 Back
BackOrganic Chemistry Midterm Study Guidance – Resonance, Hybridization, Acidity, Isomerism, and Conformations
Study Guide - Smart Notes

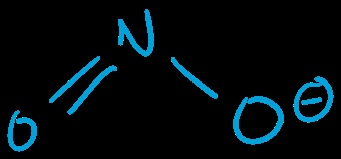
Q1. Draw the nitrite ion (NO2–), including all important resonance structures (assume N is the central atom).
Background
Topic: Resonance and Molecular Structure
This question tests your understanding of resonance structures, formal charges, and molecular geometry for polyatomic ions.
Key Terms and Formulas:
Resonance structures: Different Lewis structures for the same molecule/ion, showing delocalization of electrons.
Formal charge:
VSEPR theory: Used to predict molecular shape based on electron pair repulsion.
Step-by-Step Guidance
Draw the Lewis structure for NO2– with N as the central atom. Place the two O atoms around N, and add the extra electron for the negative charge.
Assign single and double bonds between N and O, and distribute lone pairs to satisfy the octet rule for each atom.
Calculate formal charges for each atom in both resonance structures. Make sure the sum of formal charges equals –1.
Draw both resonance structures, showing the movement of electrons (curved arrows) between them.
Try solving on your own before revealing the answer!
Final Answer:
The nitrite ion has two major resonance structures, each with a double bond to one oxygen and a single bond to the other, with the negative charge delocalized over the oxygens.
Both resonance structures contribute equally to the overall structure, stabilizing the ion.
Q1a. Indicate the shape of the nitrite ion.
Background
Topic: Molecular Geometry (VSEPR Theory)
This part tests your ability to use VSEPR theory to predict the shape of a molecule based on electron domains.
Key Terms:
Bent: Shape resulting from two bonding domains and one lone pair on the central atom.
Linear: Shape with two bonding domains and no lone pairs.
Trigonal planar: Shape with three bonding domains and no lone pairs.
Step-by-Step Guidance
Count the number of electron domains (bonding and lone pairs) around the central N atom.
Use VSEPR theory to determine the ideal geometry for three electron domains.
Consider the effect of lone pairs on the actual molecular shape.


Try solving on your own before revealing the answer!
Final Answer:
The nitrite ion is bent due to the presence of a lone pair on the nitrogen atom, resulting in a bond angle less than 120°.
Q1b. Bubble each TRUE statement about nitrite ion.
Background
Topic: Polarity, Bond Dipoles, and Acid/Base Properties
This part tests your understanding of bond polarity, molecular polarity, and acid/base strength.
Key Terms:
Bond polarity: Difference in electronegativity between atoms causes dipole moments.
Molecular polarity: Net dipole moment of the molecule.
Conjugate acid/base: Relationship between acids and their bases after proton transfer.
Step-by-Step Guidance
Compare electronegativity of N and O to determine bond polarity and direction of dipole arrows.
Assess whether the overall molecule is polar or nonpolar based on its shape and dipole moments.
Consider the acid/base properties of nitrite ion and its conjugate acid (HNO2).
Try solving on your own before revealing the answer!
Final Answer:
Each N–O bond is polar, bond polarization arrows point toward the O's, and nitrite ion is not a strong base. Its conjugate acid is not a strong acid.
Q1c. Which one best describes hybridization of the N in nitrite ion?
Background
Topic: Hybridization
This part tests your ability to assign hybridization based on electron domain geometry.
Key Terms:
sp2 hybridization: Three electron domains (trigonal planar geometry).
sp3 hybridization: Four electron domains (tetrahedral geometry).
sp hybridization: Two electron domains (linear geometry).
Step-by-Step Guidance
Count the number of electron domains (bonds and lone pairs) around the N atom.
Match the number of domains to the correct hybridization type.
Try solving on your own before revealing the answer!
Final Answer:
The nitrogen atom in nitrite ion is sp2 hybridized, corresponding to three electron domains.
Q1d. Which best describes the charge on each oxygen atom in nitrite ion?
Background
Topic: Formal Charge Calculation
This part tests your ability to assign formal charges to atoms in a polyatomic ion.
Key Terms:
Formal charge: See formula above.
Step-by-Step Guidance
Calculate the formal charge for each oxygen atom in both resonance structures.
Average the charges if resonance delocalizes the negative charge.
Try solving on your own before revealing the answer!
Final Answer:
Each oxygen atom in nitrite ion has a formal charge of –½, due to resonance delocalization of the negative charge.
