 Back
BackMolecular Structure and Properties in Organic Chemistry: Foundations and Applications
Study Guide - Smart Notes

Introduction to Molecular Structure and Properties
Overview of Organic Chemistry
Organic chemistry is the branch of chemistry focused on molecules containing carbon and hydrogen. Carbon's unique ability to form strong bonds with a variety of elements results in millions of stable molecules, each with distinct physical properties and reactivity. Most organic molecules also contain nitrogen, oxygen, and sometimes halogens, phosphorus, or sulfur.
Lewis Structures and the Octet Rule
Lewis Dot Structures and Valence Electrons
Lewis structures represent atoms and their valence electrons as dots around the element symbol. Valence electrons are the outermost electrons, most reactive, and determine bonding behavior. Atoms form covalent bonds to achieve the stable electron configuration of the nearest noble gas, typically following the octet rule (eight valence electrons for second-row elements). 
Octet Rule: Atoms bond to achieve eight valence electrons.
Examples: Methane (CH4), Ammonia (NH3), Formaldehyde (CH2O).
Formal Charge
Formal charge is the difference between the number of valence electrons in a free atom and the number assigned in a molecule. Formula:
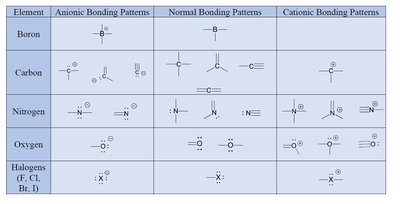
Bonding Patterns: Atoms may have normal, anionic, or cationic bonding patterns depending on their formal charge.
Constitutional Isomers
Constitutional isomers have the same molecular formula but different connectivity of atoms.
Hydrocarbons: Molecules containing only hydrogen and carbon.
Saturated Hydrocarbons: Maximum number of hydrogens (alkanes), formula CnH2n+2.
Unsaturated Hydrocarbons: Contain double or triple bonds (alkenes, alkynes).
VSEPR Theory and Molecular Shape
Valence Shell Electron Pair Repulsion (VSEPR) Theory
VSEPR theory predicts molecular geometry by considering repulsion between electron clouds (bonding and lone pairs). The steric number (number of charge clouds) determines the electron arrangement and bond angles. 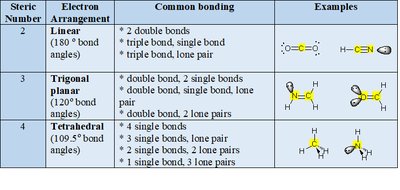
Steric Number 2: Linear (180°)
Steric Number 3: Trigonal planar (120°)
Steric Number 4: Tetrahedral (109.5°)
Wedge-Dash and Bond-Line Structures
Wedge: Bond coming out of the plane.
Dash: Bond going behind the plane.
Zig-zag lines: Used for longer carbon chains.
Electronegativity, Bonding, and Charge Distribution
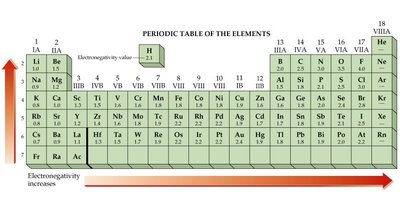
Electronegativity and Bond Types
Electronegativity is the tendency of an atom to attract electrons in a bond. The difference in electronegativity determines bond type:
Ionic Bond: Complete electron transfer (large difference).
Polar Covalent Bond: Unequal sharing (moderate difference).
Nonpolar Covalent Bond: Equal sharing (small difference).
Molecular Charge Distribution
Charge distribution in molecules can be visualized using electrostatic potential maps, which show regions of partial positive and negative charge. 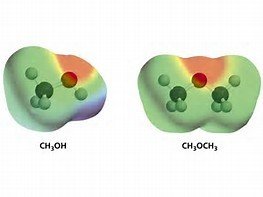
Dipole Moments and Molecular Polarity
Dipole Moment
The dipole moment () is a measure of molecular polarity, calculated as: where is the magnitude of the partial charge and is the distance between charges.
Molecular Dipole: The vector sum of individual bond dipoles, influenced by molecular geometry.
Intermolecular Forces
Types of Intermolecular Forces
Ion-Ion: Strongest, between charged ions.
Hydrogen Bond: Between H and N, O, or F.
Permanent Dipole-Dipole: Between polar molecules.
London Dispersion: Weak, transient dipoles in nonpolar molecules.
Hydrogen Bonding and the Hydrophobic Effect
Hydrogen Bond Donor: Molecule with H attached to N, O, or F.
Hydrogen Bond Acceptor: Molecule with lone pair on N, O, or F.
Hydrophobic Effect: Nonpolar molecules aggregate in water, increasing entropy.
