 Back
BackEthers and Epoxides: Structure, Properties, and Reactions
Study Guide - Smart Notes

Ethers and Epoxides
Introduction to Ethers
Ethers are a class of organic compounds characterized by an oxygen atom connected to two alkyl or aryl groups. Their general formula is R—O—R', where R and R' can be the same or different groups, resulting in symmetrical or unsymmetrical ethers. Ethers are important in both laboratory and industrial chemistry due to their unique properties and applications.
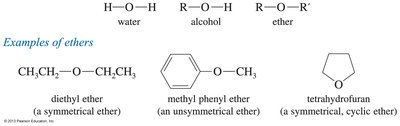
Definition: Ethers are compounds with the structure R—O—R', where the oxygen atom bridges two carbon-containing groups.
Types: Symmetrical ethers (both groups identical) and unsymmetrical ethers (groups different).
Examples: Diethyl ether (symmetrical), methyl phenyl ether (unsymmetrical), tetrahydrofuran (cyclic ether).
Structure and Polarity of Ethers
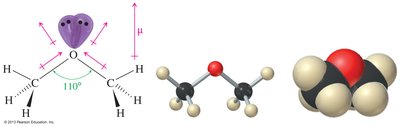
The oxygen atom in ethers is sp3 hybridized, resulting in a bent molecular geometry. The C—O—C bond angle is approximately 110°, and the C—O bonds are polar due to the electronegativity of oxygen.
Hybridization: Oxygen is sp3 hybridized.
Geometry: Bent shape with a tetrahedral arrangement around oxygen.
Bond Angle: C—O—C angle is about 110°.
Polarity: Ethers have polar C—O bonds, but the overall molecule is less polar than alcohols.
Physical Properties of Ethers
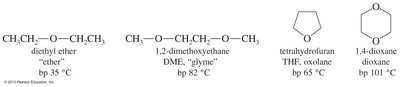
Ethers generally have lower boiling points than alcohols of similar molecular weight because they cannot form hydrogen bonds with themselves. However, the lone pairs on oxygen allow ethers to accept hydrogen bonds from water, making some ethers soluble in water.
Boiling Points: Lower than alcohols due to lack of hydrogen bonding between ether molecules.
Solubility: Ethers can accept hydrogen bonds from water, increasing their solubility.
Comparison: Diethyl ether is more soluble in water than pentane but less than t-butanol.
Ethers as Solvents
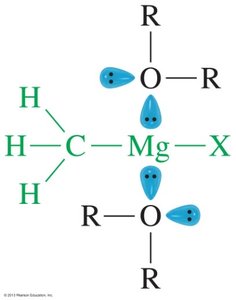
Ethers are widely used as solvents in organic chemistry because they are relatively unreactive and can dissolve both polar and nonpolar substances. They act as Lewis bases and are especially useful for dissolving organometallic compounds.
Unreactivity: Ethers are stable toward strong bases and many reagents.
Solvent Properties: Can dissolve a wide range of compounds.
Lewis Base: Ethers coordinate to metal cations, stabilizing organometallic reagents.
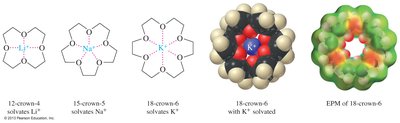
Crown Ether Complexes
Crown ethers are cyclic polyethers that can encapsulate metal cations within their ring structure. The size of the crown ether determines which cation it can best solvate. This property allows crown ethers to dissolve polar salts in nonpolar solvents and is utilized in various applications, including antibiotics.
Structure: Cyclic ethers with multiple oxygen atoms.
Complexation: Crown ethers selectively bind metal cations based on ring size.
Applications: Used to solubilize salts and in biological systems.
Nomenclature of Ethers
Ethers can be named using common names or IUPAC names. The common name lists the two alkyl groups in alphabetical order followed by "ether." The IUPAC name treats the smaller group as an "alkoxy" substituent on the larger alkane.
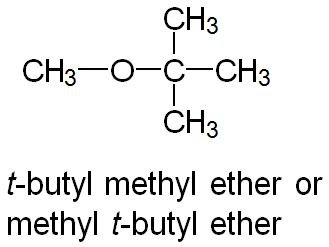
Common Names: Name both groups and add "ether" (e.g., ethyl methyl ether).
