 Back
BackOrgo 1 Review: Essential Concepts and Mechanisms
Study Guide - Smart Notes

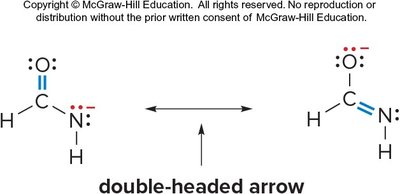
Resonance
Resonance Structures and Stability
Resonance is a fundamental concept in organic chemistry, describing the delocalization of electrons within molecules. Resonance structures are alternative Lewis structures for a molecule, differing only in the placement of electrons (not atoms).
Resonance structures are separated by a double-headed arrow.
Atoms remain in the same positions; only electrons (lone pairs and pi bonds) move.
Electron movement is shown with curved arrows.
Delocalization of electrons increases molecular stability, resulting in a resonance-stabilized molecule.
Major contributors to the resonance hybrid are those with the lowest energy (most stable).
Minor contributors are less stable due to charge separation or incomplete octets.
Resonance is crucial for understanding reactivity, acidity, and basicity in organic molecules.
Example: Resonance in amides, where electron delocalization occurs between the oxygen and nitrogen atoms.
Functional Groups
Overview of Common Functional Groups
Functional groups are specific groups of atoms within molecules that determine the chemical properties and reactivity of those molecules. Recognizing functional groups is essential for predicting reactions and interpreting spectra.
Alkane: C–H, C–C single bonds
Alkene: C=C double bond
Alkyne: C≡C triple bond
Alcohol: –OH group
Aldehyde: –CHO group
Ketone: –CO– group
Carboxylic acid: –COOH group
Amine: –NH2, –NHR, –NR2
Amide: –CONH2, –CONHR, –CONR2
Ester: –COOR
Nitrile: –C≡N
Spectroscopy
Infrared (IR) Spectroscopy
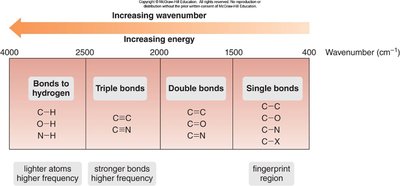
IR spectroscopy is used to identify functional groups in organic molecules by measuring bond stretching and bending vibrations. Each functional group absorbs IR radiation at characteristic wavenumbers.
Absorption occurs due to changes in dipole moment during vibration.
Characteristic absorptions help distinguish between different functional groups.
Intensity and position of peaks provide information about the type and environment of bonds.
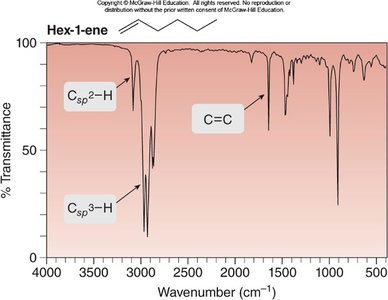
Example: IR spectrum of hex-1-ene shows Csp2–H and C=C stretches.
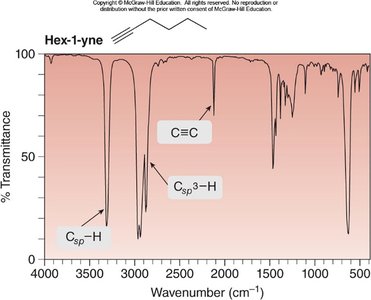
Example: IR spectrum of hex-1-yne shows C≡C and Csp–H stretches.
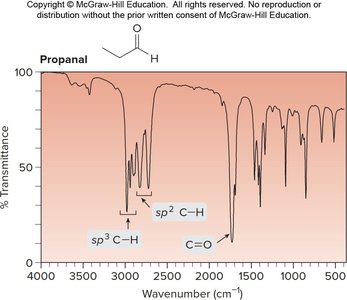
Example: IR spectrum of propanal shows C=O and C–H stretches.
Example: IR spectrum of butan-2-one shows a strong C=O stretch.
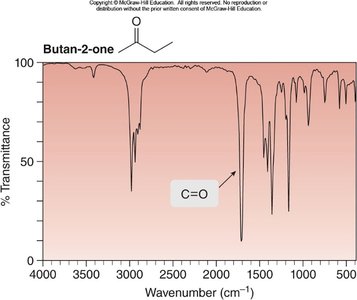
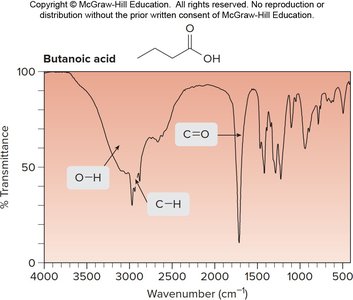
Example: IR spectrum of butanoic acid shows O–H and C=O stretches.
Example: IR spectrum of propanamide shows N–H and C=O stretches.
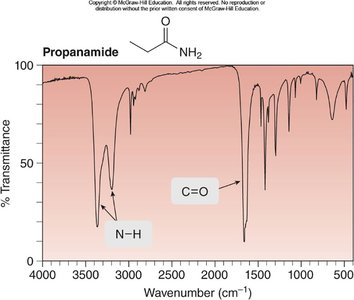
Table: Characteristic IR absorptions for functional groups.
Compound type | Absorption (cm-1) | Intensity |
|---|---|---|
Alkane C–H | 3000–2850 | strong |
Alkene C=C | 1650 | medium |
Alkyne C≡C | 2250 | medium |
Alcohol O–H | 3600–3200 | strong, broad |
Aldehyde C=O | 1725 | strong |
Ketone C=O | 1715 | strong |
Carboxylic acid C=O | 1710 | strong |
Amide C=O | 1680–1630 | strong |
Nitrile C≡N | 2250 | medium |
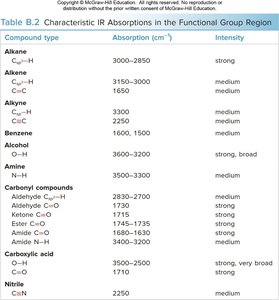
Diagram: IR wavenumber regions for different bond types.
1H NMR Spectroscopy
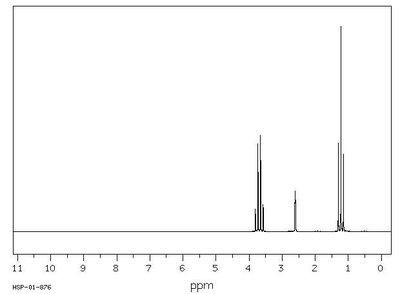
Proton NMR (1H NMR) provides detailed information about the hydrogen atoms in a molecule, including their environment, number, and interactions with neighboring protons.
Number of signals: Number of unique proton environments.
Integration: Number of protons in each environment.
Chemical shift (ppm): Indicates proximity to electronegative atoms or functional groups.
Splitting (coupling): Shows how many neighboring protons are within three bonds (n+1 rule).
Coupling constants (J values): Quantify the interaction between coupled protons.
Example: NMR spectrum with multiple peaks corresponding to different proton environments.
Nucleophilic Substitution Reactions
Mechanism and Key Concepts
Nucleophilic substitution reactions involve the replacement of a leaving group (LG) by a nucleophile (Nu) on an electrophilic carbon. The nature of the substrate, nucleophile, and leaving group determines the mechanism (SN1 or SN2).
Electrophile: Lewis acid, typically a carbon attached to a leaving group.
Nucleophile: Lewis base, provides electrons to form a new bond.
Leaving group: The group that departs with a pair of electrons; weaker bases are better leaving groups.
SN2: Concerted, one-step mechanism; strong nucleophile, primary substrate preferred; inversion of configuration.
SN1: Two-step mechanism; weak nucleophile, tertiary substrate preferred; racemization and possible carbocation rearrangement.
Example: Substitution of a halide by a nucleophile.

