 Back
BackOrganic Chemistry Reasoning Questions: Halogen Compounds, Alcohols, Phenols, Aldehydes, Ketones, and Carboxylic Acids
Study Guide - Smart Notes

Q1. Why is the thionyl chloride method preferred for preparing alkyl chloride from alcohols?
Background
Topic: Preparation of Alkyl Halides from Alcohols
This question tests your understanding of the methods used to convert alcohols to alkyl chlorides, specifically why thionyl chloride (SOCl2) is often chosen.
Key Terms and Formulas
Thionyl chloride (SOCl2)
Alkyl chloride (RCl)
Alcohol (ROH)
Reaction:
Step-by-Step Guidance
Consider the products of the reaction: alkyl chloride, sulfur dioxide (SO2), and hydrogen chloride (HCl).
Think about the physical properties of SO2 and HCl (both are gases) and how this affects the purity of the alkyl chloride.
Compare this method to other methods for preparing alkyl chlorides from alcohols, such as using HCl or PCl5.
Reflect on why the escape of gases leads to a pure product.
Try solving on your own before revealing the answer!

Q3. Why is o-nitrophenol more steam volatile than p-nitrophenol?
Background
Topic: Physical Properties of Phenols
This question tests your understanding of intermolecular and intramolecular hydrogen bonding and their effects on boiling points and volatility.
Key Terms and Formulas
Intramolecular hydrogen bonding
Intermolecular hydrogen bonding
Steam volatility
Step-by-Step Guidance
Identify the structural difference between o-nitrophenol and p-nitrophenol.
Consider how hydrogen bonding occurs in each compound (intra vs. inter).
Think about how intramolecular hydrogen bonding affects the ability of molecules to associate with each other.
Relate this to the boiling point and steam volatility.
Try solving on your own before revealing the answer!

Q4. Why does t-butyl chloride on heating with sodium methoxide give 2-methylpropene instead of t-butyl methyl ether?
Background
Topic: Elimination vs. Substitution Reactions
This question tests your understanding of the competition between nucleophilic substitution and elimination reactions, especially with tertiary alkyl halides.
Key Terms and Formulas
t-butyl chloride (tert-butyl chloride)
Sodium methoxide (NaOCH3)
Elimination (E2) vs. Substitution (SN1/SN2)
Reaction:
Step-by-Step Guidance
Identify the structure of t-butyl chloride and sodium methoxide.
Consider the steric hindrance around the tertiary carbon in t-butyl chloride.
Think about whether nucleophilic substitution or elimination is favored under these conditions.
Analyze why elimination leads to the formation of 2-methylpropene.
Try solving on your own before revealing the answer!

Q5. Why is the boiling point of ethanol higher than methoxymethane?
Background
Topic: Intermolecular Forces and Boiling Points
This question tests your understanding of hydrogen bonding and dipole-dipole interactions in organic molecules.
Key Terms and Formulas
Hydrogen bonding
Dipole-dipole interaction
Ethanol (C2H5OH)
Methoxymethane (CH3OCH3)
Step-by-Step Guidance
Identify the types of intermolecular forces present in ethanol and methoxymethane.
Compare the strength of hydrogen bonding to dipole-dipole interactions.
Relate the strength of these forces to boiling point.
Think about how molecular structure affects the ability to form hydrogen bonds.
Try solving on your own before revealing the answer!
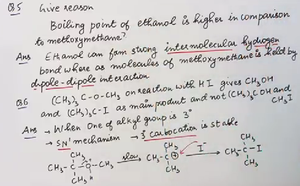
Q6. Why does (CH3)3C-O-CH2CH3 react with HI to give CH3OH and (CH3)3C-I as main products?
Background
Topic: SN1 Mechanism and Carbocation Stability
This question tests your understanding of the SN1 mechanism and the stability of tertiary carbocations.
Key Terms and Formulas
SN1 mechanism
Carbocation stability
Reaction:
Step-by-Step Guidance
Identify the alkyl groups present in the ether.
Consider which group forms a more stable carbocation upon cleavage.
Analyze the mechanism of the reaction with HI (SN1 pathway).
Think about why the tertiary group forms the iodide and the other forms the alcohol.
Try solving on your own before revealing the answer!
Q7. Why are lower alcohols water soluble, whereas higher alcohols are water insoluble?
Background
Topic: Solubility and Hydrogen Bonding
This question tests your understanding of how molecular size and hydrogen bonding affect solubility in water.
Key Terms and Formulas
Hydrogen bonding
Hydrocarbon chain length
Solubility
Step-by-Step Guidance
Consider the ability of alcohols to form hydrogen bonds with water.
Think about how increasing hydrocarbon chain length affects solubility.
Analyze why hydrogen bonding becomes less effective in higher alcohols.
Relate this to the balance between hydrophilic and hydrophobic parts of the molecule.
Try solving on your own before revealing the answer!

