Back
BackFree Radical Chemistry and Radical Polymerization
Study Guide - Smart Notes

Free Radical Chemistry
Introduction to Free Radical Chemistry
Free radical chemistry is a fundamental area in organic chemistry, focusing on reactions that proceed via species with unpaired electrons (radicals). These reactions are crucial for understanding the reactivity of alkanes, halogenation processes, and the synthesis of polymers.
Alkane Reactivity
General Reactivity of Alkanes
Alkanes are generally unreactive due to their strong C–H and C–C bonds. However, they can undergo two main types of reactions without catalysts:
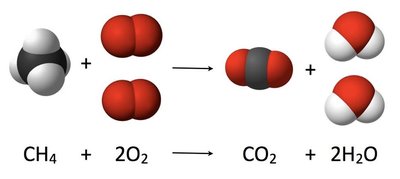
Combustion: Complete oxidation to CO2 and H2O.
Halogenation: Substitution of hydrogen atoms by halogens (chlorination or bromination) via a radical mechanism.
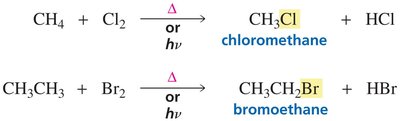
Chlorination and Bromination of Alkanes
Halogenation of alkanes is a radical substitution reaction that requires high temperature or light to initiate.
Chlorination and bromination are the most common radical halogenations.
Fluorination is too violent, and iodination is too unreactive for practical use.
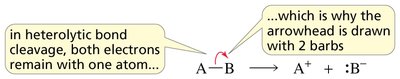
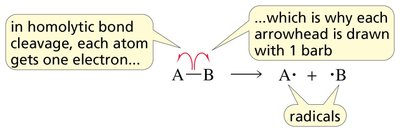
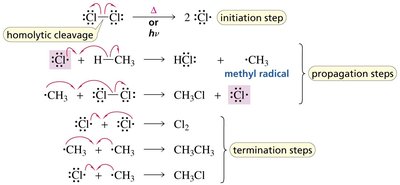
Mechanism of Radical Halogenation
The halogenation of alkanes proceeds via a chain mechanism with three main steps:
Initiation: Homolytic cleavage of a halogen molecule to form two halogen radicals.
Propagation: Radicals react with alkanes to form alkyl radicals and new halogen radicals, propagating the chain.
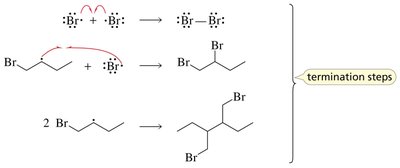
Termination: Two radicals combine to form a stable molecule, ending the chain reaction.
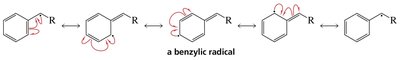
Radical Stability
The stability of alkyl radicals increases with substitution:
Tertiary radical > Secondary radical > Primary radical > Methyl radical
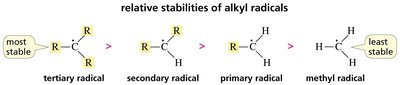
This trend is due to hyperconjugation and the ability of alkyl groups to donate electron density to the radical center.
Why Only Chlorination and Bromination?
Fluorine radicals react too violently, causing uncontrollable reactions.
Iodine radicals are too unreactive to abstract hydrogen atoms from alkanes.
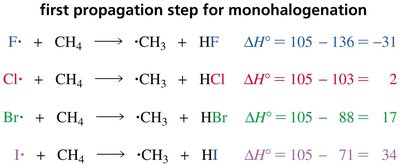
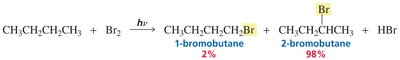
Product Distribution and Selectivity
Bromine is more selective than chlorine in radical halogenation, favoring the formation of the most stable (usually tertiary) radical intermediate.

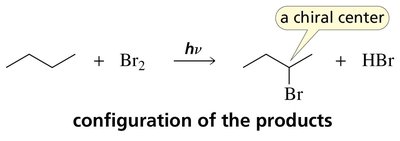
Stereochemistry of Radical Substitution
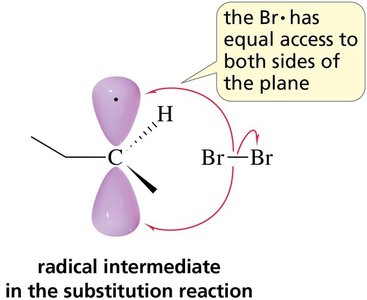
Radical intermediates are planar, allowing attack from either side, which can lead to racemic mixtures if a chiral center is formed.
