 Back
BackExamples of Pericyclic Reactions in Organic Synthesis
Study Guide - Smart Notes

Pericyclic Reactions in Organic Synthesis
Pericyclic reactions are a class of organic reactions that proceed via concerted, cyclic transition states. These reactions are fundamental in the construction of complex molecular architectures and are widely used in the synthesis of natural products and pharmaceuticals. The following study notes provide detailed examples of pericyclic reactions, focusing on their mechanisms and synthetic applications.
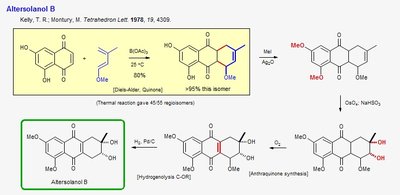
Altersolanol B Synthesis
Altersolanol B is a natural product synthesized via pericyclic reactions, specifically the Diels-Alder reaction. This reaction forms a six-membered ring by the concerted interaction of a diene and a dienophile.
Diels-Alder Reaction: A [4+2] cycloaddition between a conjugated diene and a dienophile, resulting in a cyclohexene ring.
Regioselectivity: The reaction produces a major isomer due to electronic and steric effects.
Further Transformations: The product undergoes methylation, hydrogenation, and oxidation to yield Altersolanol B and related compounds.
Key Equation:
Example: Synthesis of Altersolanol B demonstrates the utility of pericyclic reactions in constructing complex polycyclic frameworks.
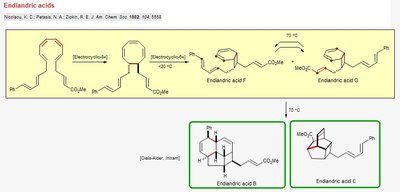
Endiandric Acids Synthesis
Endiandric acids are synthesized through a series of pericyclic reactions, including electrocyclic ring closures and Diels-Alder cycloadditions. These reactions enable the rapid assembly of polycyclic structures from linear precursors.
Electrocyclic Reaction: A pericyclic process where a π system undergoes ring closure or opening, governed by orbital symmetry rules.
Diels-Alder Reaction: Used to form additional rings in the endiandric acid framework.
Thermal Conditions: The reactions are often thermally induced, favoring specific isomers.
Key Equation:
Example: Synthesis of Endiandric acid B and C illustrates the power of pericyclic reactions in natural product synthesis.
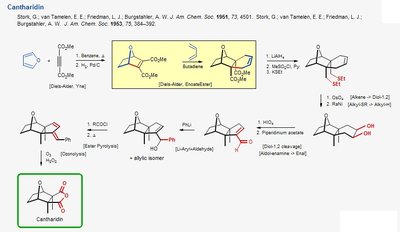
Cantharidin Synthesis
Cantharidin is a bicyclic compound synthesized using pericyclic reactions, including the Diels-Alder reaction and subsequent functional group transformations.
Diels-Alder Reaction: Formation of the bicyclic core from a diene and a dienophile.
Functionalization: Steps such as reduction, oxidation, and esterification are used to elaborate the structure.
Key Equation:
Example: The synthesis of Cantharidin highlights the efficiency of pericyclic reactions in constructing complex ring systems.
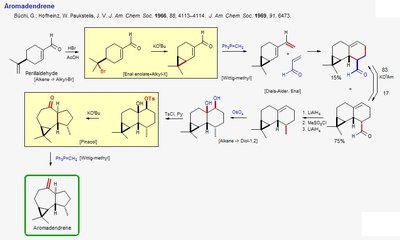
Aromadendrene Synthesis
Aromadendrene is a sesquiterpene synthesized through pericyclic reactions, including alkene-alkyne cycloadditions and Diels-Alder reactions. These steps are crucial for forming the fused ring system characteristic of aromadendrene.
Alkene-Alkyne Cycloaddition: A pericyclic reaction forming new rings from unsaturated precursors.
Diels-Alder Reaction: Used to further build the polycyclic structure.
Functional Group Manipulation: Includes oxidation, reduction, and methylation to achieve the final product.
Key Equation:
Example: The synthesis of Aromadendrene demonstrates the versatility of pericyclic reactions in terpene chemistry.
Summary Table: Types of Pericyclic Reactions
The following table summarizes the main types of pericyclic reactions illustrated in the examples above:
Reaction Type | Mechanism | Example Compound | Key Features |
|---|---|---|---|
Diels-Alder | [4+2] cycloaddition | Altersolanol B, Cantharidin | Forms six-membered rings, regioselective |
Electrocyclic | Ring closure/opening | Endiandric acids | Thermally induced, orbital symmetry rules |
Alkene-Alkyne Cycloaddition | Cycloaddition | Aromadendrene | Forms fused rings, versatile |
Additional info: Pericyclic reactions are governed by the Woodward-Hoffmann rules, which predict the stereochemistry and feasibility of these reactions based on orbital symmetry. These reactions are essential tools in organic synthesis for constructing complex molecular architectures efficiently.
