 Back
BackCovalent Bonding and Shapes of Molecules: Foundations of Organic Chemistry
Study Guide - Smart Notes

Chapter 1: Covalent Bonding and Shapes of Molecules
What is Organic Chemistry?
Organic chemistry is the study of carbon compounds, which typically also contain hydrogen, oxygen, and nitrogen. The unique bonding properties of carbon allow for a vast diversity of molecular structures and reactivity, forming the basis of life and many synthetic materials.
Shells: Regions around the nucleus where electrons are likely to be found. Each shell can hold up to electrons, where n is the shell number.
Subshells: Shells are divided into s, p, d, and f subshells, with increasing numbers of orbitals (s=1, p=3, d=5, f=7).
Electron Configuration: The arrangement of electrons in an atom, following the Aufbau Principle, Pauli Exclusion Principle, and Hund’s Rule.
Delocalization: Electrons can be spread over several atoms, especially in conjugated systems.
Example: The electron configuration of carbon is .
Principles of Electron Configuration
Aufbau Principle: Electrons fill lower energy orbitals first (e.g., 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, etc.).
Pauli Exclusion Principle: Each orbital can hold two electrons with opposite spins.
Hund’s Rule: Electrons occupy degenerate orbitals singly before pairing, with parallel spins.
Types of Energy in Chemistry
Potential Energy: Stored energy that can be released (e.g., chemical bonds).
Kinetic Energy: Energy of motion.
Thermal Energy: Energy associated with temperature (heat).
Ionization Potential: The energy required to remove an electron from an atom or molecule. It is higher for electrons closer to the nucleus due to stronger electrostatic attraction.
Lewis Dot Structures and Valence Electrons
Lewis dot structures represent the valence electrons (outermost electrons) of an atom, which are involved in bonding and chemical reactions.
Valence Electrons: Electrons in the outermost shell; determine chemical reactivity.
Lewis Dot Structure: Symbol of the element surrounded by dots representing valence electrons.
Lewis Model of Bonding
Covalent Bond: Formed by sharing electrons between atoms.
Ionic Bond: Formed by transfer of electrons, resulting in attraction between cations and anions.
Octet Rule: Atoms tend to gain, lose, or share electrons to achieve eight electrons in their valence shell (exceptions: H, B, Al).
Electronegativity: The ability of an atom to attract shared electrons in a bond.
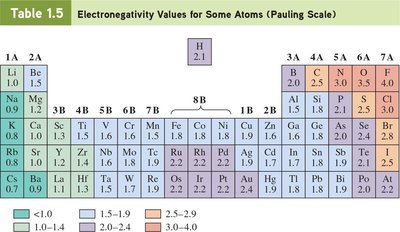
Bond Polarity:
Nonpolar Covalent Bond: Electronegativity difference < 0.5
Polar Covalent Bond: Electronegativity difference between 0.5 and 1.9
Ionic Bond: Electronegativity difference > 1.9
Lewis Structures for Molecules and Polyatomic Ions
Count total valence electrons.
Determine atom connectivity.
Connect atoms with single bonds, then distribute remaining electrons as lone pairs to satisfy the octet rule (with exceptions).
Use double or triple bonds if necessary to complete octets.
Formal Charge
Formal charge is used to determine the most stable Lewis structure for a molecule or ion.
Formula:
The sum of formal charges must equal the overall charge of the molecule or ion.
Dative (Coordinate) Bonds
A dative bond forms when both electrons in a shared pair come from the same atom. Common in compounds with boron or aluminum.
Functional Groups in Organic Chemistry
Functional groups are specific groups of atoms within molecules that determine the characteristic chemical reactions of those molecules.
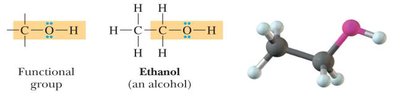
Alcohols: Contain an -OH group bonded to a carbon atom. Classified as primary (1°), secondary (2°), or tertiary (3°) based on the number of carbon atoms attached to the carbon bearing the -OH group.
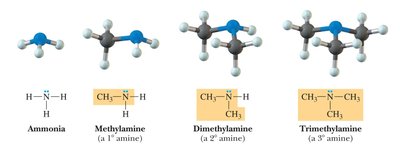
Amines: Contain a nitrogen atom bonded to one, two, or three carbon atoms (primary, secondary, tertiary amines).
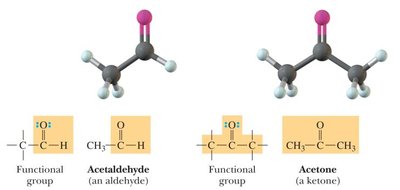
Aldehydes and Ketones: Both contain a carbonyl group (C=O). Aldehydes have at least one hydrogen attached to the carbonyl carbon; ketones have two carbons attached.
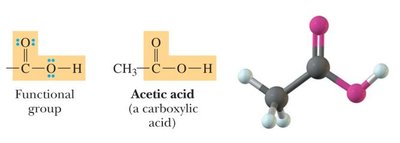
Carboxylic Acids: Contain a -COOH group (carboxyl group: carbonyl + hydroxyl).
Esters: Derivatives of carboxylic acids where the -OH is replaced by an -OR group (R = alkyl group).
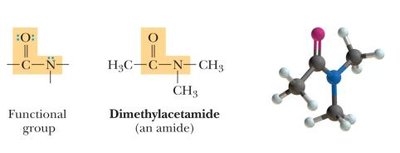
Amides: Derivatives of carboxylic acids where the -OH is replaced by an amine group (-NH2, -NHR, or -NR2).