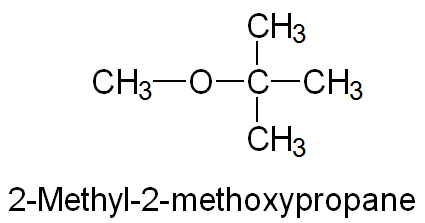
IUPAC Names: Name the larger group as the parent alkane; the smaller group as an alkoxy substituent (e.g., methoxyethane).
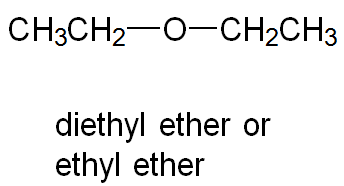
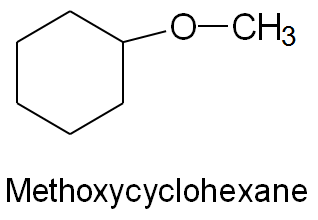
Cyclic Ethers
Cyclic ethers are rings containing oxygen atoms. Common examples include oxetane, tetrahydrofuran (THF), tetrahydropyran, and 1,4-dioxane. Epoxides (oxiranes) are three-membered cyclic ethers with significant ring strain.
Examples: THF, dioxane, oxetane, epoxides.
Epoxides: Highly reactive due to ring strain.
Preparation of Ethers
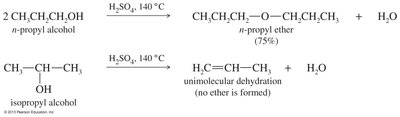
Ethers can be synthesized by several methods, including the Williamson ether synthesis, bimolecular dehydration of alcohols, and alkoxymercuration-demercuration.
Williamson Ether Synthesis: SN2 reaction of an alkoxide ion with a primary alkyl halide or tosylate.
Bimolecular Dehydration: Condensation of two alcohols in the presence of acid.
Alkoxymercuration-Demercuration: Addition of alcohol to an alkene using mercuric acetate, followed by reduction.
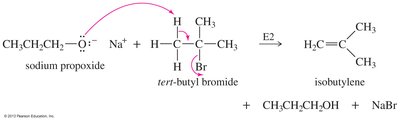
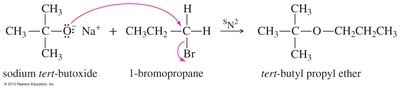
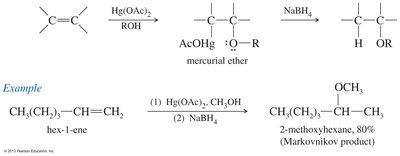
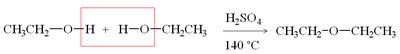
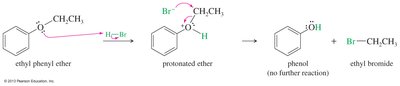
Cleavage of Ethers
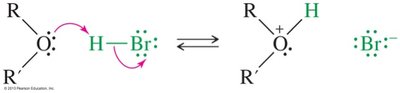
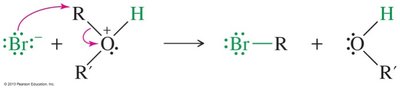
Ethers are generally unreactive, but they can be cleaved by strong acids such as HI and HBr. The mechanism involves protonation of the oxygen, followed by nucleophilic attack by the halide ion.
Reactivity: HI is more reactive than HBr.
Mechanism: Protonation, SN2 displacement, and further reaction of alcohol (except phenols).
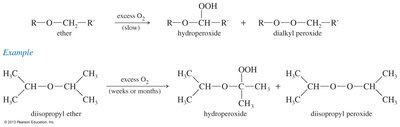
Autoxidation of Ethers
Ethers can slowly oxidize in the presence of atmospheric oxygen to form hydroperoxides and dialkyl peroxides, which are highly explosive. Proper storage and handling are essential to prevent accidents.
Products: Hydroperoxides and dialkyl peroxides.
Precautions: Store ethers in tightly sealed containers and avoid distilling to dryness.
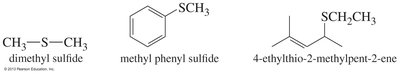
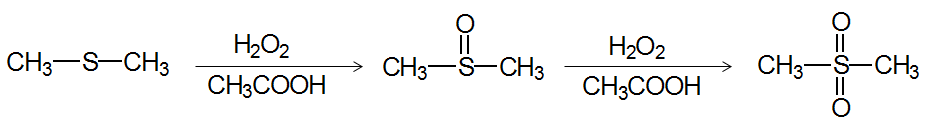
Thioethers (Sulfur Analogs of Ethers)
Thioethers are sulfur analogs of ethers, with the general formula R—S—R'. They can be synthesized by methods similar to ethers and undergo oxidation to sulfoxides and sulfones.
