Back
BackConjugated Systems, Orbital Symmetry, and Ultraviolet Spectroscopy
Study Guide - Smart Notes

Chapter 15: Conjugated Systems, Orbital Symmetry, and Ultraviolet Spectroscopy
15.1 Introduction to Diene Classifications
Conjugated systems are fundamental in organic chemistry, especially in the context of dienes, which are hydrocarbons containing two double bonds. The arrangement of these double bonds determines their classification and chemical properties.
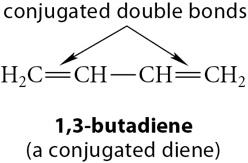
Conjugated Dienes: Double bonds are separated by a single bond, allowing for delocalization of electrons.
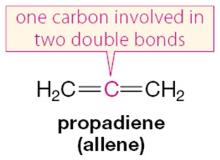
Cumulenes (Allenes): One carbon is involved in two double bonds, resulting in unique bonding and reactivity.
Non-conjugated (Isolated) Dienes: Double bonds are separated by more than one single bond, preventing electron delocalization.
Example: 1,3-butadiene is a classic conjugated diene, while propadiene (allene) is a cumulene.
15.2 Stabilities of Dienes
The stability of dienes is often compared using heats of hydrogenation. Conjugated dienes are more stable than isolated dienes due to electron delocalization.
Heats of Hydrogenation: Lower heat released indicates greater stability.
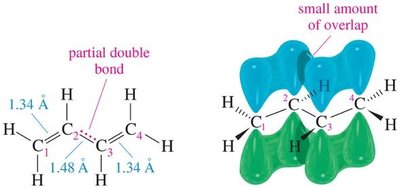
Bond Lengths: Conjugation shortens the bond between the central carbons (e.g., C2–C3 in 1,3-butadiene).
Stability Order: Conjugated dienes > isolated dienes > alkynes.
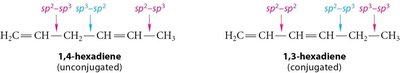
15.3 Molecular Orbital Picture of a Conjugated System
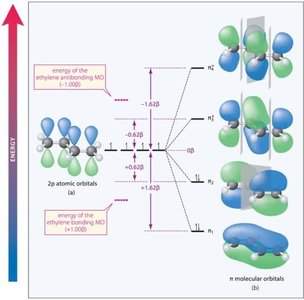
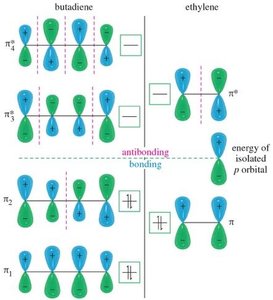
Conjugated systems are best understood through molecular orbital (MO) theory, which explains their stability and reactivity.
Bonding: Increased s-character in bonds shortens bond length and increases strength.
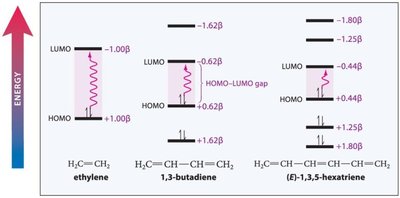
MO Diagram: Conjugated systems have multiple π molecular orbitals, with lower energy than isolated alkenes.
Resonance Stability: The delocalization of π electrons lowers the overall energy of the molecule.
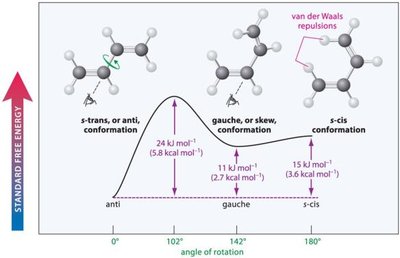
15.3 Conformations of Dienes
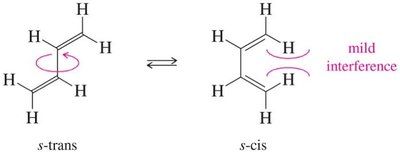
Dienes can adopt different conformations, affecting their stability and reactivity. The s-trans conformation is generally more stable than the s-cis due to reduced steric repulsion.
s-trans Conformation: Most stable due to minimal van der Waals repulsions.
s-cis Conformation: Less stable due to increased steric interference.