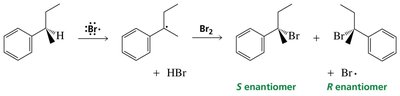
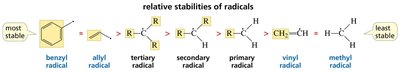
Allylic and Benzylic Radical Substitution
Reactivity at Allylic and Benzylic Positions
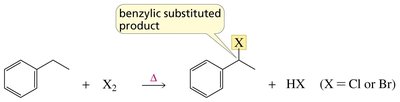
Allylic and benzylic hydrogens are especially reactive in radical halogenation due to resonance stabilization of the resulting radicals.
Allylic radical: Radical adjacent to a double bond, stabilized by resonance.
Benzylic radical: Radical adjacent to a benzene ring, highly stabilized by resonance.
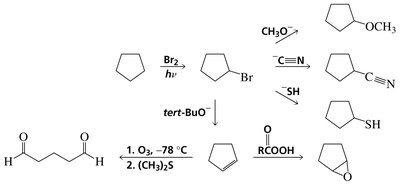

Selective Halogenation at Allylic and Benzylic Positions
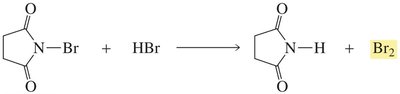
Halogenation at these positions is often performed using N-bromosuccinimide (NBS) to maintain a low concentration of Br2 and avoid addition to double bonds.
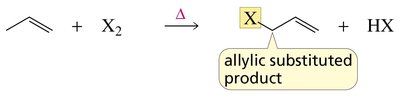
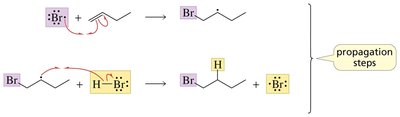
Radical Addition to Alkenes
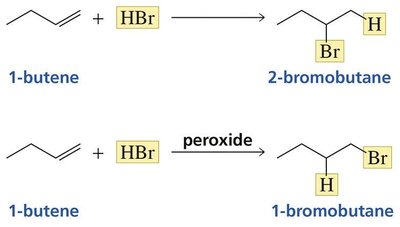
Anti-Markovnikov Addition of HBr
In the presence of peroxides, HBr adds to alkenes via a radical mechanism, resulting in anti-Markovnikov product formation. Peroxides do not affect HCl or HI addition.
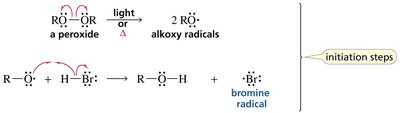
Initiation: Peroxide decomposes to form alkoxy radicals, which generate bromine radicals.
Propagation: Bromine radical adds to the alkene, forming the more stable radical intermediate, which then abstracts a hydrogen from HBr.
Termination: Combination of two radicals.
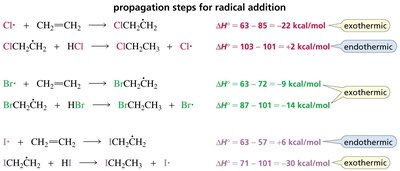
Thermodynamics of Radical Addition
For radical addition to proceed, both propagation steps must be exothermic. This is true for HBr but not for HCl or HI.
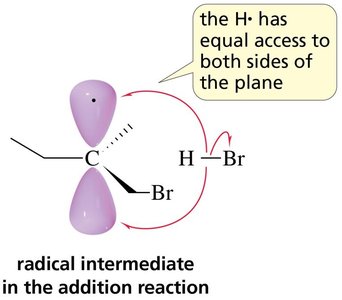
Stereochemistry of Radical Addition
Radical intermediates in addition reactions are planar, allowing attack from either side and leading to racemic mixtures if a chiral center is formed.
Radical Polymerization
Introduction to Polymers
Polymers are large molecules formed by linking repeating units (monomers) through chain reactions. Chain-growth (addition) polymers are synthesized via radical, cationic, or anionic mechanisms.
