 Back
BackCovalent Bonding and Shapes of Molecules: Foundations of Organic Chemistry
Study Guide - Smart Notes

Covalent Bonding and Shapes of Molecules
Introduction to Organic Chemistry
Organic chemistry is the study of carbon compounds, which typically also contain hydrogen, oxygen, and nitrogen. The unique properties of carbon allow for a vast array of molecular structures and reactivity, forming the basis of life and synthetic materials.
Organic compounds are defined by the presence of carbon atoms.
Common elements in organic molecules: H, O, N.
Electrons are found in shells around the nucleus, with quantized energy levels.
Electrons are delocalized in many organic molecules, contributing to stability and reactivity.
Atomic Structure and Electron Configuration
Atoms consist of electrons arranged in shells and subshells. Understanding electron configuration is essential for predicting chemical bonding and molecular structure.
Shells contain up to electrons, where n is the shell number.
Subshells: s (1 orbital), p (3 orbitals), d (5 orbitals), f (7 orbitals).
Electrons fill orbitals according to three rules:
Aufbau Principle: Fill lower energy levels first.
Pauli Exclusion Principle: Maximum two electrons per orbital, with opposite spins.
Hund’s Rule: Electrons occupy degenerate orbitals singly before pairing.
Types of Energy in Chemistry
Energy is the capacity to do work. In chemistry, energy is stored in chemical bonds and released during reactions.
Potential Energy: Stored energy, released under certain conditions.
Kinetic Energy: Energy of motion.
Thermal Energy: Heat energy.
Ionization Potential: Energy required to remove an electron from an atom.
Lewis Dot Structures and Valence Electrons
Lewis dot structures represent the valence electrons of atoms, which are crucial for bond formation and chemical reactivity.
Valence electrons are in the outermost shell and participate in bonding.
Lewis dot structure: Element symbol surrounded by dots representing valence electrons.
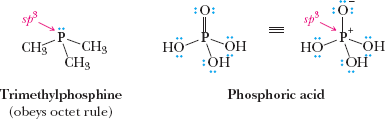
Octet rule: Atoms tend to achieve eight electrons in their valence shell (exceptions: H, B, Al).
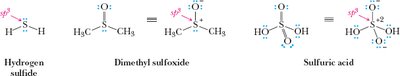
Bonding Models: Ionic, Covalent, and Polar Covalent Bonds
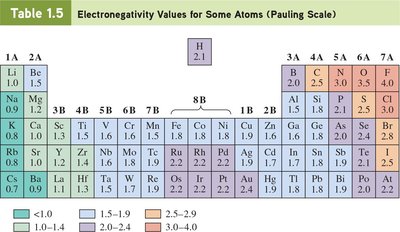
Chemical bonds form through the sharing or transfer of electrons. The nature of the bond depends on the difference in electronegativity between atoms.
Ionic bonds: Formed by transfer of electrons, resulting in cations and anions.
Covalent bonds: Formed by sharing electrons between atoms.
Polar covalent bonds: Unequal sharing of electrons due to differences in electronegativity.
Nonpolar covalent bonds: Equal sharing of electrons.
Electronegativity: Measure of an atom’s ability to attract shared electrons.
Functional Groups in Organic Chemistry
Functional groups are specific groups of atoms within molecules that determine the chemical properties and reactivity of those molecules. They are essential for classification and nomenclature in organic chemistry.
Alcohols: Contain an -OH group bonded to carbon.
Amines: Contain a nitrogen atom bonded to one, two, or three carbon atoms.
Aldehydes and Ketones: Contain a carbonyl group (C=O); aldehydes have at least one hydrogen attached to the carbonyl carbon, ketones have two carbons attached.
Carboxylic acids: Contain a -COOH group.
Esters: Derivatives of carboxylic acids where the -OH is replaced by a carbon group.
Amides: Derivatives of carboxylic acids where the -OH is replaced by an amine.
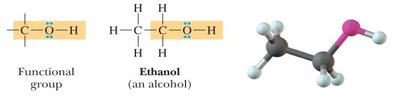
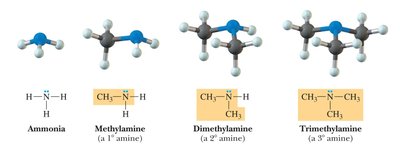
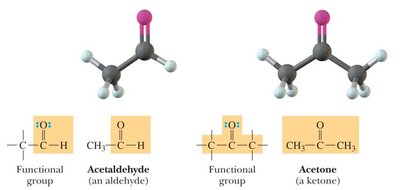
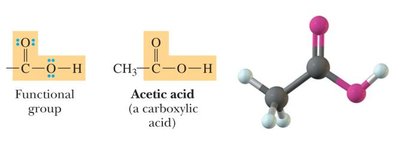
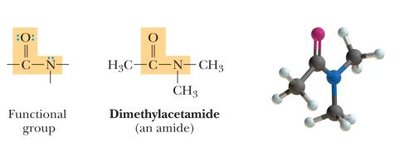
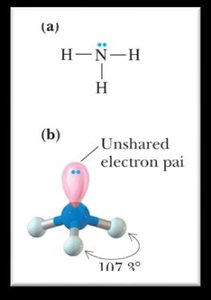
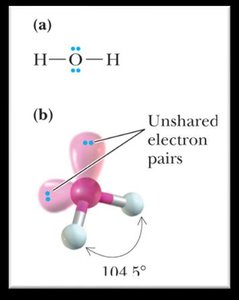
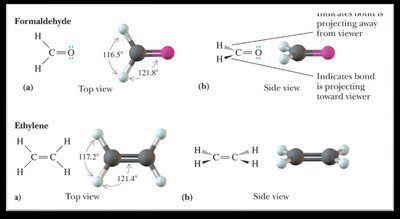
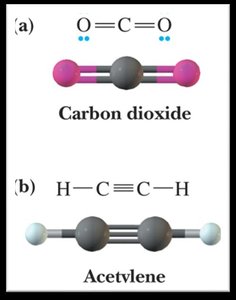
Bond Angles and Molecular Shapes (VSEPR Theory)
The Valence Shell Electron Pair Repulsion (VSEPR) theory predicts the shapes and bond angles of molecules based on the repulsion between electron pairs around a central atom.