Table: Common nucleophiles in organic chemistry.
Negatively charged nucleophiles | Neutral nucleophiles | |
|---|---|---|
Oxygen | OH-, OR-, CH3CO2- | H2O, ROH |
Nitrogen | N3- | NH3, RNH2 |
Carbon | CN-, HC≡C- | |
Halogen | Cl-, Br-, I- | |
Sulfur | HS-, RS- | H2S, RSH |
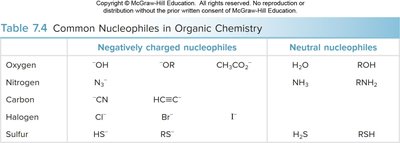
Table: Good leaving groups for nucleophilic substitution.
Starting material | Leaving group | Conjugate acid | pKa |
|---|---|---|---|
R–Cl | Cl- | HCl | -7 |
R–Br | Br- | HBr | -9 |
R–I | I- | HI | -10 |
R–OH2+ | H2O | H3O+ | -1.7 |

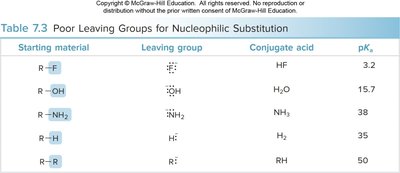
Table: Poor leaving groups for nucleophilic substitution.
Starting material | Leaving group | Conjugate acid | pKa |
|---|---|---|---|
R–F | F- | HF | 3.2 |
R–OH | OH- | H2O | 15.7 |
R–NH2 | NH2- | NH3 | 38 |
R–H | H- | H2 | 35 |
R–R' | R'- | RH | 50 |
SN2 Mechanism: One-step, simultaneous bond formation and breaking.