Structure: R—S—R' (sulfur replaces oxygen).
Preparation: Williamson-type synthesis using thiolates.
Oxidation: Thioethers can be oxidized to sulfoxides and sulfones.


Synthesis of Epoxides
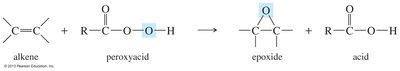
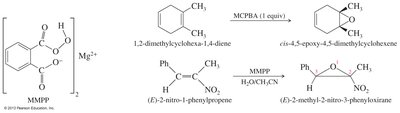
Epoxides are three-membered cyclic ethers formed by the oxidation of alkenes using peroxyacids such as meta-chloroperoxybenzoic acid (MCPBA). The reaction is stereospecific and occurs via syn addition.
Reagents: Peroxyacids (e.g., MCPBA).
Mechanism: Syn addition to the alkene.
Selectivity: Electron-rich double bonds react faster.
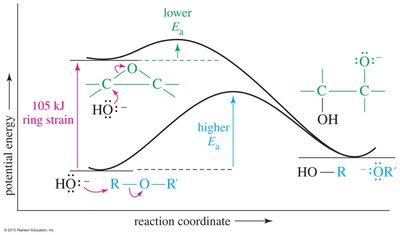
Epoxide Ring Opening Reactions
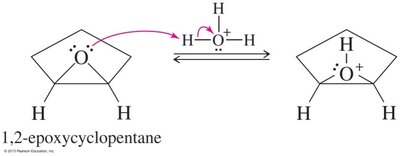
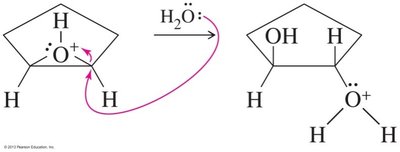
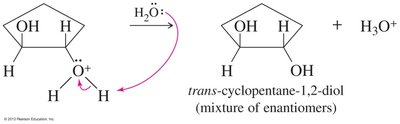
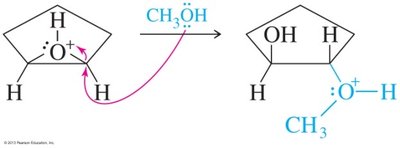
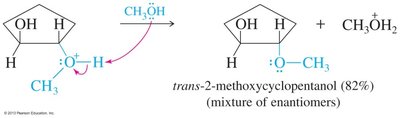
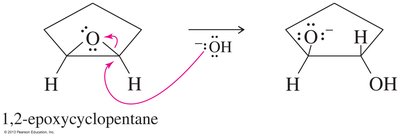
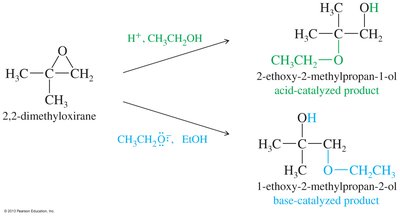
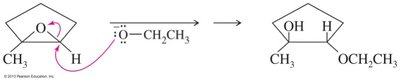
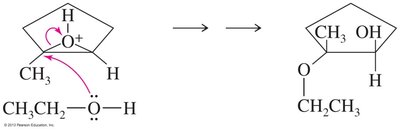
Epoxides can be opened by acids, bases, or nucleophiles. The regioselectivity depends on the reaction conditions: under acidic conditions, nucleophiles attack the more substituted carbon; under basic conditions, they attack the less hindered carbon.
Acid-Catalyzed Opening: Protonation followed by nucleophilic attack.
Base-Catalyzed Opening: Nucleophile attacks the less hindered carbon.
Organometallic Opening: Grignard and organolithium reagents open epoxides at the less hindered carbon.
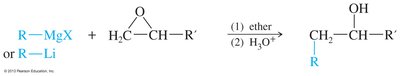
Method | Reagents | Product |
|---|---|---|
Williamson Ether Synthesis | Alkoxide + Alkyl Halide | Ether (SN2) |
Bimolecular Dehydration | Alcohol + Acid | Ether |
Alkoxymercuration-Demercuration | Alkene + Alcohol + Hg(OAc)2 | Ether (Markovnikov) |
Epoxide Synthesis | Alkene + Peroxyacid | Epoxide |
Additional info: The notes above expand on the brief points in the original materials, providing academic context, definitions, and examples for each topic. Images included are directly relevant to the explanations and reinforce key concepts in ether and epoxide chemistry.
