 Back
BackMaterials Chemistry of Solids: Structure, Properties, and Synthesis
Study Guide - Smart Notes

Introduction to Materials Chemistry
Overview of Solids and Their Importance
Materials chemistry focuses on the synthesis, structure, and properties of solid materials, which are fundamental to technological, scientific, and societal advancements. Solids exhibit a wide range of properties, including mechanical, electrical, magnetic, optical, and surface characteristics, all of which are determined by their atomic structure and composition.
Mechanical Properties: Strength, stiffness, hardness, ductility
Electrical Properties: Electronic and ionic conductivity, superconductivity, dielectric constant
Magnetic Properties: Susceptibility, hysteresis, coercivity
Optical Properties: Colour, fluorescence, phosphorescence, non-linear effects
Surface Properties: Surface area, sorption capacity, catalytic activity
Understanding the relationship between structure and properties is essential for designing materials with desired functionalities.
Crystal Structures and Descriptors
Basic Definitions and Structural Concepts
Crystals are solids with translational periodicity in three dimensions. Their structure is described using lattices, unit cells, motifs, and symmetry operations.
Lattice: An infinite array of points with identical environments, defined by lattice vectors (a, b, c) and angles (α, β, γ).
Unit Cell: The smallest repeating unit that reproduces the entire structure via translation.
Motif (Basis): The set of atomic coordinates associated with each lattice point.
Symmetry Operations: Rotations, mirror planes, and centers of symmetry define the space group.
Classic structure types include ionic, metallic, covalent, molecular, and macromolecular crystals.
Example: NaCl (rock salt) structure is cubic, with Na and Cl ions occupying interpenetrating face-centered cubic lattices. All octahedral holes are filled by the counter ion.


Polyhedral and Plane Descriptions
Structures can also be described using coordination polyhedra (e.g., octahedra, tetrahedra) and lattice planes (hkl), which are important for understanding surface properties and diffraction.
Edge-sharing octahedra: Common in NaCl-type structures.
Lattice planes: Indexed by Miller indices (hkl), with symmetry-related sets denoted by curly brackets {hkl}.
Types of Crystals
Classification of Solid Structures
Crystals are classified based on the nature of their bonding and constituent particles:
Ionic: e.g., NaCl, CaF2
Metallic: e.g., Fe-Ni alloys, galvanized zinc
Covalent: e.g., diamond, quartz
Molecular: e.g., water ice, amethyst
Macromolecular: e.g., proteins, polypropylene
Large entities: e.g., nanoparticles, viruses, opals


