Back
BackElectrocyclic Reactions: Ring Closures and Openings in Organic Chemistry
Study Guide - Smart Notes

Electrocyclic Reactions
Introduction to Electrocyclic Reactions
Electrocyclic reactions are a class of pericyclic reactions in organic chemistry where a conjugated π system undergoes a ring closure or ring opening, resulting in the formation or breaking of a cyclic structure. These reactions are governed by the number of π electrons involved and the conditions (heat or light) under which the reaction occurs.
Electrocyclic ring closure: A linear π system forms a ring via a concerted mechanism.
Electrocyclic ring opening: A cyclic π system opens to form a linear conjugated system.
Key factors: Number of π electrons (4n or 4n+2), stereochemistry, and reaction conditions (thermal or photochemical).
Electron Count and Reaction Conditions
The stereochemistry of electrocyclic reactions depends on the electron count and whether the reaction is induced by heat or light. The Woodward-Hoffmann rules predict the outcome:
4n π electrons: Thermal reactions proceed via conrotatory motion; photochemical reactions proceed via disrotatory motion.
4n+2 π electrons: Thermal reactions proceed via disrotatory motion; photochemical reactions proceed via conrotatory motion.
Example: For a system with 6 π electrons (4n+2, n=1), thermal ring closure is disrotatory, while photochemical ring closure is conrotatory.
Examples of Electrocyclic Ring Closures and Openings
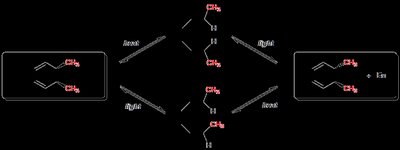
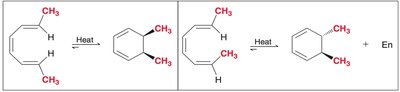
Electrocyclic reactions can be illustrated using substituted cyclohexadiene and octatriene systems. The stereochemistry of the products is determined by the mode of rotation (conrotatory or disrotatory) and the substituents' positions.
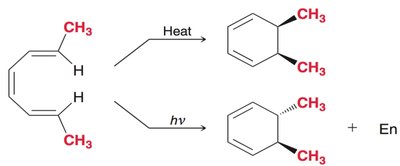
Thermal ring closure: Disrotatory for 4n+2 π electrons.
Photochemical ring closure: Conrotatory for 4n+2 π electrons.
Thermal ring opening: Disrotatory for 4n+2 π electrons.
Photochemical ring opening: Conrotatory for 4n+2 π electrons.
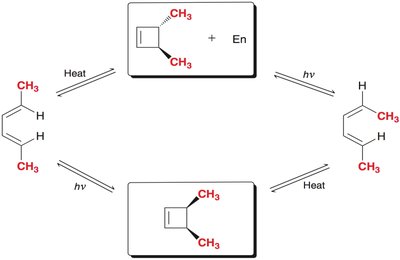
Stereochemistry of Electrocyclic Reactions
The stereochemical outcome of electrocyclic reactions is determined by the symmetry of the molecular orbitals and the mode of rotation. The substituents' orientation (cis or trans) in the product depends on whether the reaction is conrotatory or disrotatory.
Conrotatory motion: Both terminal groups rotate in the same direction, leading to trans stereochemistry.
Disrotatory motion: Terminal groups rotate in opposite directions, leading to cis stereochemistry.
Example: In a thermal ring closure of hexatriene (6 π electrons), disrotatory motion results in cis product.
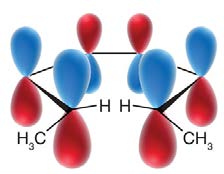
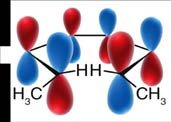
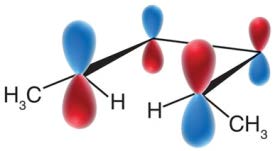
Woodward-Hoffmann Rules and Molecular Orbitals
The Woodward-Hoffmann rules use molecular orbital symmetry to predict the stereochemistry of electrocyclic reactions. The highest occupied molecular orbital (HOMO) of the reactant determines the allowed mode of rotation.
Disrotatory: Allowed for 4n+2 π electrons under thermal conditions.
Conrotatory: Allowed for 4n π electrons under thermal conditions.
Equation: The electron count is given by .
Summary Table: Electrocyclic Reaction Stereochemistry
Electron Count | Thermal Reaction | Photochemical Reaction |
|---|---|---|
4n π electrons | Conrotatory | Disrotatory |
4n+2 π electrons | Disrotatory | Conrotatory |
Applications and Importance
Electrocyclic reactions are important in organic synthesis, especially in the construction of cyclic compounds and the study of reaction mechanisms. Understanding the stereochemistry and electron count allows chemists to predict product outcomes and design efficient synthetic routes.