Linear: 180° bond angle (2 bonding groups, 0 lone pairs).
Trigonal planar: 120° bond angle (3 bonding groups, 0 lone pairs).
Tetrahedral: 109.5° bond angle (4 bonding groups, 0 lone pairs).
Trigonal pyramidal: Less than 109.5° (3 bonding groups, 1 lone pair).
Bent: Less than 120° or 109.5° (2 bonding groups, 1 or 2 lone pairs).
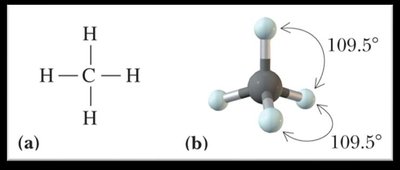
Polarity of Molecules and Dipole Moments
Molecular polarity depends on both the presence of polar bonds and the spatial arrangement of atoms. The dipole moment is a vector sum of individual bond dipoles.
Nonpolar molecules: Bond dipoles cancel due to symmetry (e.g., CO2, CCl4).
Polar molecules: Bond dipoles do not cancel, resulting in a net dipole moment (e.g., H2O, NH3).
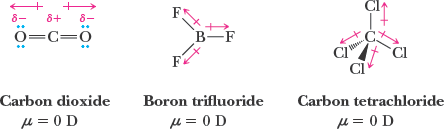
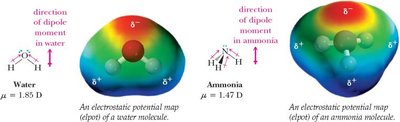
Hybridization of Atomic Orbitals
Hybridization explains the observed shapes and bond angles in molecules by combining atomic orbitals into new hybrid orbitals.
sp3 hybridization: Combination of one s and three p orbitals; tetrahedral geometry.
sp2 hybridization: Combination of one s and two p orbitals; trigonal planar geometry.
sp hybridization: Combination of one s and one p orbital; linear geometry.
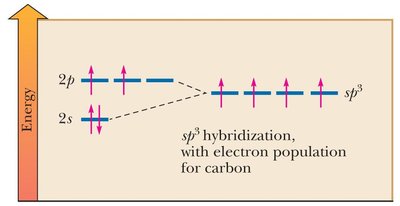
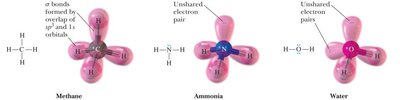
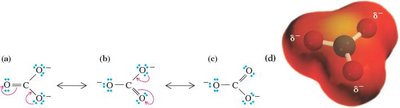
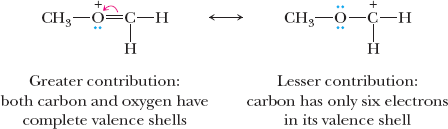
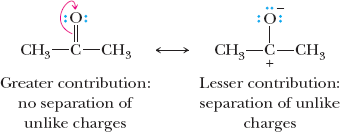
Resonance Structures
Resonance describes molecules or ions that are best represented by two or more contributing Lewis structures. The actual structure is a hybrid of these forms.
Contributing structures: Individual Lewis structures connected by double-headed arrows.
Curved arrows: Indicate electron movement in resonance and reaction mechanisms.
Rules: All contributing structures must have the same number of valence electrons, obey covalent bonding rules, and differ only in electron arrangement.
Relative importance: Structures with filled valence shells, minimal charge separation, and negative charge on more electronegative atoms contribute more.

Summary Table: VSEPR Geometries and Bond Angles
Lone Pairs | Bonding Groups | Electron Geometry | Molecular Geometry | Bond Angle |
|---|---|---|---|---|
0 | 2 | Linear | Linear | 180° |
0 | 3 | Trigonal planar | Trigonal planar | 120° |
1 | 2 | Trigonal planar | Bent | <120° |
0 | 4 | Tetrahedral | Tetrahedral | 109.5° |
1 | 3 | Tetrahedral | Trigonal pyramidal | <109.5° |
2 | 2 | Tetrahedral | Bent | <109.5° |
0 | 5 | Trigonal bipyramidal | Trigonal bipyramidal | 90°, 120°, 180° |
1 | 4 | Trigonal bipyramidal | Seesaw | 90°, 120°, 180° |
2 | 3 | Trigonal bipyramidal | T-shaped | 90°, 180° |
3 | 2 | Trigonal bipyramidal | Linear | 180° |
0 | 6 | Octahedral | Octahedral | 90°, 180° |
1 | 5 | Octahedral | Square pyramidal | 90°, 180° |
2 | 4 | Octahedral | Square planar | 90°, 180° |
Additional info: Academic context and examples were added to clarify and expand upon brief points in the original material, ensuring completeness and self-contained study notes.
