 Back
BackOrganic Chemistry Practice Exam – NMR, Bond Strength, and Reaction Mechanisms
Study Guide - Smart Notes

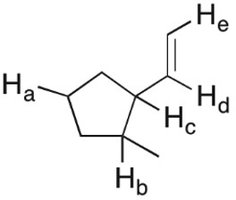
Q28. In the molecule shown, which of the highlighted C-H bonds (labeled from a to e) is expected to have the lowest bond dissociation energy?
Background
Topic: Bond Dissociation Energy (BDE) in Organic Molecules
This question tests your understanding of how the structure and environment of a C-H bond affect its bond dissociation energy, particularly in the context of allylic, vinylic, and alkyl positions in cycloalkenes.
Key Terms and Concepts:
Bond Dissociation Energy (BDE): The energy required to homolytically cleave a bond, producing radicals.
Allylic Hydrogen: A hydrogen atom attached to a carbon adjacent to a double bond (alkene).
Vinylic Hydrogen: A hydrogen atom directly attached to a carbon-carbon double bond.
Alkyl Hydrogen: A hydrogen atom attached to a saturated (sp3) carbon not adjacent to a double bond.
Step-by-Step Guidance
Examine the structure and identify the hybridization and position of each labeled hydrogen (Ha, Hb, Hc, Hd, He).
Recall that allylic C-H bonds (adjacent to a double bond) typically have lower BDEs than vinylic or alkyl C-H bonds due to resonance stabilization of the resulting radical.
Vinylic C-H bonds (directly on the double bond) have higher BDEs because the resulting radical is less stabilized.
Compare the positions: Determine which hydrogen is allylic, which is vinylic, and which are simple alkyl hydrogens.
Predict which labeled hydrogen corresponds to the lowest BDE based on its position relative to the double bond and the potential for resonance stabilization.
Try solving on your own before revealing the answer!
Final Answer: C-Hc (Hydrogen labeled 'c')
The C-H bond labeled 'c' is allylic to the double bond, meaning that homolytic cleavage produces an allylic radical, which is resonance stabilized. This makes its bond dissociation energy the lowest among the options.
