 Back
BackNuclear Magnetic Resonance (NMR) Spectroscopy: Principles, Interpretation, and Applications
Study Guide - Smart Notes

Chapter 13: Nuclear Magnetic Resonance (NMR) Spectroscopy
Introduction to Nuclear Magnetic Resonance (NMR) Spectroscopy
NMR spectroscopy is a fundamental analytical technique in organic chemistry, used for elucidating the structure of organic molecules. It relies on the interaction between nuclear spins and an external magnetic field, providing detailed information about the chemical environment of nuclei, especially hydrogen (1H) and carbon (13C).
NMR is the most powerful tool for organic structure determination.
Nuclei with odd atomic numbers or masses possess nuclear spin, generating a magnetic moment.
For 1H, spin states are +1/2 or –1/2.
The spinning nucleus acts as a circulating positive charge, creating a magnetic field.

Principles of NMR: Nuclear Spin States and Resonance
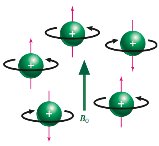
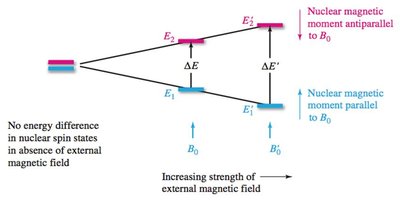
Nuclear spin states are randomly oriented in the absence of an external magnetic field. When a magnetic field is applied, spins align either with or against the field, resulting in different energy levels. Irradiation with radiofrequency causes transitions between these states, known as resonance.
Without a magnetic field, nuclear spins are randomly oriented.
Applying a magnetic field causes alignment and energy splitting between +1/2 and –1/2 states.
The energy difference () is proportional to the magnetic field strength ().
Resonance occurs when nuclei absorb energy and flip spin states.
Gyromagnetic ratio () is a constant for each nucleus.

Equation:
NMR Spectrometer and Its Elements
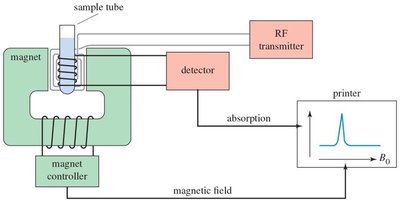
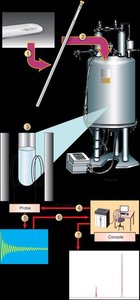
An NMR spectrometer consists of a powerful magnet, a radiofrequency transmitter, and a detector. The sample is placed in a magnetic field, irradiated with radio waves, and the emitted signals are detected and processed to produce an NMR spectrum.
Magnet: Generates a strong, uniform magnetic field.
RF transmitter: Applies radiofrequency pulses to excite nuclei.
Detector: Measures emitted radio waves as nuclei relax.
Modern spectrometers use pulsed radio waves and detect free induction decay.
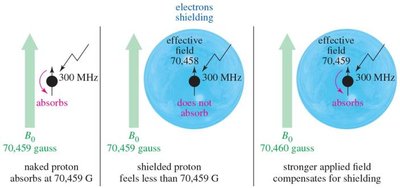
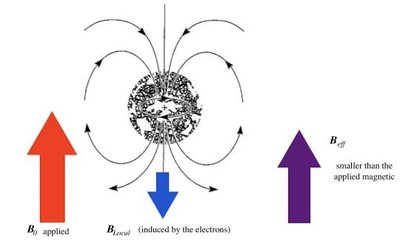
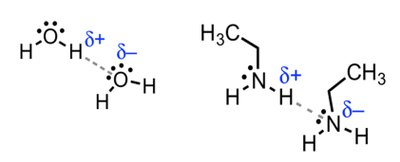
Magnetic Shielding and Effect of Shielding
Protons are surrounded by electrons, which shield them from the external magnetic field. The degree of shielding depends on the electron density around the nucleus, affecting the resonance frequency and chemical shift.
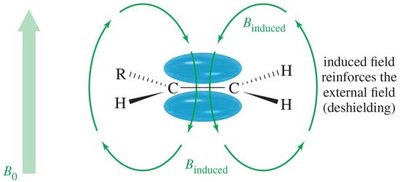
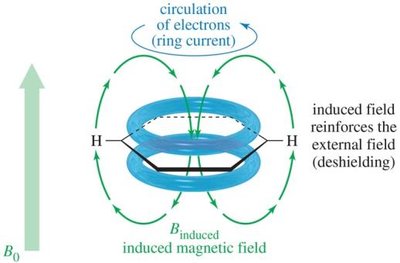
Electrons create an induced magnetic field opposing the external field.
Protons in different environments are shielded to varying extents.
Effective field felt by the nucleus:
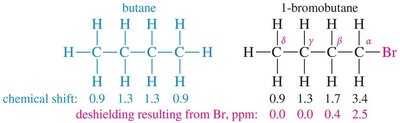
Hydrogen atoms near electronegative groups are deshielded and resonate at higher frequencies (downfield).
Chemical Shift (δ)
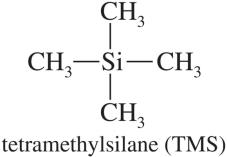
The chemical shift (δ) is a measure of the resonance frequency of a nucleus relative to a reference compound, typically tetramethylsilane (TMS). It reflects the electronic environment of the nucleus and is reported in parts per million (ppm).
δ scale: Arbitrary scale calibrated with TMS at 0 ppm.
TMS: Highly shielded protons, used as internal standard.
Protons near electronegative atoms are deshielded and appear at higher δ values.
Most 1H chemical shifts fall between 0 and 10 ppm.

