 Back
BackCovalent Bonding and Shapes of Molecules: Foundations of Organic Chemistry
Study Guide - Smart Notes

Covalent Bonding and Shapes of Molecules
Introduction to Organic Chemistry
Organic chemistry is the study of carbon compounds, which typically also contain hydrogen, oxygen, and nitrogen. The unique bonding properties of carbon allow for a vast array of molecular structures, making organic chemistry central to biological and material sciences.
Organic compounds usually contain C, H, O, and N.
Electrons are found in shells around the nucleus, with quantized energy levels.
Electrons are delocalized in many organic molecules, contributing to stability and reactivity.
Atomic Structure and Electron Configuration
Atoms have electrons arranged in shells and subshells, which determine their chemical properties. Understanding electron configuration is essential for predicting bonding and molecular structure.
Each shell can hold up to electrons, where n is the shell number.
Subshells: s (1 orbital), p (3 orbitals), d (5 orbitals), f (7 orbitals).
Ground state electron configuration follows three rules:
Aufbau Principle: Fill lower energy levels first.
Pauli Exclusion Principle: Maximum two electrons per orbital, with opposite spins.
Hund’s Rule: Electrons occupy degenerate orbitals singly before pairing.
Types of Energy in Chemistry
Energy is the ability to do work and is fundamental to chemical reactions.
Potential Energy: Stored energy that can be released.
Kinetic Energy: Energy of motion.
Thermal Energy: Heat energy.
Ionization Potential: Energy required to remove an electron from an atom.
Lewis Dot Structures and Valence Electrons
Lewis dot structures represent the valence electrons of atoms, which are crucial for bond formation and chemical reactivity.
Valence electrons: Electrons in the outermost shell involved in bonding.
Lewis dot structure: Element symbol surrounded by dots representing valence electrons.
Example: Carbon has 4 valence electrons.
Bonding Models: Ionic, Covalent, and Polar Covalent Bonds
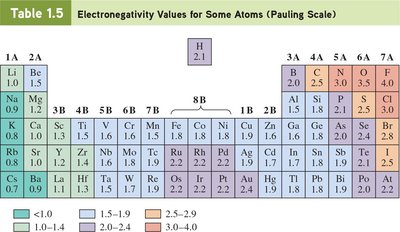
Atoms achieve stable electron configurations through ionic or covalent bonding. The nature of the bond depends on the difference in electronegativity between the atoms.
Ionic bonds: Formed by transfer of electrons (anion and cation attraction).
Covalent bonds: Formed by sharing electrons.
Octet rule: Atoms tend to achieve eight electrons in their valence shell.
Electronegativity: Measure of an atom’s ability to attract shared electrons.
Nonpolar covalent bond: Electronegativity difference < 0.5.
Polar covalent bond: Electronegativity difference between 0.5 and 1.9.
Polar bonds have a dipole moment.
Lewis Structures for Molecules and Polyatomic Ions
Lewis structures help visualize the arrangement of atoms and electrons in molecules and ions.
Count total valence electrons.
Determine atom connectivity.
Connect atoms with single bonds, then distribute remaining electrons as lone pairs.
Form double or triple bonds if necessary.
Formal Charge
Formal charge is used to determine the distribution of electrons in molecules and ions.
Formula:
The sum of formal charges must equal the total charge of the molecule or ion.
Dative Bonds
Dative (coordinate covalent) bonds occur when one atom provides both electrons for a bond, common in molecules with incomplete valence shells (e.g., boron, aluminum).
Example:
Functional Groups in Organic Chemistry
Functional groups are specific groups of atoms within molecules that determine the chemical properties and reactions of those molecules.
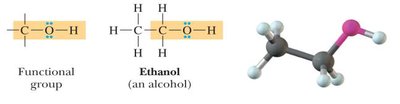
Alcohol: -OH group bonded to carbon.
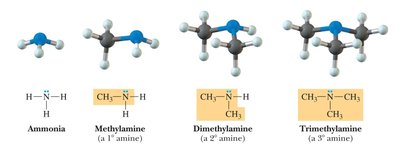
Amines: Nitrogen atom bonded to one, two, or three carbon atoms.
Aldehydes: Carbonyl group (C=O) with at least one hydrogen attached.
Ketones: Carbonyl group (C=O) with two carbons attached.
Carboxylic acids: -COOH group.
Esters: Derivative of carboxylic acid with H replaced by a carbon.
Amides: Derivative of carboxylic acid with -OH replaced by an amine.