Q8. Why does phenol not give protonation reaction readily?
Background
Topic: Resonance and Reactivity of Phenol
This question tests your understanding of resonance effects and the availability of lone pairs for protonation.
Key Terms and Formulas
Resonance
Lone pairs on oxygen
Protonation
Step-by-Step Guidance
Identify the location of lone pairs on the oxygen atom in phenol.
Consider how resonance delocalizes these electrons into the benzene ring.
Analyze why this makes the lone pairs less available for protonation.
Relate this to the reactivity of phenol compared to other alcohols.
Try solving on your own before revealing the answer!

Q9. Out of 2-chloroethanol and ethanol, which is more acidic and why?
Background
Topic: Inductive Effect and Acidity
This question tests your understanding of how substituents affect the acidity of alcohols via the inductive effect.
Key Terms and Formulas
Inductive effect
Acidity
2-chloroethanol (ClCH2CH2OH)
Ethanol (CH3CH2OH)
Step-by-Step Guidance
Identify the effect of the chlorine atom on the electron density of the O-H bond.
Analyze how the inductive effect influences the acidity of the alcohol.
Compare the acidity of 2-chloroethanol and ethanol based on their structures.
Think about why electron-withdrawing groups increase acidity.
Try solving on your own before revealing the answer!

Q10. Why is the C-O-H bond angle in alcohols slightly less than the tetrahedral angle (109.5°)?
Background
Topic: Molecular Geometry and Lone Pair Repulsion
This question tests your understanding of VSEPR theory and how lone pairs affect bond angles.
Key Terms and Formulas
Tetrahedral angle (109.5°)
Lone pair repulsion
Alcohol structure
Step-by-Step Guidance
Identify the electron pairs around the oxygen atom in alcohols.
Consider how lone pairs exert greater repulsion than bonding pairs.
Analyze why this repulsion reduces the bond angle below 109.5°.
Relate this to the observed geometry of alcohols.
Try solving on your own before revealing the answer!

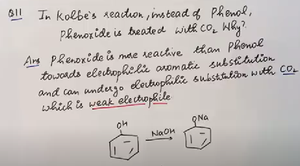
Q11. In Kolbe's reaction, why is phenoxide treated with CO2 instead of phenol?
Background
Topic: Electrophilic Aromatic Substitution
This question tests your understanding of the reactivity of phenoxide ion compared to phenol in Kolbe's reaction.
Key Terms and Formulas
Kolbe's reaction
Phenoxide ion
Electrophilic aromatic substitution
CO2 as a weak electrophile
Step-by-Step Guidance
Identify the difference in reactivity between phenol and phenoxide ion.
Consider why phenoxide ion is more reactive towards electrophiles.
Analyze the role of CO2 as an electrophile in the reaction.
Think about why phenol does not react as efficiently with CO2.
Try solving on your own before revealing the answer!
Q1 (Aldehydes, Ketones, and Carboxylic Acids). Why are aldehydes more reactive than ketones towards nucleophilic reagents?
Background
Topic: Reactivity of Carbonyl Compounds
This question tests your understanding of steric and electronic effects in carbonyl chemistry.
Key Terms and Formulas
Nucleophilic addition
Steric hindrance
Electron releasing groups
General structure: (aldehyde) vs. (ketone)
Step-by-Step Guidance
Compare the number and size of alkyl groups attached to the carbonyl carbon in aldehydes and ketones.
Analyze how steric hindrance affects the approach of nucleophiles.
Consider the electronic effects of alkyl groups on the carbonyl carbon.
Relate these effects to the overall reactivity towards nucleophilic addition.
Try solving on your own before revealing the answer!