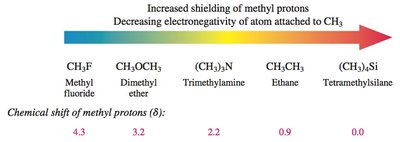

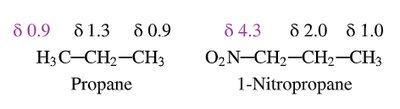
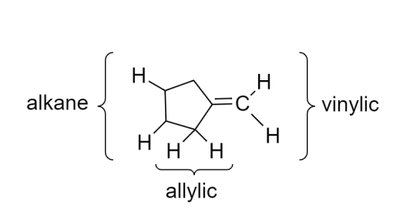
Characteristic Values of Chemical Shifts
Chemical shifts are influenced by the type of proton and its environment. The main regions are:
Saturated region: Methyl, methylene, methine protons (0–3 ppm).
Allylic region: Protons adjacent to double bonds (~2.5–3.5 ppm).
O, N, Halogen-attached region: Protons near electronegative atoms (~3.5–4.5 ppm).
Vinylic protons: Protons on double bonds (~5–6 ppm).
Aromatic region: Protons on aromatic rings (~7–8 ppm).

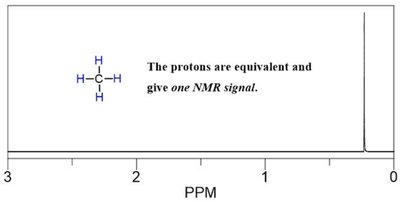
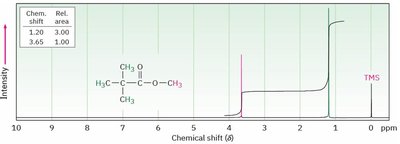
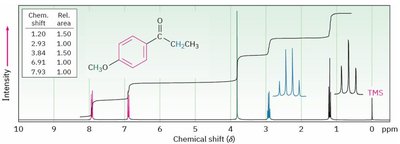
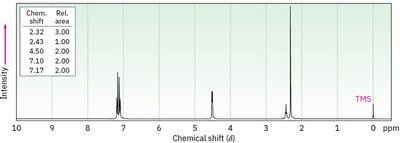
Integration
Integration measures the area under each NMR peak, which is proportional to the number of protons contributing to that signal. It allows determination of the relative number of each type of proton in the molecule.
Integration curves are S-shaped and represent the number of protons.
More protons = more intense signal.
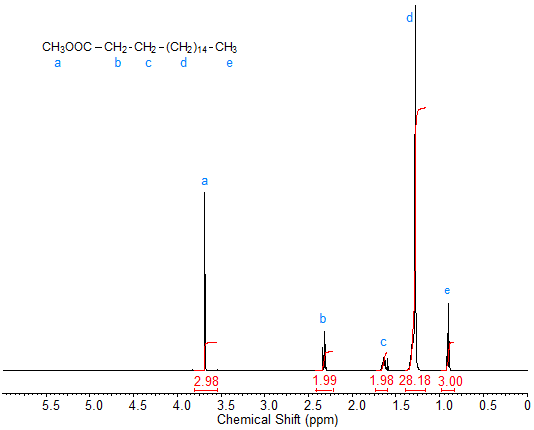
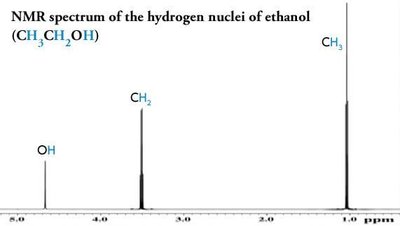
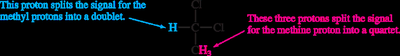
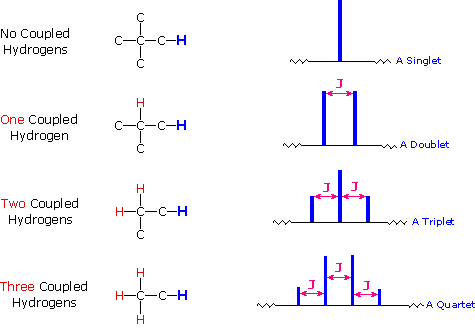
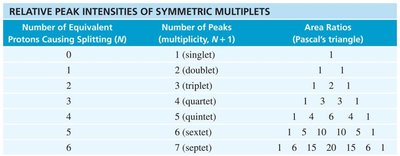
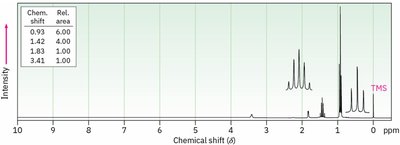
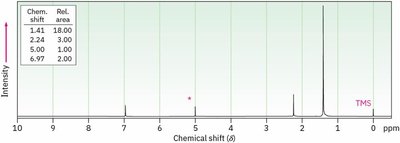
Splitting: Multiplicity and Coupling
Signal splitting occurs due to spin-spin coupling between non-equivalent protons on adjacent atoms. The multiplicity follows the n + 1 rule, where n is the number of equivalent neighboring protons.
Rule 1: Chemically equivalent protons do not split each other's signals.
Rule 2: A proton with n equivalent neighbors is split into n + 1 peaks.
Coupling constant (J): The spacing between split peaks, measured in Hz.
Multiplet intensities follow Pascal's triangle.
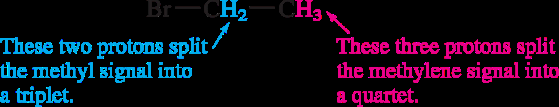
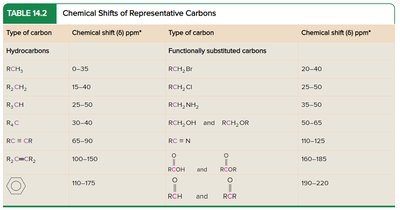
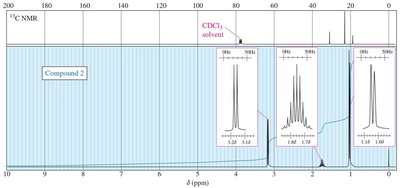
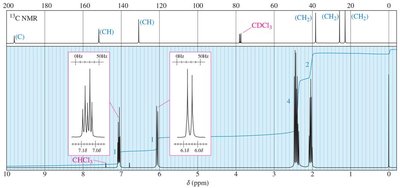
13C NMR Spectroscopy
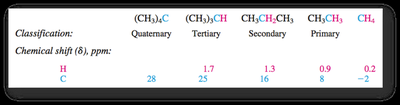
13C NMR provides information about the carbon skeleton of organic molecules. 13C is less abundant than 12C, so spectra require longer acquisition times. Chemical shifts are influenced by electronegativity and hybridization.
Electronegative substituents cause deshielding and higher δ values.
sp2 carbons are less shielded than sp3; sp carbons are more shielded than sp2.
DEPT experiments distinguish between CH, CH2, and CH3 carbons.