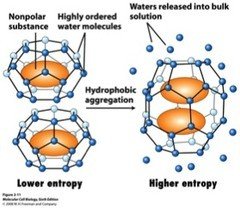
Micelles and Amphipathic Molecules
Detergents are amphipathic, with a hydrophilic head and hydrophobic tail, forming micelles in water. 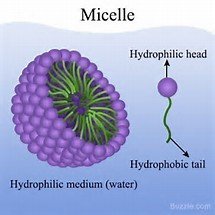
Molecular Properties: Boiling Point, Melting Point, and Solubility
Boiling and Melting Points
Boiling Point: Increases with stronger intermolecular forces and larger surface area.
Melting Point: Influenced by molecular shape and ability to pack into a lattice.
Solubility
Polar solutes: Dissolve in polar solvents.
Nonpolar solutes: Dissolve in nonpolar solvents.
Hydrogen bonding: Enhances solubility in water.
Condensed Formula and Bond-Line Structures
Structural Representations
Condensed Formula: Groups atoms together (e.g., CH3CH2CH2CH3).
Bond-Line Structure: Shows carbon skeleton as zig-zag lines, omits C and H atoms for simplicity.
Primary, Secondary, Tertiary, Quaternary Carbons: Classified by the number of other carbons attached.
Functional Groups in Organic Molecules
Classification by Functional Group
Functional groups are recurring bonding patterns that define the physical properties and chemical reactivity of organic molecules. 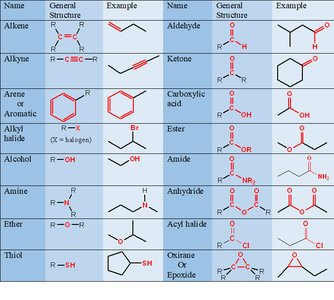
Examples: Alkene, Alkyne, Aromatic, Alcohol, Amine, Ether, Thiol, Aldehyde, Ketone, Carboxylic acid, Ester, Amide, Anhydride, Acyl halide, Epoxide.
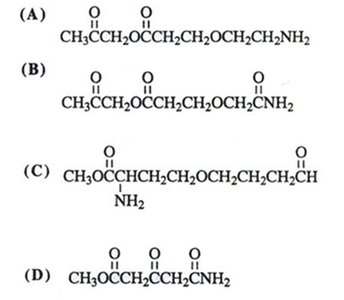
Functional Group Identification Practice
Practice identifying and labeling functional groups in complex molecules.
Summary Table: Bonding Patterns of Common Elements
Element | Anionic Bonding Patterns | Normal Bonding Patterns | Cationic Bonding Patterns |
|---|---|---|---|
Boron | -B | B | B+ |
Carbon | -C | C | C+ |
Nitrogen | -N | N | N+ |
Oxygen | -O | O | O+ |
Halogens | -X | X | X+ |
Summary Table: Electron Arrangement and Steric Number
Steric Number | Electron Arrangement | Common Bonding | Examples |
|---|---|---|---|
2 | Linear (180°) | 2 double bonds, triple bond/single bond, triple bond/lone pair | CO2, HCN |
3 | Trigonal planar (120°) | Double bond/2 single bonds, double bond/single bond/lone pair | H2CO, CH2NH |
4 | Tetrahedral (109.5°) | 4 single bonds, 3 single bonds/1 lone pair, 2 single bonds/2 lone pairs | CH4, NH3, H2O |
Summary Table: Common Functional Groups
Name | General Structure | Example | Name | General Structure | Example |
|---|---|---|---|---|---|
Alkene | R-CH=CH-R | Ethene | Aldehyde | R-CHO | Acetaldehyde |
Alkyne | R-C≡C-R | Ethyne | Ketone | R-CO-R | Acetone |
Arene | Benzene ring | Benzene | Carboxylic acid | R-COOH | Acetic acid |
Alcohol | R-OH | Ethanol | Ester | R-COOR | Ethyl acetate |
Amine | R-NH2 | Methylamine | Amide | R-CONH2 | Acetamide |
Ether | R-O-R | Diethyl ether | Anhydride | R-CO-O-CO-R | Acetic anhydride |
Thiol | R-SH | Ethanethiol | Acyl halide | R-COCl | Acetyl chloride |
Epoxide | Three-membered ring with O | Oxirane |
Summary Table: Functional Group Identification Practice
Structure | Contains |
|---|---|
A | Amide, Ether, Amine |
B | Amide, Amine |
C | Ether, Amide, Amine |
D | Ether, Amide |
Additional info: These notes provide foundational concepts in molecular structure, bonding, and properties, essential for understanding organic chemistry at the college level. The included tables and images reinforce key ideas and support visual learning.
