 Back
BackFamilies of Carbon Compounds, Functional Groups, and Physical Properties (with IR Spectroscopy)
Study Guide - Smart Notes

Families of Carbon Compounds
Hydrocarbons
Hydrocarbons are organic compounds composed solely of carbon and hydrogen atoms. They are classified based on the types of bonds between carbon atoms and their structural features.
Alkanes: Saturated hydrocarbons with only single bonds between carbon atoms. They can form chains or rings and are generally less reactive than other hydrocarbons.
Alkenes: Unsaturated hydrocarbons containing at least one carbon–carbon double bond. They are more reactive than alkanes due to the presence of the double bond.
Alkynes: Unsaturated hydrocarbons with at least one carbon–carbon triple bond. Alkynes are even more reactive than alkenes.
Aromatic Compounds: Contain a special type of ring structure, most commonly the benzene ring, which is stabilized by resonance.
Example: Methane (alkane), ethene (alkene), ethyne (alkyne), benzene (aromatic).





Functional Groups
Definition and Importance
Functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. They define the family to which a compound belongs and influence its physical and chemical properties.
Alkyl Groups: Derived from alkanes by removing a hydrogen atom (e.g., methyl, ethyl).
Phenyl and Benzyl Groups: Phenyl is derived from benzene, benzyl includes a methylene group attached to benzene.
Alkyl Halides: Contain a halogen atom (F, Cl, Br, I) attached to an alkyl group.
Alcohols: Contain a hydroxyl group (–OH) attached to an alkyl group.
Ethers: Have an oxygen atom connected to two alkyl or aryl groups (R–O–R).
Amines: Contain nitrogen atoms bonded to alkyl groups (R–NH2, R2NH, R3N).
Aldehydes and Ketones: Contain a carbonyl group (C=O); aldehydes have it at the end, ketones in the middle.
Carboxylic Acids, Esters, Amides: Contain carboxyl, ester, or amide functional groups, respectively.
Nitriles: Contain a cyano group (–C≡N).
Example: Ethanol (alcohol), acetone (ketone), acetic acid (carboxylic acid).





Intermolecular Forces and Physical Properties
Types of Intermolecular Forces
Intermolecular forces are the forces that act between molecules, affecting their physical properties such as melting point, boiling point, and solubility.
Ionic Forces: Very strong forces between ions (e.g., sodium chloride lattice).
Ion-Dipole Forces: Moderate strength, occur between ions and polar molecules.
Hydrogen Bonds: Moderate to weak, but stronger than other dipole interactions; occur when hydrogen is bonded to N, O, or F.
Dipole-Dipole Forces: Weak, occur between polar molecules.
Dispersion Forces (London Forces): Variable strength, present in all molecules, especially nonpolar ones.
Boiling Points
The boiling point of a compound depends on the strength of intermolecular forces and is pressure dependent. Stronger forces lead to higher boiling points.

Solubility
Solubility is determined by the principle "like dissolves like"—compounds with similar polarity dissolve in each other. Water solubility is often defined as dissolving at least 3 g in 100 mL of water.

Polar Covalent Bonds and Dipole Moments
Bond Polarity
Bond polarity arises from differences in electronegativity between atoms. Polar covalent bonds have unequal sharing of electrons, resulting in partial charges (δ+ and δ–).
Dipole Moment: A measure of bond polarity, expressed in debyes (D). Calculated as where is the distance and is the charge.
Examples: Water (1.85 D), Methanol (1.70 D), Chloroethane (1.87 D).
Dipole Moments in Alkenes
Geometric isomers (cis/trans) of alkenes can have different dipole moments due to the arrangement of substituents.