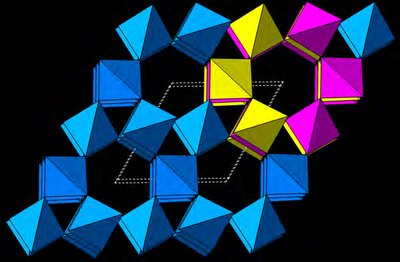
Classic Inorganic Binary Structures
Summary Table of Structure Types
Many binary inorganic compounds adopt archetypal structures based on the filling of polyhedral holes in close-packed lattices.
Structure Type | Coordination | Site Type & Filling | Formula |
|---|---|---|---|
NaCl (Rock Salt) | 6:6 | All octahedral | AB |
ZnS (Wurtzite/Sphalerite) | 4:4 | ½ tetrahedral (alternate) | AB |
CaF2 (Fluorite) | 8:4 | All tetrahedral | AB2 |
CdI2 (Cadmium Iodide) | 6:3 | ½ octahedral (alternate layers) | A2B |
TiO2 (Rutile) | 6:3 | Half octahedral (ordered framework) | AB2 |
Additional info: Many more complex structures are derived from these archetypes by filling different polyhedral holes or by introducing defects.
Intrinsic Properties of Solids
Mechanical Stiffness (Young's Modulus)
Young's modulus (E) quantifies the stiffness of a material and is defined as the ratio of stress to strain in the linear elastic region:
Stress (σ): Force per unit area,
Strain (ε): Fractional elongation,
Young's Modulus:
The modulus is determined by the bond stiffness and packing density of bonds in the lattice.
Example: The Born-Mayer equation relates bond stretching to modulus:
Thermal Expansion
Materials expand with temperature due to the anharmonicity of the bond potential. The linear thermal expansion coefficient (αL) is given by:
Thermal expansion is related to the depth and shape of the bonding curve.
Thermal Conductivity
Thermal conductivity (λ) in solids is carried by lattice vibrations (phonons) and conduction electrons. The relationship is:
Thermal diffusivity (DT) is related to conductivity, heat capacity, and density:
Dielectric Properties
Dielectrics and Polarization
Dielectric materials are electrical insulators that can be polarized by an applied electric field. The capacitance of a parallel plate capacitor is:
Adding a dielectric increases capacitance by the relative permittivity (εr):
Polarization mechanisms include electron cloud distortion, bond stretching, molecular rotation, and ion movement.
Piezoelectric, Pyroelectric, and Ferroelectric Materials
Piezoelectric: Develops polarization under mechanical strain or vice versa.
Pyroelectric: Develops temporary polarization when heated or cooled.
Ferroelectric: Exhibits spontaneous polarization and forms domains below the Curie temperature.
Example: BaTiO3 is a classic ferroelectric perovskite, with high dielectric constant and domain formation.
Phase Diagrams and Thermodynamics
Phase Diagrams
Phase diagrams show the stability regions of different phases as a function of temperature, pressure, and composition. They are essential for understanding phase transitions and guiding synthesis.
Critical Point: The end point of a phase equilibrium curve.
Triple Point: Where three phases coexist.
Composition-Temperature Diagrams: Used for binary systems, showing miscibility, eutectic, and intermediate compounds.
Lever Rule
The lever rule is used to determine the fraction of phases in a two-phase region:
,
Kinetic Effects in Solid Synthesis
Solid State Diffusion and Nucleation
Solid state reactions are often slow due to limited diffusion. Diffusion coefficient (D) follows Arrhenius behavior:
Nucleation requires overcoming an energy barrier, with the critical radius (r*) and nucleation rate determined by thermodynamic and kinetic factors.
Synthetic Techniques for Solids
Liquid Phase Syntheses
Melts: Ingredients are melted and cooled; used for glasses and alloys.
Czochralski Method: Single crystal growth from melt, especially for semiconductors.
Hydrothermal/Solvothermal: Growth in solvent at high temperature and pressure; used for quartz and zeolites.
Precipitation: Formation of solids from solution, e.g., TiO2 production.
Sol-Gel: Formation of gels from molecular precursors, followed by calcination.
Dry Syntheses
Solid State Reactions: Mixing and heating powders; slow due to diffusion.
Mechanosynthesis: Mechanical stress accelerates reactions.
Combustion/Flame Synthesis: Exothermic reactions or flame spray produce powders.
Sealed Tube/Vapour Transport: Thermal gradients drive vapor phase reactions.
Physical and Chemical Vapour Deposition (PVD/CVD): Thin film growth by condensation or decomposition of vapors.
Summary Table: Synthesis Methods and Applications
Method | Scale | Application | Key Property | Structure | Example |
|---|---|---|---|---|---|
Melt | 64M tonne/yr | Aircraft | Specific strength | CCP Al | Al alloys |
Czochralski | $10B/yr | Electronics | Semiconductor | Diamond | Silicon |
Solvothermal | 2000 tonne/yr | Watches | Piezoelectric | α-quartz | Quartz |
Precipitation | 10M tonne/yr | Paint | Optical pigment | Anatase/Rutile | TiO2 |
Sol-gel | $10B/yr | Chromatography | Surface area | Amorphous | Silica |
Solid state reaction | 10M tonne/yr | Capacitors | Ferroelectric | Perovskite | BaTiO3 |
Calcination | 5000M tonne/yr | Construction | Mechanical | Rock salt | Cement (CaO) |
Vapour Transport | 2.5M tonne/yr | Chemical industry | Corrosion resistance | CCP Ni | Nickel |
Combustion | $1.8B/yr | Transformer | Magnetism | Spinel (ferrite) | Ni0.5Zn0.5Fe2O4 |
Flame synthesis | 10M tonne/yr | Paint | Optical pigment | Rutile | TiO2 |
CVD | $2B/yr | LEDs | Semiconductor | Wurtzite | GaN |
Conclusion
Materials chemistry integrates structural, thermodynamic, and kinetic principles to design and synthesize solids with tailored properties. Understanding the interplay between synthesis methods, crystal structure, and intrinsic properties is essential for advancing technology and addressing societal challenges.
