 Back
BackOrganic Chemistry Nomenclature and Isomerism Study Guide
Study Guide - Smart Notes

Q9. What is the name of the following compound?
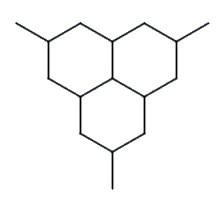
Background
Topic: Alkane Nomenclature
This question tests your ability to apply IUPAC rules for naming branched alkanes. You need to identify the longest carbon chain, number the chain to give substituents the lowest possible numbers, and name and order the substituents correctly.
Key Terms and Formulas
Longest Chain: The main chain of carbon atoms in the molecule.
Substituents: Groups attached to the main chain (e.g., methyl, ethyl, isopropyl).
Numbering: Assign numbers to the main chain so that substituents get the lowest possible numbers.
Alphabetical Order: List substituents alphabetically in the name.
Step-by-Step Guidance
Identify the longest continuous carbon chain in the structure. This will determine the parent name (e.g., octane, decane).
Number the carbon chain from the end that gives the substituents the lowest possible numbers.
Identify and name each substituent group attached to the main chain (e.g., isopropyl, methyl).
Assign the correct numbers to each substituent based on their position on the main chain.
Combine the substituent names and numbers in alphabetical order, followed by the parent chain name.
Try solving on your own before revealing the answer!
Final Answer: 4-isopropyloctane
The longest chain is octane, and the isopropyl group is attached to the fourth carbon. The name follows IUPAC rules for alkane nomenclature.
