 Back
BackFoundations of Organic Chemistry: Atomic Structure, Bonding, and Molecular Geometry
Study Guide - Smart Notes

Introduction to Atomic Structure and Orbitals
Atomic Structure
Atoms are the fundamental building blocks of matter, consisting of a nucleus (containing protons and neutrons) surrounded by electrons in defined energy levels or shells. The arrangement of these subatomic particles determines the chemical properties of each element.
Protons: Positively charged particles in the nucleus.
Neutrons: Neutral particles in the nucleus.
Electrons: Negatively charged particles in orbitals around the nucleus.
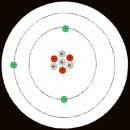
Atomic Orbitals
Electrons occupy regions of space called atomic orbitals. The most common types are s and p orbitals:
s-Orbital: Spherical in shape and can hold up to two electrons.
p-Orbitals: Dumbbell-shaped, oriented along the x, y, and z axes, each holding up to two electrons (six total for all three p orbitals).
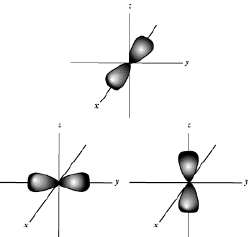
Energy Levels and Electron Configuration
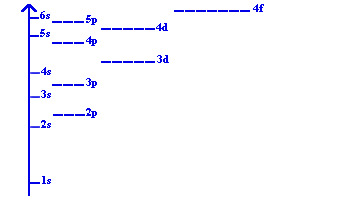
Electrons fill orbitals in order of increasing energy, following the Aufbau principle, Pauli exclusion principle, and Hund's rule. The energy diagram below shows the relative energies of different orbitals:
Ionic and Covalent Bonding
Ionic Bonding
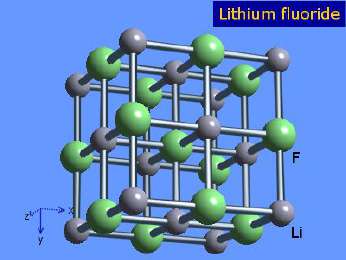
Ionic bonds form when electrons are transferred from one atom to another, resulting in oppositely charged ions that attract each other. For example, lithium fluoride (LiF) forms when lithium donates an electron to fluorine:
Li+: Cation (lost an electron)
F-: Anion (gained an electron)
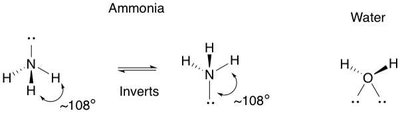
Covalent Bonding and Molecular Geometry
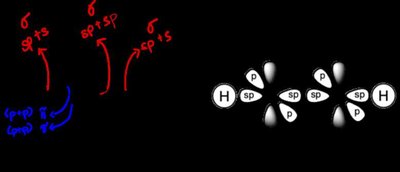
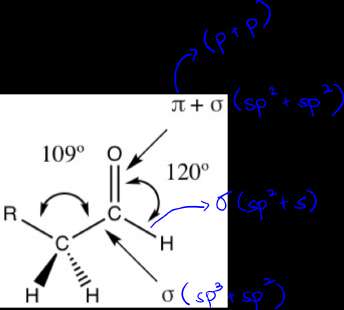
Covalent bonds involve the sharing of electron pairs between atoms. The geometry of molecules is determined by the hybridization of atomic orbitals:
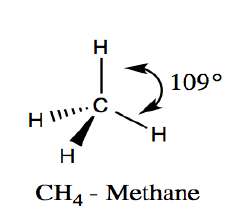
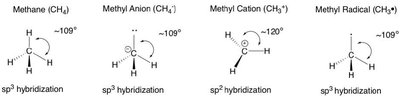
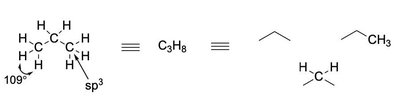
sp3 Hybridization: Tetrahedral geometry, bond angles ~109.5° (e.g., methane, CH4).
Electron Configuration and Hybridization
Electron Configuration
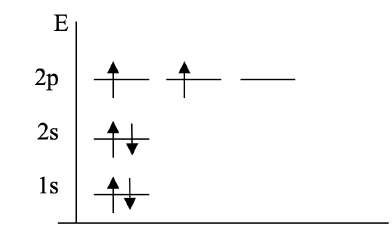
Electrons fill orbitals according to specific rules, resulting in characteristic electron configurations for each element. The diagram below shows the filling of 1s, 2s, and 2p orbitals for a second-row element:
Hybridization
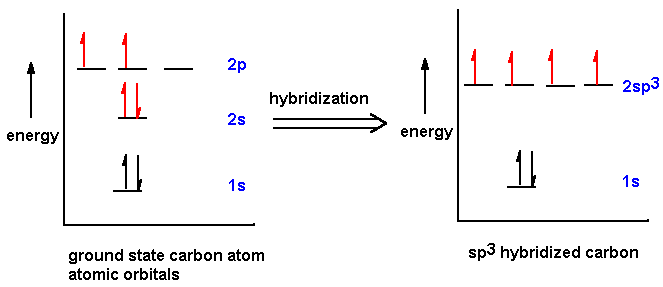
Hybridization is the mixing of atomic orbitals to form new hybrid orbitals suitable for bonding:
sp3 Hybridization: Four equivalent orbitals, tetrahedral geometry (e.g., methane).
sp2 Hybridization: Three equivalent orbitals, trigonal planar geometry (e.g., alkenes).
sp Hybridization: Two equivalent orbitals, linear geometry (e.g., alkynes).