Gauche/Skew Conformation: Intermediate stability.
15.4 Allylic Cations
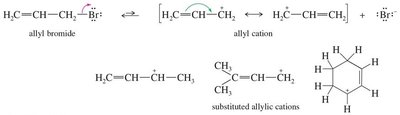
Resonance Stabilization of Allylic Cations
Allylic cations are carbocations where the positive charge is adjacent to a double bond, allowing for resonance stabilization.
Resonance: The charge is delocalized over multiple carbons, increasing stability.
Stability Comparison: 2° allylic cation ≈ 3° carbocation; 1° allylic cation ≈ 2° carbocation.
Example: Allyl bromide forms an allylic cation upon loss of Br-.
15.5 1,2- and 1,4-Addition to Conjugated Dienes
Mechanism and Product Distribution
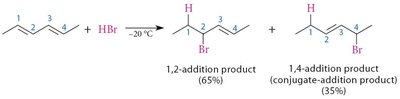
Conjugated dienes can undergo addition reactions at two positions, leading to different products.
1,2-Addition: Occurs at adjacent carbons.
1,4-Addition: Occurs at the terminal carbons of the diene system.
Example: Addition of HBr to buta-1,3-diene yields both 1,2- and 1,4-products.
15.6 Kinetic vs Thermodynamic Control in Addition Reactions
Reaction Conditions and Product Formation
The outcome of addition reactions to conjugated dienes depends on temperature and reaction conditions.
Kinetic Control: At low temperatures, the fastest-forming product (usually 1,2-addition) predominates.
Thermodynamic Control: At higher temperatures, the more stable product (often 1,4-addition) is favored.
Reaction Diagram: Shows the energy profile and product distribution.
15.7 Allylic Radicals and Substitution
Stability and Mechanism
Allylic radicals are stabilized by resonance, and their formation can compete with addition reactions.
Stability Order: 1° < 2° < 3° < 1° allylic radical.
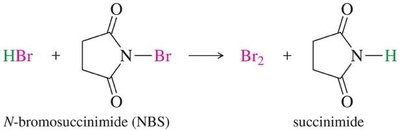
Allylic Bromination: Initiated by light, heat, or peroxides; N-bromosuccinimide (NBS) is used to maintain low Br2 concentration.
Example: NBS reacts with HBr to produce Br2 and succinimide, preventing unwanted addition across the double bond.
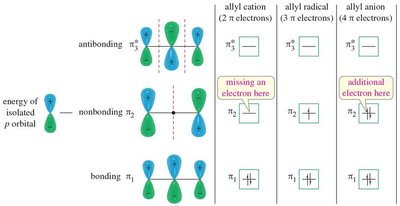
15.8/9 Molecular Orbitals of Allylic Species
Electronic Configurations
The molecular orbitals of allylic cations, radicals, and anions differ based on electron count, affecting their stability and reactivity.
Allylic Cation: 2 π electrons.
Allylic Radical: 3 π electrons.
Allylic Anion: 4 π electrons.
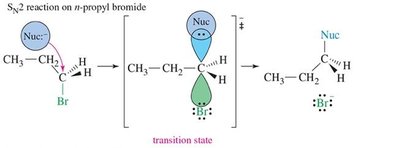
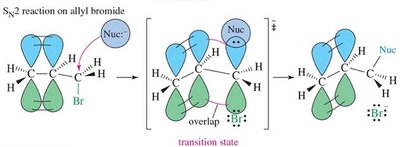
15.10 SN2 Displacement Reactions of Allylic Halides and Tosylates
Reactivity and Mechanism
Allylic halides and tosylates undergo SN2 reactions rapidly due to stabilization of the transition state by conjugation.
Transition State: Stabilized by overlap with π orbitals, lowering activation energy.
Reactivity: Allylic halides react faster than normal alkyl halides.
