 Back
BackElectronic Effects and Reactive Intermediates in Organic Chemistry
Study Guide - Smart Notes

Electronic Effects in Organic Chemistry
Inductive Effect
The inductive effect is a permanent electronic effect observed in organic molecules, resulting from the polarization of σ-bonds due to differences in electronegativity between atoms. This effect leads to the displacement of electron density along a carbon chain, either towards or away from a substituent group.
Negative Inductive Effect (–I): Electron-withdrawing groups (EWG) pull electron density towards themselves. Examples: –NO2, –CN, –COOH, F, Cl, Br, I, –OH, –C6H5.
Positive Inductive Effect (+I): Electron-donating groups (EDG) push electron density away from themselves. Examples: (CH3)3C–, (CH3)2CH–, –C2H5, –CH3.
Inductive effects are short-range and decrease rapidly with distance from the substituent.
Applications:
Explains acid and base strength in organic compounds.
Stabilizes carbocations: More alkyl groups (+I effect) increase carbocation stability.
Influences reactivity and physical properties of molecules.
Electronegativity and Bond Polarity
Electronegativity is the tendency of an atom to attract shared electrons in a chemical bond. The difference in electronegativity between two atoms determines the polarity of the bond:
If the difference is large, the bond is polar (e.g., C–F).
If the difference is small or zero, the bond is nonpolar (e.g., C–H).
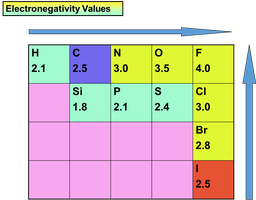
Example: The C–F bond is highly polarized due to the large electronegativity difference (C: 2.5, F: 4.0), while the C–H bond is nearly nonpolar (C: 2.5, H: 2.1).
Resonance (Mesomeric) Effect
Definition and Types
The resonance effect (or mesomeric effect) involves the delocalization of electrons in a conjugated π-system, resulting in multiple contributing structures (canonical forms) that differ only in the position of electrons, not atoms.
–M or –R Effect: Electron-withdrawing groups (e.g., –NO2, –CHO) pull electrons towards themselves through resonance.
+M or +R Effect: Electron-donating groups (e.g., –OH, –OR, –Cl) push electrons away from themselves through resonance.
Key Points:
Resonance stabilizes molecules by lowering their potential energy.
Resonance structures are imaginary; the actual molecule is a resonance hybrid.
Rules for drawing resonance structures include planarity, conjugation, and conservation of charge.
Electromeric Effect
Definition and Example
The electromeric effect is a temporary effect observed in the presence of a reagent, involving the complete transfer of π-electrons in an unsaturated system (such as alkenes or carbonyls) to one of the atoms, resulting in a transient charge separation.
Occurs only in the presence of an attacking reagent.
Example: Addition of HBr to an alkene, where the π-electrons shift to form a carbocation and a bromide ion.
Hyperconjugation
Definition and Significance
Hyperconjugation is the stabilizing interaction resulting from the delocalization of electrons in a σ-bond (usually C–H or C–C) with an adjacent empty or partially filled p-orbital or π-orbital. This leads to increased stability of carbocations, alkenes, and radicals.
More hyperconjugative structures mean greater stability.
Example: The tert-butyl cation ((CH3)3C+) is more stable than the ethyl cation (CH3CH2+) due to more C–H bonds available for hyperconjugation.
Hyperconjugation involves σ-orbitals, while resonance involves π-orbitals.
Steric Effect
The steric effect refers to the influence of the physical size and spatial arrangement of atoms or groups within a molecule on its reactivity and properties. Bulky groups can hinder reactions by preventing close approach of reactants.
Classification of Organic Reactions
By Structural Change
Addition: Atoms are added to double or triple bonds (e.g., alkene + HBr → alkyl bromide).
Substitution: An atom or group is replaced by another (e.g., alkyl halide + OH– → alcohol + halide ion).
Elimination: Atoms are removed to form double or triple bonds (e.g., alcohol → alkene + H2O).
Rearrangement: Atoms rearrange within the molecule (e.g., hydride shift in carbocation rearrangement).
Condensation, Hydrolysis, Combustion: Other important reaction types.
By Reaction Type
Acid-Base Reactions
Oxidation and Reduction Reactions
By Functional Group
Reactions are also classified based on the functional group involved (e.g., alcohols, alkenes, alkynes).
Mechanisms of Organic Reactions
Arrow Notation
Curved arrows are used to show the movement of electrons during bond breaking and making:
Homolysis: Each atom gets one electron from the bond (forms radicals).
Heterolysis: Both electrons go to one atom (forms ions).
Reactive Intermediates
Types and Properties
Free Radicals: Neutral species with an unpaired electron; highly reactive.
Carbocations: Positively charged carbon species; act as electrophiles.
Carbanions: Negatively charged carbon species; act as nucleophiles.
Carbenes: Neutral species with a divalent carbon atom; can be both electrophilic and nucleophilic.
Many organic reactions involve the interaction of nucleophiles (electron-rich) and electrophiles (electron-deficient).
Stability of Carbocations
Carbocation stability increases with the number of alkyl groups attached to the positively charged carbon due to the +I inductive effect. Order of stability:
Tertiary > Secondary > Primary > Methyl
Applications of Inductive Effect
Acid Strength: Electron-withdrawing groups increase acid strength; electron-donating groups decrease it.
Base Strength: Aliphatic amines are more basic than ammonia due to +I effect; aniline is less basic due to –I and +R effects of the phenyl group.
Summary Table: Inductive Effects of Common Groups
Group | Effect |
|---|---|
–NO2, –CN, –COOH, F, Cl, Br, I | –I (Electron-withdrawing) |
(CH3)3C–, (CH3)2CH–, –C2H5, –CH3 | +I (Electron-donating) |