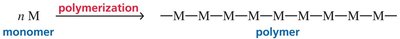
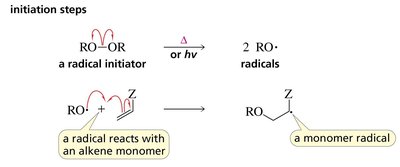
Mechanism of Radical Polymerization
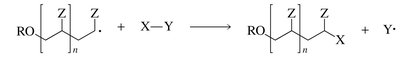
Initiation: Formation of radicals from initiators (e.g., peroxides).
Propagation: Radical adds to monomer, generating a new radical that continues the chain.
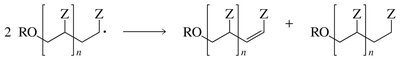
Termination: Two radicals combine or disproportionate, ending the chain.
Chain Transfer: Radical is transferred to another molecule, altering the polymer chain.
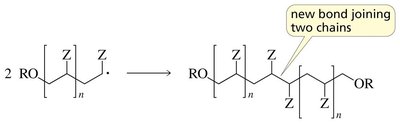
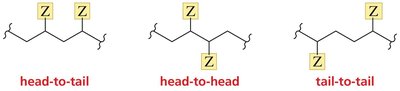
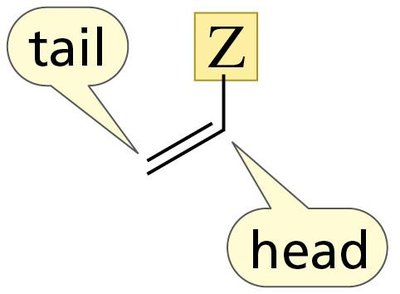
Head-to-Tail Linkage in Polymerization
Monosubstituted ethylenes typically polymerize via head-to-tail addition, which is favored due to electronic effects.
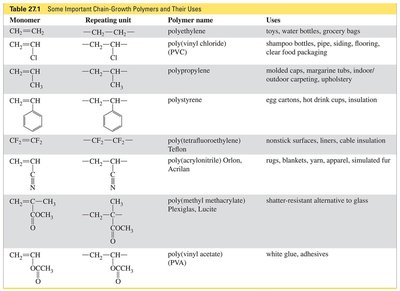
Examples and Applications of Chain-Growth Polymers
Many everyday materials are made from chain-growth polymers, including polyethylene, polyvinyl chloride (PVC), polystyrene, and Teflon. The properties and uses of these polymers depend on their monomer units and structure.
Monomer | Repeating Unit | Polymer Name | Uses |
|---|---|---|---|
CH2=CH2 | –CH2–CH2– | polyethylene | toys, water bottles, grocery bags |
CH2=CHCl | –CH2–CHCl– | poly(vinyl chloride) (PVC) | shampoo bottles, pipes, siding, flooring, clear food packaging |
CH2=CH–CH3 | –CH2–CH(CH3)– | polypropylene | molded cups, margarine tubs, indoor/outdoor carpeting, synthetic fibers |
CH2=CH–Ph | –CH2–CH(Ph)– | polystyrene | egg cartons, hot drink cups, insulation |
CF2=CF2 | –CF2–CF2– | poly(tetrafluoroethylene) Teflon | nonstick surfaces, linens, cable insulation |
CH2=CH–CN | –CH2–CH(CN)– | poly(acrylonitrile) Orlon, Acrilan | rugs, blankets, yarn, apparel, simulated fur |
CH2=C(CH3)COOCH3 | –CH2–C(CH3)(COOCH3)– | poly(methyl methacrylate) Plexiglas, Lucite | shatter-resistant alternative to glass |
CH2=CHOCOCH3 | –CH2–CH(OCOCH3)– | poly(vinyl acetate) (PVA) | white glue, adhesives |
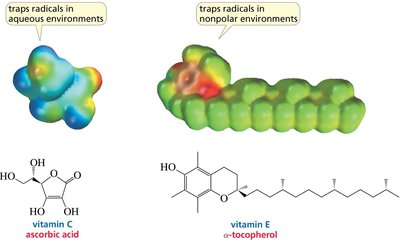
Biological Relevance: Vitamins as Radical Traps
Role of Vitamins C and E
Vitamins C and E act as antioxidants, trapping free radicals and protecting biological systems from oxidative stress. Vitamin C is effective in aqueous environments, while vitamin E functions in nonpolar environments such as cell membranes.