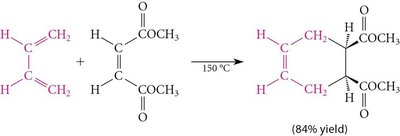
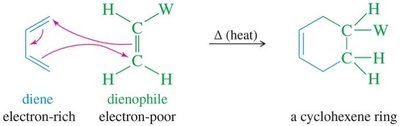
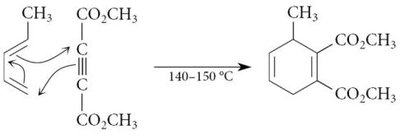
15.11 The Diels-Alder Reaction
Mechanism and Applications
The Diels-Alder reaction is a [4+2] cycloaddition between a conjugated diene and a dienophile, forming a six-membered ring. It is a key method for constructing cyclic compounds in organic synthesis.
Diene: Electron-rich species.
Dienophile: Electron-poor alkene or alkyne.
Product: Cyclohexene or bicyclic compounds.
Example: Reaction of 1,3-butadiene with methyl vinyl ketone.
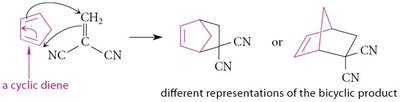
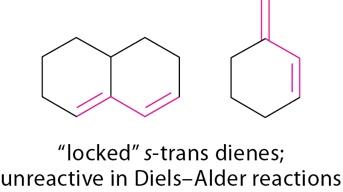
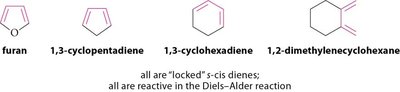
Effect of Diene Conformation
Diene conformation significantly affects reactivity in the Diels-Alder reaction. Dienes locked in s-trans conformation are unreactive, while those locked in s-cis are highly reactive.
s-trans Dienes: Unreactive in Diels-Alder reactions.
s-cis Dienes: Highly reactive.
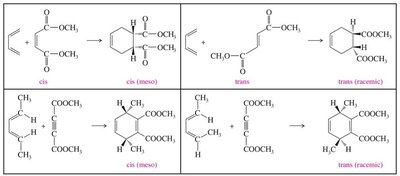
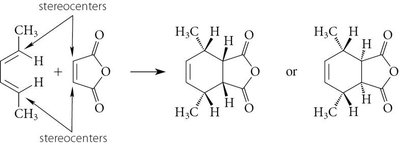
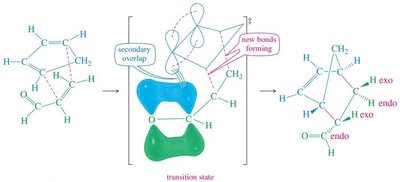
Stereochemistry of the Diels-Alder Reaction
The Diels-Alder reaction is stereospecific, often resulting in syn-addition. The endo rule describes the preference for endo products due to secondary orbital interactions.
Endo Rule: Electron-withdrawing groups on the dienophile interact with the diene's π orbitals.
Product Stereochemistry: Can be endo or exo, depending on the transition state.
15.12 The Diels-Alder as a Pericyclic Reaction
Orbital Symmetry and Conservation
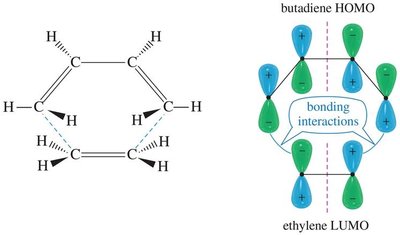
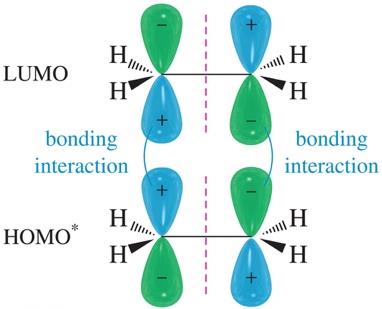
The Diels-Alder reaction is a pericyclic reaction, governed by the conservation of orbital symmetry. Constructive overlap of molecular orbitals stabilizes the transition state.