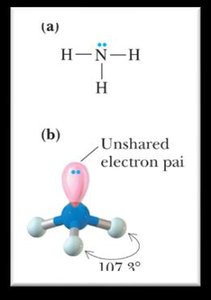
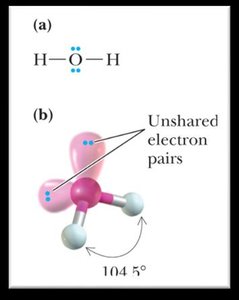
Bond Angles and Molecular Shapes (VSEPR Theory)
The Valence Shell Electron Pair Repulsion (VSEPR) theory predicts the geometry of molecules based on the repulsion between electron pairs around a central atom.
Lone Pairs | Bonding Groups | Electron Geometry | Molecular Geometry | Bond Angle |
|---|---|---|---|---|
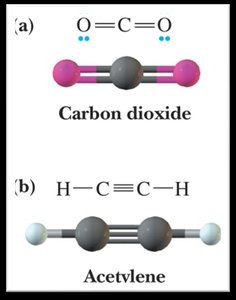
0 | 2 | Linear | Linear | 180° |
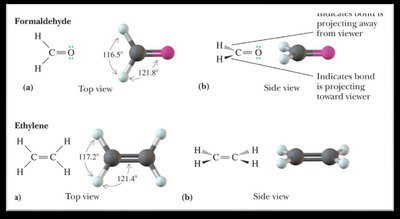
0 | 3 | Trigonal planar | Trigonal planar | 120° |
1 | 2 | Trigonal planar | Bent | <120° |
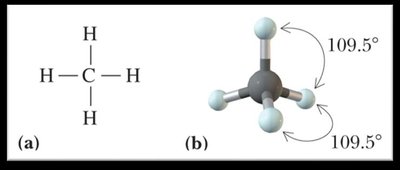
0 | 4 | Tetrahedral | Tetrahedral | 109.5° |
1 | 3 | Tetrahedral | Trigonal pyramidal | <109.5° |
2 | 2 | Tetrahedral | Bent | <109.5° |
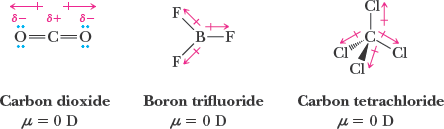
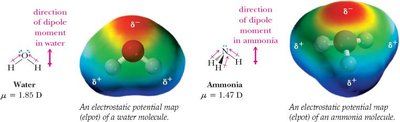
Polarity of Molecules
A molecule is polar if it has a net dipole moment, which depends on both the polarity of individual bonds and the molecular geometry. The vector sum of bond dipoles determines the overall molecular dipole moment.
Nonpolar molecules can have polar bonds if the bond dipoles cancel due to symmetry (e.g., CO2, CCl4).
Polar molecules have a net dipole moment (e.g., H2O, NH3).
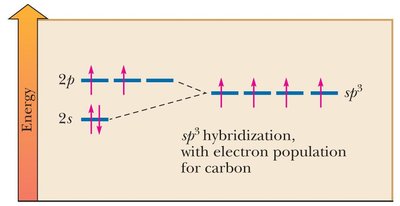
Hybridization of Atomic Orbitals
Hybridization describes the mixing of atomic orbitals to form new hybrid orbitals suitable for the pairing of electrons to form chemical bonds.
sp3 Hybridization: Mixing of one s and three p orbitals, forming four equivalent sp3 orbitals (tetrahedral geometry, 109.5° bond angles).
sp2 Hybridization: Mixing of one s and two p orbitals, forming three sp2 orbitals (trigonal planar geometry, 120° bond angles).
sp Hybridization: Mixing of one s and one p orbital, forming two sp orbitals (linear geometry, 180° bond angles).
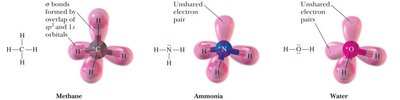
Resonance
Resonance is a concept used to represent the delocalization of electrons within molecules where a single Lewis structure is insufficient. The actual structure is a hybrid of all valid resonance forms.
Resonance structures are connected by double-headed arrows.
Curved arrows indicate the movement of electrons (not atoms).
Rules: All resonance forms must have the same number of valence electrons, obey covalent bonding rules, and differ only in the arrangement of electrons.
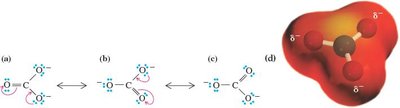

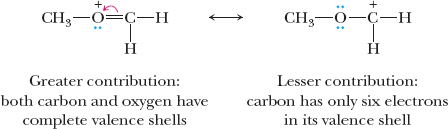
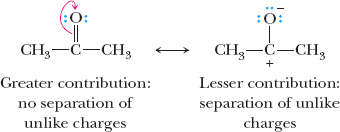
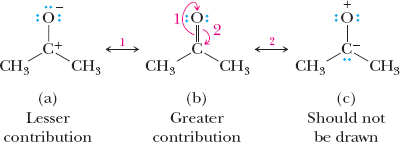
Relative Importance of Resonance Structures
Structures with filled valence shells are most important.
Structures with minimal separation of unlike charges are favored.
Negative charges are more stable on more electronegative atoms.
Summary Table: Common Functional Groups
Functional Group | General Structure | Example |
|---|---|---|
Alcohol | -OH | Ethanol |
Aldehyde | -CHO | Acetaldehyde |
Ketone | RCOR' | Acetone |
Carboxylic Acid | -COOH | Acetic acid |
Ester | -COOR | Ethyl acetate |
Amide | -CONH2 | Dimethylacetamide |
Amine | -NH2, -NHR, -NR2 | Methylamine |
Additional info: This chapter provides foundational concepts for understanding molecular structure, bonding, and reactivity in organic chemistry. Mastery of these topics is essential for all subsequent chapters in organic chemistry.
