 Back
BackChapter 5 part 2 Optical Activity, Chirality, and Stereoisomerism in Organic Chemistry
Study Guide - Smart Notes

Optical Activity and Chirality
Introduction to Chirality
Chirality is a fundamental concept in organic chemistry, describing molecules that are non-superimposable on their mirror images. Such molecules are called chiral, and their mirror images are known as enantiomers. Chirality is crucial because it leads to different interactions with biological systems and polarized light.
Chiral molecules have at least one carbon atom bonded to four different groups (a stereocenter).
Enantiomers are pairs of molecules that are non-superimposable mirror images of each other.
Chirality is analogous to handedness: just as left and right hands are mirror images but not identical, so are enantiomers.
Optical Activity
Optical activity refers to the ability of chiral compounds to rotate plane-polarized light. This property is used to distinguish between enantiomers and assess the purity of chiral substances.
Specific rotation ([α]): The angle by which a chiral molecule rotates plane-polarized light, measured in degrees.
Clockwise rotation is defined as positive (+), or dextrorotatory (d).
Counterclockwise rotation is negative (−), or levorotatory (l).
The specific rotation is calculated using the formula:
Where is the observed rotation, is the concentration (g/mL), and is the path length (dm).
Enantiomers rotate light to the same degree but in opposite directions.
Biological Importance of Chirality
Chirality is essential in biology because many biomolecules (proteins, sugars, DNA, steroids, hormones) are chiral. Enantiomers can interact differently with biological systems, leading to:
Different smells or tastes
Different efficacy or potency of drugs
One enantiomer being therapeutic, the other toxic
Racemic Mixtures
A racemic mixture contains equal amounts of both enantiomers. Such mixtures are optically inactive because the rotations cancel each other out, resulting in a net zero rotation of polarized light.
Racemic mixtures are often produced in chemical synthesis.
Separating enantiomers (resolution) can be challenging and costly.
Enzymes in nature often produce only one enantiomer (stereoselectivity).
Measuring Optical Purity and Enantiomeric Excess
When a mixture contains unequal amounts of enantiomers, its optical purity or enantiomeric excess (ee) can be calculated:
Optical Purity (O.P.):
Enantiomeric Excess (ee):
Enantiomeric excess is more commonly used than optical purity.
Stereoisomerism: Diastereomers and Meso Compounds
Diastereomers
Diastereomers are stereoisomers that are not mirror images of each other. They have different physical and chemical properties, such as boiling/melting points, crystallinity, and reactivity.
Unlike enantiomers, diastereomers behave differently even in achiral environments.
They arise when a molecule has two or more stereocenters.
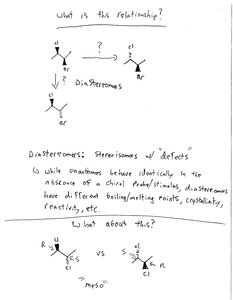
Meso Compounds
A meso compound is an achiral molecule that contains multiple stereocenters but has an internal plane of symmetry, making it superimposable on its mirror image. Meso compounds do not exhibit optical activity.
Meso compounds are a special case where a molecule with stereocenters is achiral due to symmetry.
Example: Tartaric acid has two stereocenters but one meso form.
Multiple Stereocenters and Isomer Count
Molecules with multiple stereocenters can have several stereoisomers. The maximum number of stereoisomers is , where is the number of stereocenters, but this number is reduced if meso forms are present.
Enantiomers: Non-superimposable mirror images
Diastereomers: Stereoisomers that are not mirror images
Meso compounds: Achiral despite having stereocenters
Summary Table: Types of Stereoisomers
Type | Mirror Image? | Optical Activity | Physical Properties |
|---|---|---|---|
Enantiomers | Yes (non-superimposable) | Equal and opposite | Identical (except for optical activity) |
Diastereomers | No | Different | Different |
Meso Compounds | Superimposable | None | Unique |
Practice Problem Example
Problem: 6.00 g of 2-butanol is dissolved in 40 mL of H2O and placed in a 200 mm tube. The observed rotation is -4.05°. What is the specific rotation ([α])? Is this the d or l isomer? What would be the [α] of the other isomer?
Given: , g/mL, mm dm
Calculation:
This is the l (levorotatory) isomer. The d isomer would have .
Key Takeaways
Chirality and optical activity are central to understanding organic molecules and their interactions.
Enantiomers have identical physical properties except for their effect on polarized light and interactions with other chiral substances.
Diastereomers have different physical and chemical properties.
Meso compounds are achiral despite having stereocenters.
Understanding these concepts is essential for predicting molecular behavior in chemical and biological systems.
