 Back
BackNucleophilic Substitution: SN1 and SN2 Mechanisms
Study Guide - Smart Notes

Substitution vs. Elimination Reactions
Introduction to Nucleophilic Substitution
Nucleophilic substitution reactions are fundamental processes in organic chemistry, where a nucleophile replaces a leaving group attached to a carbon atom. These reactions are classified mainly as SN1 (unimolecular nucleophilic substitution) and SN2 (bimolecular nucleophilic substitution), each with distinct mechanisms, kinetics, and stereochemical outcomes.
SN2 Reactions
Mechanism of SN2 Reactions
In the SN2 mechanism, the nucleophile attacks the substrate carbon from the side opposite the leaving group, resulting in a single concerted step where bond formation and bond breaking occur simultaneously. This leads to inversion of configuration at the carbon center.
Nucleophile (Nu−): Donates an electron pair to the substrate carbon.
Leaving Group (LG): Departs with the electron pair from the original bond.
Transition State: Both the nucleophile and leaving group are partially bonded to the carbon in a high-energy state.
Stereochemistry: Always results in inversion of configuration (Walden inversion).
Example: The reaction of hydroxide ion (HO−) with methyl bromide (CH3Br) to form methanol (CH3OH) and bromide ion (Br−).
Kinetics of SN2 Reactions
The rate of an SN2 reaction depends on the concentrations of both the nucleophile and the substrate, making it a second-order reaction:
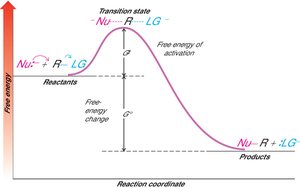
Energy Profile of SN2 Reactions
The reaction proceeds through a single transition state with no intermediates. The free energy diagram shows a single energy barrier corresponding to the transition state.
Solvent Effects in SN2 Reactions
Polar Aprotic Solvents: Best for SN2 reactions (e.g., DMSO, DMF) because they solvate cations but not anions, leaving the nucleophile 'naked' and more reactive.
Polar Protic Solvents: Hinder SN2 by stabilizing the nucleophile through hydrogen bonding.
Leaving Groups in SN2 Reactions
A good leaving group is a weak base that can stabilize the negative charge after departure. Examples include I−, Br−, Cl−, TsO−, MsO−, H2O, and NH3.
Substrate Structure and Reactivity
Methyl > 1° > 2° >> 3°: SN2 reactions are fastest with methyl and primary substrates due to minimal steric hindrance.
Tertiary substrates: Generally unreactive in SN2 due to steric bulk.
SN1 Reactions
Mechanism of SN1 Reactions
SN1 reactions proceed via a two-step mechanism. First, the leaving group departs, forming a carbocation intermediate. Then, the nucleophile attacks the planar carbocation, which can occur from either side, leading to racemization if the center is chiral.
Step 1 (Rate-Determining): Loss of the leaving group to form a carbocation.
Step 2: Nucleophilic attack on the carbocation.
Stereochemistry: Results in a racemic mixture due to planar carbocation intermediate.
Kinetics of SN1 Reactions
The rate of an SN1 reaction depends only on the concentration of the substrate:
The nucleophile does not participate in the rate-determining step.
Carbocation Stability
Order of Stability: 3° > 2° >> 1° > methyl
Stabilization: Carbocations are stabilized by inductive effects (electron-donating groups) and resonance (delocalization of charge).
Solvent Effects in SN1 Reactions
Polar Protic Solvents: Accelerate SN1 reactions by stabilizing the carbocation and the leaving group through solvation (e.g., water, alcohols).
Nucleophiles and Leaving Groups
Nucleophiles
Typically Lewis bases with a lone pair of electrons.
Can be neutral or negatively charged.
Examples: HO−, RO−, CN−, NH3, H2O.
Leaving Groups
Must be able to leave as a stable, weakly basic molecule or ion.
Order of leaving group ability: I− > Br− > Cl− > F−.
Comparison of SN1 and SN2 Reactions
Summary Table: Factors Favoring SN1 vs. SN2
Factor | SN1 | SN2 |
|---|---|---|
Substrate | 3° (requires formation of a relatively stable carbocation) | Methyl > 1° > 2° (requires unhindered substrate) |
Nucleophile | Weak Lewis base, neutral nucleophile, nucleophile may be the solvent (solvolysis) | Strong Lewis base, rate increased by high concentration of nucleophile |
Solvent | Polar protic (e.g., alcohols, water) | Polar aprotic (e.g., DMF, DMSO) |
Leaving group | I− > Br− > Cl− > F− (the weaker the base after the group departs, the better the leaving group) | I− > Br− > Cl− > F− (the weaker the base after the group departs, the better the leaving group) |

Key Points
SN1 is favored by: Stable carbocation formation, weak nucleophile, polar protic solvent.
SN2 is favored by: Unhindered substrate, strong nucleophile, high nucleophile concentration, polar aprotic solvent.
Summary
Understanding the differences between SN1 and SN2 mechanisms is crucial for predicting the outcomes of nucleophilic substitution reactions. Key factors include substrate structure, nucleophile strength, solvent type, and leaving group ability. Mastery of these concepts enables the rational design and analysis of organic reactions.