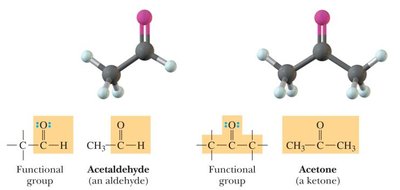
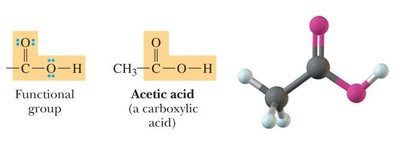
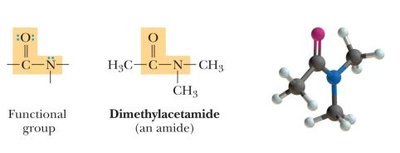
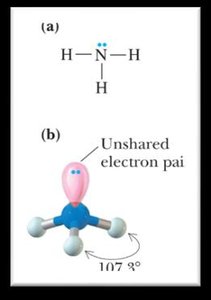
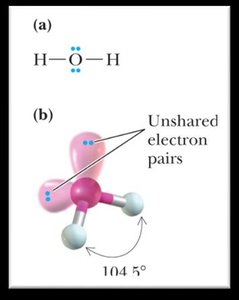
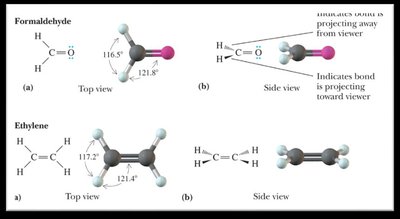
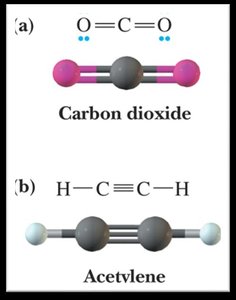
Bond Angles and Molecular Shapes (VSEPR Theory)
The Valence Shell Electron Pair Repulsion (VSEPR) theory predicts the shapes and bond angles of molecules based on the repulsion between electron pairs.
Linear: 180°
Trigonal planar: 120°
Tetrahedral: 109.5°
Trigonal pyramidal: <109.5°
Bent: <120° or <109.5°
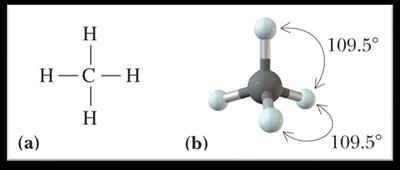
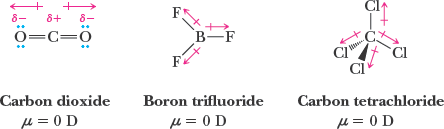
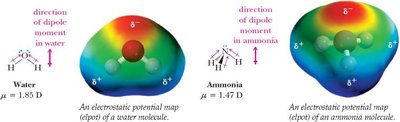
Polarity of Molecules
Molecular polarity depends on both the presence of polar bonds and the spatial arrangement of atoms. The molecular dipole moment is the vector sum of individual bond dipoles.
Nonpolar molecules: Bond dipoles cancel (e.g., CO2, CCl4).
Polar molecules: Bond dipoles do not cancel (e.g., H2O, NH3).
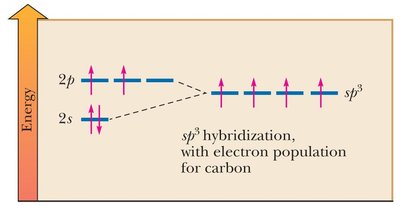
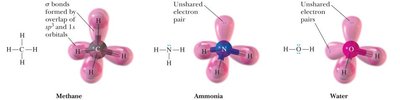
Hybridization of Atomic Orbitals
Hybridization explains the observed shapes and bond angles in molecules by combining atomic orbitals into new hybrid orbitals.
sp3 hybridization: Combination of one s and three p orbitals; tetrahedral geometry.
sp2 hybridization: Combination of one s and two p orbitals; trigonal planar geometry.
sp hybridization: Combination of one s and one p orbital; linear geometry.
Resonance Structures
Resonance describes molecules or ions that are best represented as a hybrid of multiple Lewis structures. Resonance stabilizes molecules by delocalizing electrons.
Contributing structures are connected by double-headed arrows.
Curved arrows show electron movement.
Rules: Same number of valence electrons, obey covalent bonding rules, nuclei positions unchanged.
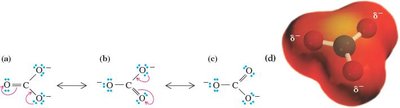

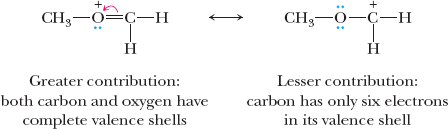
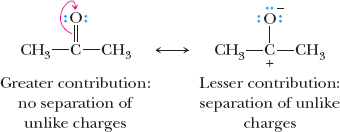
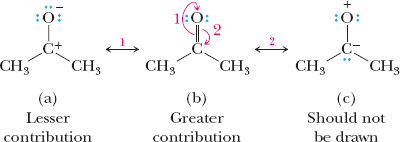
Relative Importance of Resonance Structures
Not all resonance structures contribute equally to the hybrid. The most important structures have filled valence shells, minimal charge separation, and negative charges on electronegative atoms.
Filled valence shells are prioritized.
Least separation of unlike charges is preferred.
Negative charge on more electronegative atom is favored.
Summary
This chapter provides foundational concepts in organic chemistry, including atomic structure, bonding, functional groups, molecular shapes, polarity, hybridization, and resonance. Mastery of these topics is essential for understanding the structure and reactivity of organic molecules.