Woodward-Hoffmann Rules: Predict allowed and forbidden pericyclic reactions.
HOMO-LUMO Interactions: Diene's HOMO interacts with dienophile's LUMO.
[2+2] Cycloaddition: Thermally forbidden due to antibonding overlap, but photochemically allowed.
![Forbidden [2+2] cycloaddition](https://static.studychannel.pearsonprd.tech/study_guide_files/organic-chemistry/sub_images/a12c5e34_image_25.png)
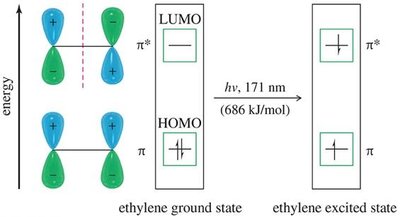
15.13 Ultraviolet Absorption Spectroscopy
Principles and Applications
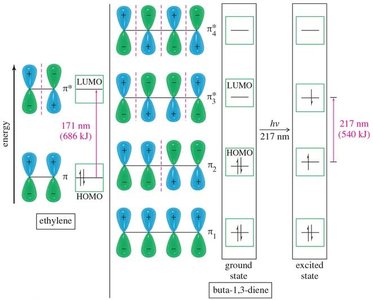
UV-VIS spectroscopy is used to probe the extent of conjugation in organic molecules. Conjugated systems absorb at longer wavelengths due to smaller HOMO-LUMO gaps.
Absorbance (A): Follows Beer’s Law:
λmax: Wavelength at maximum absorption; increases with conjugation.
Chromophore: Structural feature responsible for UV-VIS absorption.
Energy Equation:
Example: Each additional conjugated C=C adds 30–40 nm to λmax; each alkyl group adds ~5 nm.
Estimating λmax and Substituent Effects
Conformational and substituent effects influence the absorption maxima in UV-VIS spectroscopy.
s-cis Conformation: Higher λmax due to increased conjugation.
Alkyl Substituents: Each adds ~5 nm to λmax.
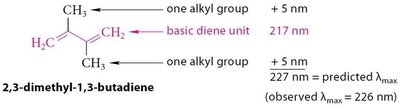
Example: 2,3-dimethyl-1,3-butadiene predicted λmax = 227 nm; observed = 226 nm.
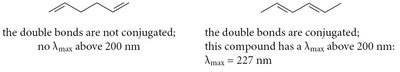
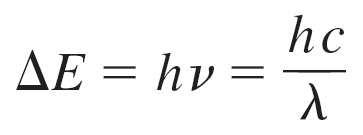
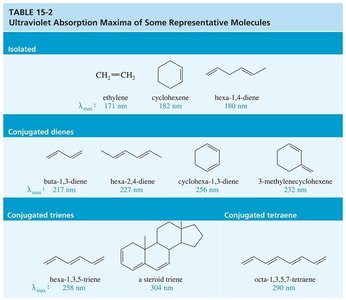
Summary Table: Ultraviolet Absorption Maxima
The following table summarizes UV absorption maxima for representative molecules:
Type | Compound | λmax (nm) |
|---|---|---|
Isolated | Ethylene | 171 |
Isolated | Cyclohexene | 182 |
Isolated | Hexa-1,4-diene | 180 |
Conjugated Diene | Buta-1,3-diene | 217 |
Conjugated Diene | Hexa-2,4-diene | 227 |
Conjugated Diene | Cyclohexa-1,3-diene | 256 |
Conjugated Diene | 3-methylhex-1-ene | 220 |
Conjugated Triene | Hexa-1,3,5-triene | 258 |
Conjugated Triene | Steroid triene | 304 |
Conjugated Tetraene | Octa-1,3,5,7-tetraene | 290 |
Key Equations
Beer’s Law:
Energy of Absorption:

Conclusion
Conjugated systems exhibit unique stability, reactivity, and spectroscopic properties due to electron delocalization. Understanding their molecular orbitals, addition mechanisms, and UV-VIS absorption is essential for predicting their behavior in organic reactions.