 Back
BackOrganometallic Compounds: Structure, Reactivity, and Coupling Reactions
Study Guide - Smart Notes

Organometallic Compounds
Definition and General Properties
Organometallic compounds are molecules that contain a covalent bond between a carbon atom and a metal. These compounds are central to organic synthesis due to their unique reactivity, which arises from the polarization of the carbon–metal bond. The nature of this bond depends on the relative electronegativities of carbon and the metal involved.
Organometallic bond: A covalent bond between carbon and a metal atom.
Polarity: The bond is polarized such that carbon can act as a nucleophile when bonded to less electronegative metals.
Reactivity: Organometallic compounds are highly reactive with water and other protic solvents due to their basic and nucleophilic character.
Electronegativity and Bond Polarity
The reactivity of organometallic compounds is largely determined by the difference in electronegativity between carbon and the metal. Metals less electronegative than carbon (such as Li, Mg, Cu) impart a partial negative charge to carbon, making it nucleophilic.
Electronegativity: The tendency of an atom to attract electrons in a bond.
Trend: As the electronegativity difference increases, the carbon becomes more nucleophilic and basic.
Types of Organometallic Compounds
Organolithium Compounds
Organolithium compounds contain a carbon–lithium bond. Lithium is monovalent and forms highly reactive organometallics that are strong bases and nucleophiles.
Preparation: Typically synthesized by reacting alkyl or aryl halides with lithium metal in an aprotic solvent such as hexane.
Reactivity: Reacts with a wide range of electrophiles, including carbonyl compounds and epoxides.
Organomagnesium Compounds (Grignard Reagents)
Organomagnesium compounds, commonly known as Grignard reagents, have the general formula RMgX, where R is an alkyl or aryl group and X is a halide. Magnesium is divalent and forms a bond with both the carbon and the halogen.
Preparation: Formed by reacting alkyl or aryl halides with magnesium metal in dry ether or THF.
Reactivity: Strong nucleophiles and bases, react with carbonyl compounds, epoxides, and other electrophiles.
Comparison of Organolithium and Grignard Reagents
Both organolithium and Grignard reagents are strong nucleophiles and bases, but organolithium compounds are generally more reactive due to a larger electronegativity difference between carbon and lithium.
Organolithium (CH3Li): Electronegativity difference = 1.5 (more reactive)
Grignard (CH3MgBr): Electronegativity difference = 1.3 (less reactive)
Reactivity: Both react with acidic protons and electrophiles, but not with alkyl halides.
Reactivity of Organometallic Compounds
As Bases
Organolithium and Grignard reagents are extremely strong bases and will react with any compound containing an acidic hydrogen, such as alcohols, water, thiols, and carboxylic acids. Even trace moisture can destroy these reagents by forming alkanes.
Example: Reaction with water:
As Nucleophiles
These reagents are also strong nucleophiles, attacking electrophilic centers such as carbonyl groups (in aldehydes, ketones, esters) and epoxides to form new carbon–carbon bonds.
Limitation: Do not react with alkyl halides due to competing side reactions.
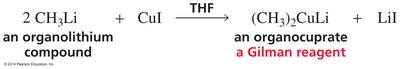
Organocuprates (Gilman Reagents)
Structure and Preparation
Organocuprates, also known as Gilman reagents, have the general formula R2CuLi. They are prepared by reacting organolithium compounds with copper(I) iodide in THF.
Example:
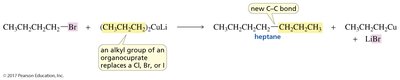
Coupling Reactions
Organocuprates are used in coupling reactions to join two carbon-containing groups, typically replacing a halogen (Cl, Br, I) on an alkyl, vinylic, or aryl halide with an alkyl group from the organocuprate. The R group cannot be secondary or tertiary due to side reactions.
Example:
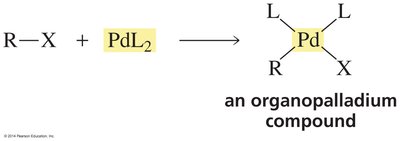
Palladium-Catalyzed Coupling Reactions
General Features
Palladium-catalyzed coupling reactions are powerful methods for forming carbon–carbon bonds, especially between vinylic or aryl halides and organometallic reagents. These reactions proceed via a catalytic cycle involving palladium complexes coordinated with ligands.
Key Reactions: Suzuki and Heck reactions
Substrates: Only vinylic or aryl halides are suitable; alkyl halides with β-hydrogens undergo elimination instead of coupling.
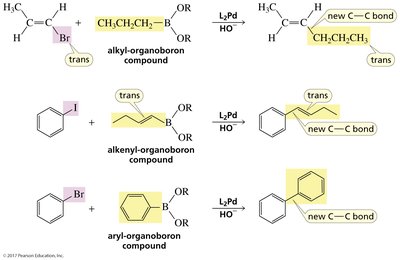
Suzuki Reaction
The Suzuki reaction couples a vinylic or aryl halide with an organoboron compound in the presence of a palladium catalyst and base, forming a new C–C bond. This reaction is widely used to add R groups onto alkenes or aromatic rings.
General equation:
Applications: Synthesis of biaryl compounds, styrenes, and substituted alkenes.

Heck Reaction
The Heck reaction couples a vinylic or aryl halide with an alkene in the presence of a palladium catalyst and a base, forming a substituted alkene. The reaction is regioselective and stereoselective, often favoring trans products.
General equation:
Best alkenes: Symmetrical alkenes, terminal alkenes, or those with resonance stabilization of the carbocation intermediate.
Limitations and Selectivity
Only vinylic or aryl halides are suitable for these coupling reactions because alkyl halides with β-hydrogens undergo β-elimination instead of coupling. The selectivity of the reaction is influenced by the structure of the alkene and the presence of substituents.
Limitation: No β-hydrogens on sp3 carbons for successful coupling.
Regioselectivity: The new C–C bond forms at the less hindered or more electron-deficient position.
Summary Table: Common Organometallic Compounds
Type | General Formula | Preparation | Reactivity | Key Applications |
|---|---|---|---|---|
Organolithium | R–Li | Alkyl/aryl halide + Li | Strong base, nucleophile | Carbonyl addition, Gilman reagent synthesis |
Grignard (Organomagnesium) | R–MgX | Alkyl/aryl halide + Mg | Strong base, nucleophile | Carbonyl addition, epoxide opening |
Organocuprate (Gilman) | R2CuLi | 2 R–Li + CuI | Mild nucleophile | Coupling with alkyl/vinylic/aryl halides |
Organopalladium | R–PdL2 | R–X + PdL2 | Catalyst in coupling | Suzuki, Heck reactions |
Additional info: Organometallic chemistry is foundational for modern organic synthesis, enabling the construction of complex molecules through selective C–C bond formation. Mastery of these reagents and their mechanisms is essential for advanced study and research in organic chemistry.
