH): Classified as primary, secondary, or tertiary based on the carbon bearing the -OH group.
Nomenclature: IUPAC names use the longest chain with -OH, replacing -e with -ol; position of -OH indicated by number.
Properties: Solubility decreases with chain length; high boiling points due to hydrogen bonding.
Qualitative tests: Lucas test (distinguishes 1°, 2°, 3° alcohols), iodoform test (for methyl alcohols).
Carboxylic Acids (R-COOH): Named by replacing -ane with -oic acid; always terminal.
Qualitative test: Reaction with sodium hydrogen carbonate produces CO2 and a precipitate.
Ketones (RCOR): Named by replacing -e with -one; carbonyl carbon gets priority.
Qualitative test: Iodoform test for methyl ketones.
Aldehydes (R-CHO): Named by replacing -e with -al; always terminal.
Qualitative tests: Fehling's test (Cu2+ reduction), Tollens' test (silver mirror).
Amines (R-NH2): Classified as primary, secondary, or tertiary based on number of alkyl groups attached to nitrogen.
Nomenclature: Suffix -amine added to parent chain; locant N used for substituents on nitrogen.
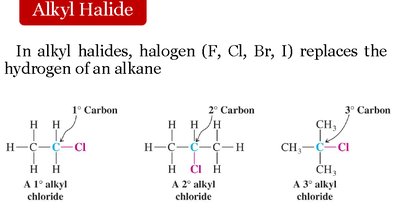
Alkyl Halides (R-X): Halogen replaces hydrogen in an alkane.
Classification: 1°, 2°, 3° based on the carbon to which halogen is attached.
Structural Representations in Organic Chemistry
Condensed, Dash, and Bond-Line Formulas
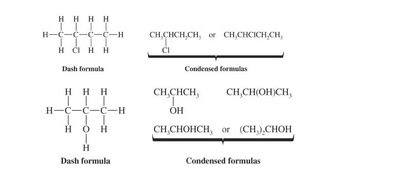
Condensed formulas: Show groups of atoms together (e.g., CH3CH2OH).
Dash formulas: Show all bonds explicitly.
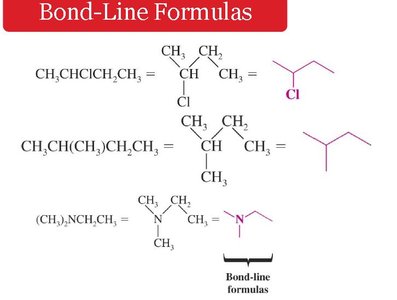
Bond-line formulas: Simplified representation showing only bonds and vertices for carbon atoms.


Summary Table: Hybridization and Bonding in Carbon Compounds
Hybridization | Geometry | Bond Angle | Example |
|---|---|---|---|
sp3 | Tetrahedral | 109.5° | Alkanes (e.g., CH4) |
sp2 | Trigonal planar | 120° | Alkenes (e.g., C2H4) |
sp | Linear | 180° | Alkynes (e.g., C2H2) |
Further Reading
Bruce P. Y.: Organic Chemistry. 5th Ed.
Carey, F: Organic Chemistry. 7th Ed.
Wade J. R. Organic Chemistry. 4th Ed.
Morrison and Boyd: Organic Chemistry. 6th Ed.
Solomons: Organic Chemistry. 4th Ed.
John McMurray: Fundamentals of Organic Chemistry. 7th Ed.
Vollhardt and Schore: Organic Chemistry – Structure and Function. 6th Ed.
Hornback J. M.: Organic Chemistry. 2nd Ed.