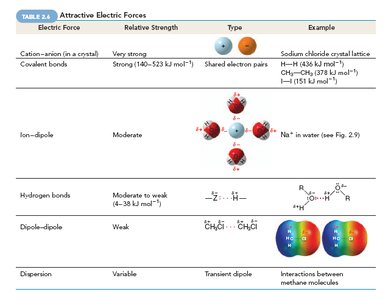
Summary of Attractive Electric Forces
The table below summarizes the main types of attractive electric forces found in organic compounds:
Electric Force | Relative Strength | Type | Example |
|---|---|---|---|
Cation–anion (in a crystal) | Very strong | Shared electron pairs | Sodium chloride crystal lattice |
Covalent bonds | Strong (140–523 kJ/mol) | Shared electron pairs | H–H, CH4 |
Ion–dipole | Moderate | Ion–dipole interaction | Na+ in water |
Hydrogen bonds | Moderate to weak (4–38 kJ/mol) | Hydrogen bonding | R–O–H...O |
Dipole–dipole | Weak | Dipole–dipole interaction | CH3Cl, CH3Br |
Dispersion | Variable | Transient dipole | Interactions between methane molecules |
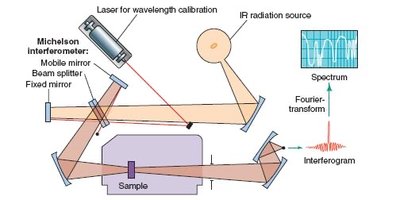
Infrared (IR) Spectroscopy
Principles and Instrumentation
IR spectroscopy is a technique used to identify functional groups in organic compounds by measuring the absorption of infrared radiation, which causes molecular vibrations. The position of absorption bands is specified in wavenumbers (cm-1).
Key Equations: , , ,
Fingerprint Region: 1500–500 cm-1 is unique for each compound.
Peak Shape: The shape and position of peaks are important for functional group identification.
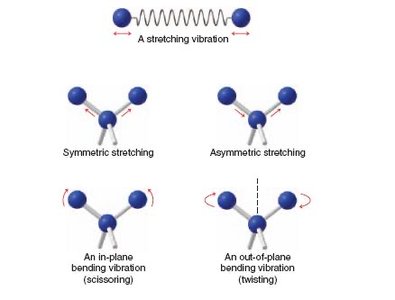
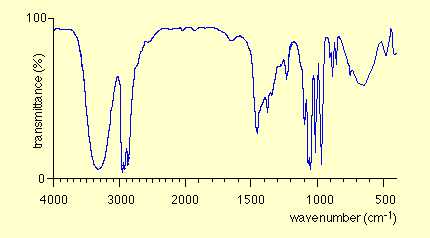

Characteristic IR Absorptions
Different functional groups absorb IR radiation at characteristic frequencies:
Alkyl C–H: 2853–2962 cm-1 (medium–strong)
Alkenyl C–H: 3010–3095 cm-1 (medium)
Alkynyl C–H: ~3300 cm-1 (strong)
Aromatic Ar–H: ~3300 cm-1 (variable)
Alcohols O–H: 3200–3550 cm-1 (broad, strong)
Carbonyl C=O: 1630–1780 cm-1 (strong)
Amines N–H: 3300–3500 cm-1 (medium)
Nitriles C≡N: 2220–2260 cm-1 (medium)
Interpreting IR Spectra
IR spectra are used to identify functional groups by analyzing the position and shape of absorption bands. Examples include spectra for octane, toluene, and 1-heptyne.
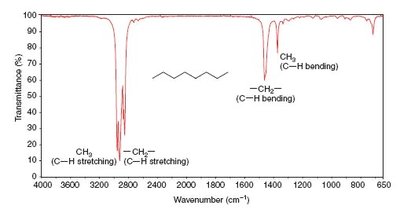
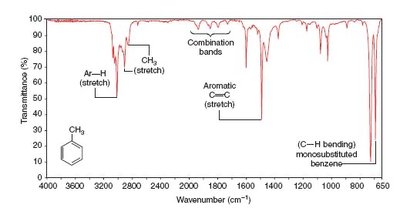


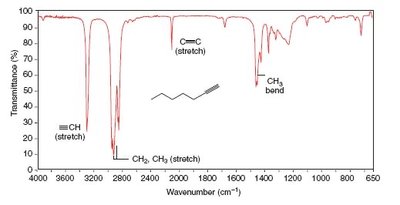



Practice and Further Study
Students are encouraged to use spectral databases such as SDBS to practice identifying functional groups in IR spectra.
Additional info: The notes cover all major families of organic compounds, their functional groups, physical properties, and IR spectroscopy, which are central to organic chemistry at the college level.