Molecular Structure and Functional Groups
Molecular Geometry and Bond Angles
The geometry of molecules is determined by the number of electron domains around the central atom and the type of hybridization. For example, methane (CH4) is tetrahedral, while ethene (C2H4) is planar due to sp2 hybridization.
Examples of Organic Molecules
Alkanes: Saturated hydrocarbons with only single bonds (sp3 hybridization).
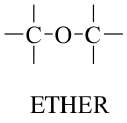
Ethers: Contain an oxygen atom connected to two alkyl or aryl groups.
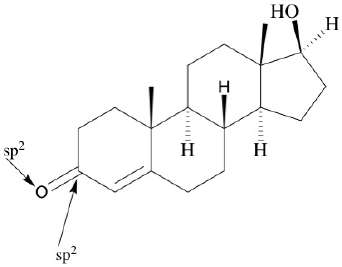
Steroids: Polycyclic molecules with multiple functional groups and stereocenters.
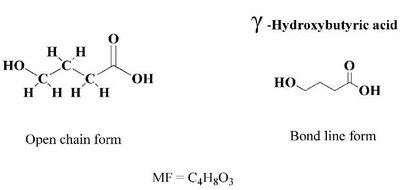
Carboxylic Acids: Contain a carboxyl group (-COOH).
Intermolecular Forces and Physical Properties
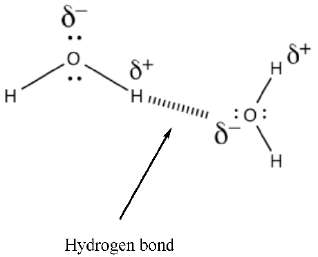
Hydrogen Bonding
Hydrogen bonds are strong intermolecular forces that occur when hydrogen is bonded to highly electronegative atoms (N, O, F). These interactions significantly affect boiling and melting points.
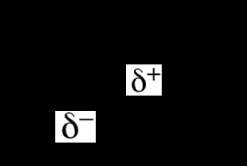
Dipole-Dipole and London Dispersion Forces
Other intermolecular forces include dipole-dipole interactions (between polar molecules) and London dispersion forces (temporary dipoles in all molecules, especially significant in nonpolar compounds).
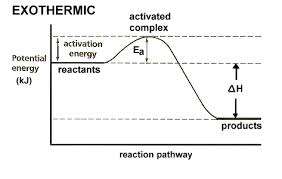
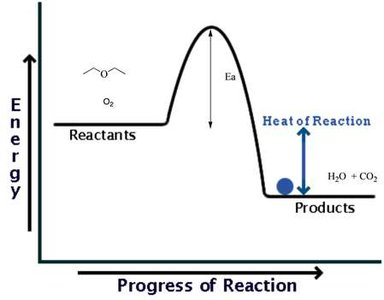
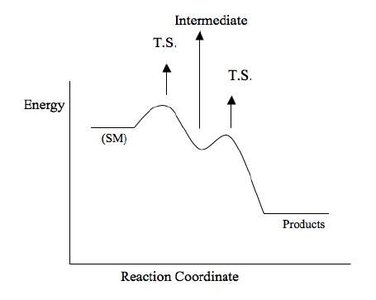
Thermodynamics and Reaction Energy Profiles
Exothermic and Endothermic Reactions
Chemical reactions involve changes in energy. Exothermic reactions release energy (negative ΔH), while endothermic reactions absorb energy (positive ΔH). The energy profile of a reaction shows the activation energy (Ea) and the difference in energy between reactants and products.
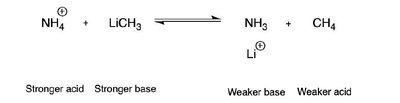
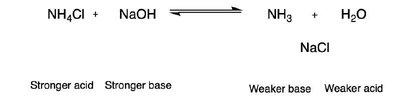
Acids, Bases, and Reaction Mechanisms
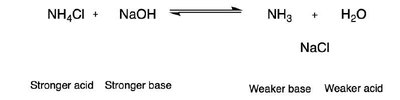
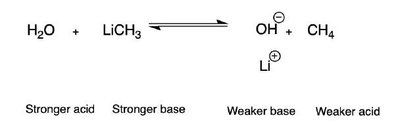
Acid-Base Equilibria
Acid-base reactions are fundamental in organic chemistry. The strength of acids and bases is compared using pKa values. The direction of acid-base equilibria can be predicted by comparing the relative strengths of acids and bases.
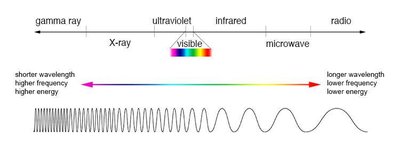
Electromagnetic Radiation and Spectroscopy
Wavelength, Frequency, and Energy
Electromagnetic radiation is characterized by its wavelength (λ), frequency (ν), and energy (E). The relationship is given by:
Shorter wavelengths correspond to higher energy and frequency.
Summary Table: Key Bonding and Hybridization Concepts
Concept | Description | Example |
|---|---|---|
sp3 Hybridization | Tetrahedral geometry, 109.5° bond angles | Methane (CH4) |
sp2 Hybridization | Trigonal planar geometry, 120° bond angles | Ethene (C2H4) |
sp Hybridization | Linear geometry, 180° bond angles | Acetylene (C2H2) |
Ionic Bonding | Electron transfer, formation of ions | Lithium fluoride (LiF) |
Covalent Bonding | Electron sharing, formation of molecules | Methane (CH4) |
Additional info: This guide covers foundational concepts from atomic structure and bonding to molecular geometry and intermolecular forces, providing a basis for further study in organic chemistry.
