 Back
BackElectrophilic Aromatic Substitution and Friedel-Crafts Reactions: Mechanisms, Substituent Effects, and Applications
Study Guide - Smart Notes

Electrophilic Aromatic Substitution (EAS)
Introduction to EAS
Electrophilic Aromatic Substitution (EAS) is a fundamental reaction in organic chemistry, where an aromatic ring reacts with an electrophile, resulting in the substitution of a hydrogen atom. Unlike alkenes, which favor addition reactions due to the stability of the resulting sigma bond, arenes (such as benzene) favor substitution because their aromatic pi system is highly stabilized by resonance.
Electrophilic Addition: Alkenes react with electrophiles via a carbocation intermediate, favoring addition due to the stability of the Nu-C sigma bond.
Electrophilic Substitution: Aromatic rings undergo substitution, preserving their resonance stabilization.
Electrophilic Limitations: Benzene will only react if the electrophile is sufficiently reactive to disrupt its resonance.
EAS Mechanism
The mechanism of EAS involves three main steps:
Preparation of the Electrophile: The electrophile is generated, often with the aid of acids or catalysts.
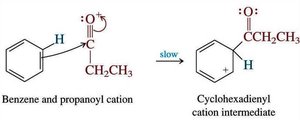
Breaking Aromaticity: The aromatic ring attacks the electrophile, forming a cyclohexadienyl cation intermediate and temporarily losing aromaticity.
Restoring Aromaticity: A base removes a proton, restoring the aromatic system.
Common EAS Reactions
Nitration: Uses HNO3 and H2SO4 to generate the nitronium ion (NO2+).
Sulfonation: Uses SO3 and H2SO4 to introduce a sulfonic acid group.
Halogenation: Requires a halogen and an iron catalyst (e.g., FeCl3).
Friedel-Crafts Alkylation and Acylation: Uses alkyl or acyl halides with AlCl3 as a catalyst.
Friedel-Crafts Reactions
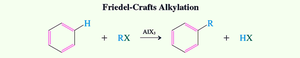
Friedel-Crafts Alkylation
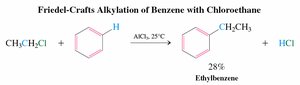
Friedel-Crafts alkylation is a method for forming sp2-sp3 carbon-carbon bonds by reacting benzene with alkyl halides in the presence of a Lewis acid catalyst (AlCl3).
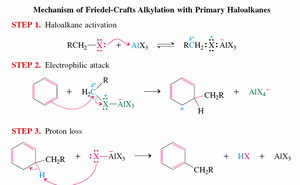
Primary Haloalkanes: Undergo activation by AlCl3, followed by electrophilic attack and proton loss.
Secondary and Tertiary Haloalkanes: Form carbocations, which are attacked by the benzene ring.
Problems: Carbocations can rearrange, leading to poor yields and polyalkylation.
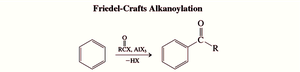
Friedel-Crafts Acylation
Friedel-Crafts acylation uses acyl halides to introduce aryl ketones, avoiding carbocation rearrangements. The key intermediate is the acylium ion, which is stabilized by resonance.
Acyl Cation: The electrophilic center is the carbonyl carbon.
Mechanism: Similar to alkylation, but more controlled and avoids polyacylation.
Acyl Chlorides and Anhydrides: Both can be used to generate the acylium ion.
Reduction: Clemmensen and Wolff-Kishner reductions can remove the acyl group, converting it to an alkyl group.
Substituent Effects and Directing Groups
Disubstitution and Regioselectivity
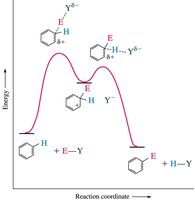
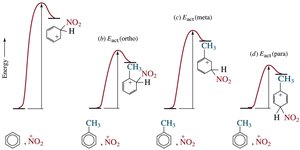
Substituents on a benzene ring influence both the reactivity and the position (regioselectivity) of further substitution. The stability of the cyclohexadienyl cation intermediate determines the preferred site of attack.
Ortho/Para Attack: Substituents that stabilize the intermediate favor ortho and para positions.
Meta Attack: Substituents that destabilize the intermediate favor meta substitution.
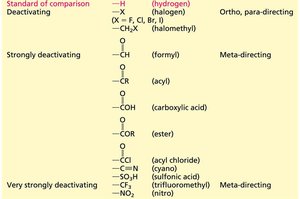
Activating and Deactivating Substituents
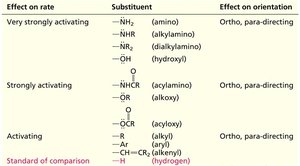
Substituents are classified based on their effect on the rate and orientation of EAS.
Activating Groups: Increase the rate and direct substitution to ortho/para positions (e.g., -NH2, -OH, -OR).
Deactivating Groups: Decrease the rate and often direct substitution to the meta position (e.g., -NO2, -CF3, -COOH).
Halogens: Are deactivating but direct to ortho/para due to their ability to donate lone pairs.
Reaction Coordinates for Substituent Effects
The energy profile for EAS changes depending on the substituent. Para substitution often has the lowest activation energy, but is not always the major product due to steric and statistical factors. 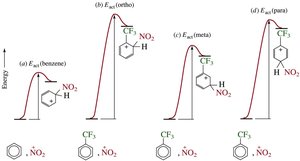
Nucleophilic Aromatic Substitution (SNAr)
Introduction to SNAr
Aryl halides can undergo nucleophilic aromatic substitution, especially when electron-withdrawing groups (such as nitro groups) are present at the ortho or para positions.
Addition-Elimination Mechanism: The nucleophile adds to the ring, forming a Meisenheimer complex, followed by elimination to restore aromaticity.
Rate Determining Step: Loss of aromaticity is the slow step.
Kinetics: SNAr reactions are second order.
Substituent Effects: Electron-withdrawing groups increase the rate; more ortho/para nitro groups allow weaker nucleophiles to react.
Activating vs. Deactivating Groups in SNAr
The nitro group can be activating or deactivating depending on the context. Other groups can also activate aryl halides for nucleophilic attack.
Summary Table: Substituent Effects on EAS
Effect on Rate | Substituent | Effect on Orientation |
|---|---|---|
Very strongly activating | –NH2, –NHR, –NR2, –OH | Ortho, para-directing |
Strongly activating | –NHCR, –OR | Ortho, para-directing |
Activating | –OCR, –R, –Ar, –CH=CR2 | Ortho, para-directing |
Deactivating | –X (halogen), –CH2X | Ortho, para-directing |
Strongly deactivating | –CHO, –COR, –COOH, –COOR | Meta-directing |
Very strongly deactivating | –COCl, –CN, –SO3H, –CF3, –NO2 | Meta-directing |
Key Equations
General EAS Mechanism:
Friedel-Crafts Alkylation:
Friedel-Crafts Acylation:
Practice Problems
Show the mechanism for nitration, sulfonation, halogenation, alkylation, and acylation of benzene.
Predict the major product for monobromination of substituted benzenes.
Explain the resonance structures that stabilize the intermediate in SNAr reactions.
Additional info:
Clemmensen reduction uses Zn(Hg) and HCl to reduce aryl ketones to alkylbenzenes.
Wolff-Kishner reduction uses hydrazine and base to achieve the same transformation.
Electron-withdrawing groups (EWGs) at ortho/para positions greatly enhance SNAr reactivity.
