 Back
BackConjugated Unsaturated Systems: Structure, Stability, and Reactions
Study Guide - Smart Notes

Conjugated Unsaturated Systems
Molecular Orbitals of Conjugated Systems
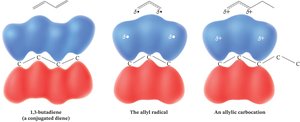
Conjugated systems are characterized by parallel p orbitals on more than two adjacent atoms, allowing for electron delocalization. This delocalization leads to unique molecular orbital arrangements and increased stability compared to isolated double bonds.
Conjugation occurs when alternating single and double bonds allow p orbitals to overlap.
All p orbitals must be aligned (coplanar) for effective conjugation.
Delocalization lowers the overall energy of the molecule.
Bond Order and p Orbital Energies
The bond order in conjugated systems is calculated as:
Higher bond order indicates stronger bonds.
In conjugated systems, bond order is distributed across the molecule, resulting in partial double bond character.
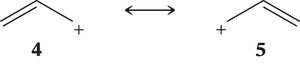
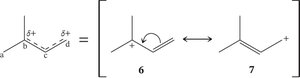
Resonance in Allylic Systems
Resonance is a key feature in conjugated and allylic systems, stabilizing cations, radicals, and anions by delocalizing charge or unpaired electrons.
Resonance structures represent different possible electron arrangements; atoms do not move, only electrons.
Proper Lewis structures are required for resonance.
Resonance stabilizes molecules by spreading charge or electrons over multiple atoms.
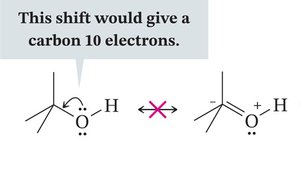
Rules for Resonance Structures
Atoms remain stationary; only electrons move.
Resonance structures must obey the octet rule; additional electron unpairing is forbidden.
Resonating groups must be coplanar for effective delocalization.
Resonance increases stability, especially when electrons are paired.
Equal resonance structures contribute equally; unequal structures contribute proportionally to their stability.
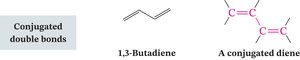
Categories of Polyunsaturated Compounds
Polyunsaturated compounds contain multiple unsaturated bonds and are classified based on the arrangement of their double bonds:
Cumulated: Two double bonds on the same carbon atom.
Conjugated: Alternating single and double bonds, allowing electron delocalization.
Isolated: Double bonds separated by more than one single bond; no delocalization.
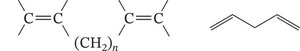
Bond Lengths in Conjugated Systems
Conjugation shortens bond lengths due to increased bond order and partial double bond character. The hybridization state of carbon atoms affects bond length.
sp and sp2 hybridized carbons form shorter sigma bonds than sp3 hybridized carbons.
Compound | Hybridization State | Bond Length (Å) |
|---|---|---|
H3C—CH3 | sp3–sp3 | 1.54 |
H2C=CH—CH3 | sp2–sp3 | 1.50 |
H2C=CH—CH=CH2 | sp2–sp2 | 1.47 |
HC≡C—CH3 | sp–sp3 | 1.46 |
HC≡C—CH=CH2 | sp–sp2 | 1.43 |
HC≡C—C≡CH | sp–sp | 1.37 |

Heats of Hydrogenation and Stability
Heats of hydrogenation are used to estimate the stability of unsaturated hydrocarbons. Lower heat of hydrogenation indicates greater stability due to conjugation.
Conjugated dienes are more stable than isolated dienes.
Stability increases with more conjugation and substitution.
UV-Visible Spectroscopy of Conjugated Systems
Conjugated systems absorb light in the UV-visible region due to their delocalized electrons. The absorption maxima (λmax) shift to longer wavelengths as conjugation increases.
Wavelength is inversely proportional to energy.
More conjugation results in higher λmax (lower energy gap).
Highly conjugated compounds can absorb visible light and appear colored.
Reactivity of Conjugated Dienes: 1,2- and 1,4-Additions
Conjugated dienes undergo addition reactions at two possible positions: 1,2- and 1,4-. The product distribution depends on reaction conditions.
1,2-Addition: Occurs at the first and second carbon; favored at low temperatures (kinetic control).
1,4-Addition: Occurs at the first and fourth carbon; favored at high temperatures (thermodynamic control).
Thermodynamic product is more stable due to more substituted double bond.
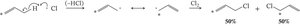
Diels-Alder Cycloaddition Reaction
The Diels-Alder reaction is a [4+2] cycloaddition between a conjugated diene and a dienophile, forming a six-membered ring. It is a key method for constructing cyclic compounds in organic synthesis.
Requires the diene to be in the s-cis conformation.
Dienophiles with electron-withdrawing groups (EWGs) are more reactive.
Electron-donating groups (EDGs) on the diene increase nucleophilicity.
Lewis acid catalysis can accelerate the reaction.
Cyclopentadiene is always s-cis and highly reactive.
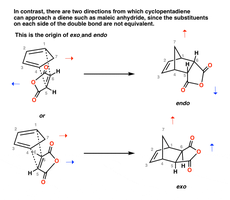
Endo and Exo Products in Bicyclic Systems
In Diels-Alder reactions involving cyclic dienes, two possible products can form: endo and exo. The endo product is typically favored due to secondary orbital interactions.
Endo addition: The substituents are oriented towards the larger bridge, maximizing interaction with EWGs.
Exo addition: The substituents are oriented away from the bridge.
Stereochemistry of both diene and dienophile is preserved in the product.
Summary Table: Types of Unsaturated Systems
Type | Structure | Electron Delocalization |
|---|---|---|
Cumulated | Two double bonds on same carbon | None |
Conjugated | Alternating single and double bonds | Yes |
Isolated | Double bonds separated by more than one single bond | None |
Key Equations
Bond Order:
Beer-Lambert Law (UV-Vis):
Additional info:
Conjugated systems are fundamental in organic chemistry, affecting color, stability, and reactivity.
Diels-Alder reactions are widely used in the synthesis of natural products and pharmaceuticals.