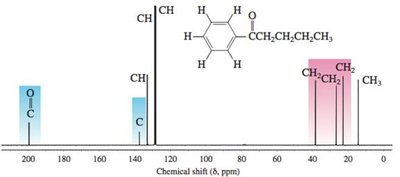
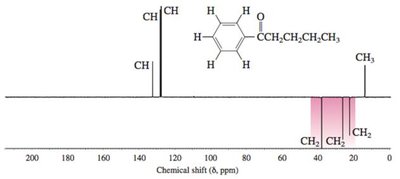
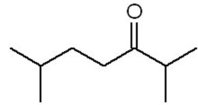
Practice Problems and Applications
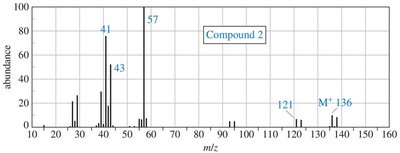
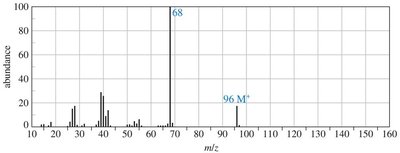
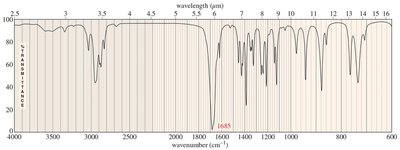
Practice problems involve assigning chemical shifts, interpreting spectra, and proposing structures based on NMR, IR, and mass spectrometry data. These exercises reinforce the principles and applications of NMR spectroscopy in organic chemistry.
Assigning chemical shifts to specific protons in molecules.
Distinguishing isomers using 13C NMR.
Interpreting combined spectroscopic data to deduce molecular structure.
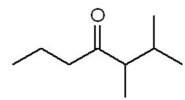
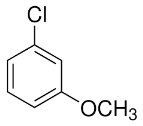
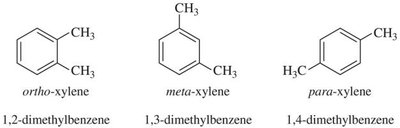
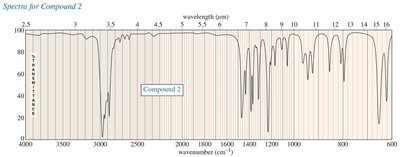
Additional info: These notes expand on brief points from the original materials, providing academic context, definitions, and examples for clarity and completeness. All images included are directly relevant to the adjacent explanations, visually reinforcing key concepts in NMR spectroscopy.