SN1 Mechanism: Two-step, carbocation intermediate.
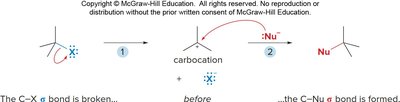
Table: Summary of alkyl halides and SN1, SN2, E1, and E2 mechanisms.
Alkyl halide type | Reaction with | Mechanism |
|---|---|---|
1° RCH2X | Strong nucleophile Strong bulky base | SN2 E2 |
2° R2CHX | Strong base and nucleophile Strong bulky base Weak base and nucleophile | SN2 and E2 E2 SN1 and E1 |
3° R3CX | Weak base and nucleophile Strong base | SN1 and E1 E2 |

Elimination Reactions
Mechanism and Key Concepts
Elimination reactions involve the removal of atoms or groups from a molecule, resulting in the formation of a double or triple bond. The two main mechanisms are E1 and E2.
E1: Two-step mechanism; weak base, tertiary substrate preferred; carbocation intermediate.
E2: One-step mechanism; strong base or bulky base, tertiary substrate preferred; concerted removal of proton and leaving group.
Zaitsev's rule: The more substituted alkene is the major product.
High reaction temperature favors elimination over substitution.
Example: E2 reaction with strong base.
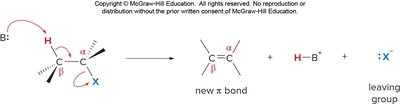
Table: Common bases used in dehydrohalogenation.
Structure | Name |
|---|---|
Na+ OH- | Sodium hydroxide |
K+ OH- | Potassium hydroxide |
Na+ OCH3- | Sodium methoxide |
Na+ OCH2CH3- | Sodium ethoxide |
K+ OC(CH3)3- | Potassium tert-butoxide |

Alcohols: Reactions and Oxidation
Alcohol Reactivity and Transformation
Alcohols are versatile functional groups but require activation for substitution or elimination reactions due to the poor leaving ability of the hydroxyl group.
OH- is a poor leaving group; must be converted to a better leaving group (e.g., protonated, tosylated).
Alcohols can undergo nucleophilic substitution or elimination after activation.
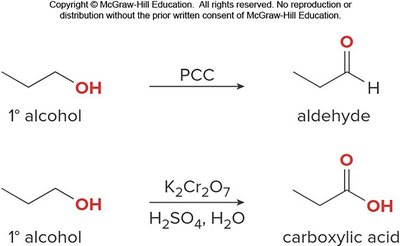
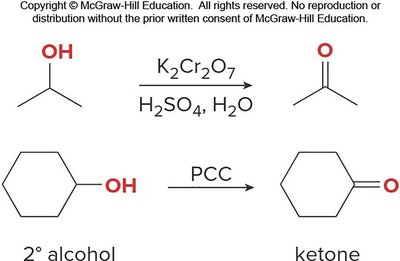
Alcohols can be oxidized to aldehydes, ketones, or carboxylic acids depending on their structure and the oxidant used.
Example: Oxidation of primary alcohols to aldehydes (PCC) or carboxylic acids (K2Cr2O7).
Example: Oxidation of secondary alcohols to ketones.
Example: Tertiary alcohols do not undergo oxidation.
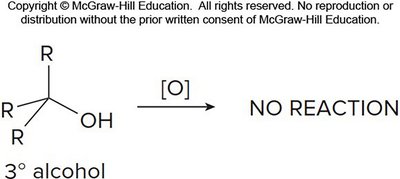
Alkenes and Alkynes: Addition and Oxidation
Electrophilic Addition to Alkenes
Alkenes and alkynes are electron-rich and undergo addition reactions with electrophiles. The regioselectivity of addition is governed by Markovnikov's and anti-Markovnikov's rules.
Markovnikov's rule: Hydrogen adds to the less substituted carbon, forming the more stable carbocation intermediate.
Anti-Markovnikov addition: Hydrogen adds to the more substituted carbon, often via radical or hydroboration mechanisms.
Alkenes can be converted to alcohols, halides, or other functional groups via addition reactions.
Example: Addition of HX, H2O/H2SO4, or BH3/H2O2 to cyclohexene.
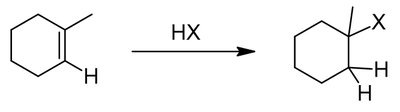
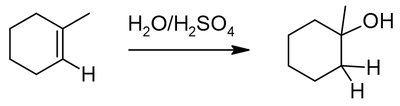
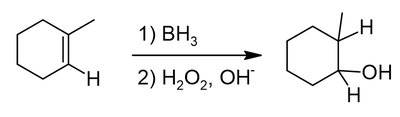
Tautomerism in Alkynes
Alkynes can undergo addition reactions leading to tautomers, which are constitutional isomers differing in the position of a double bond and a hydrogen atom. Keto forms are usually favored over enol forms.
Tautomerization is catalyzed by acid or base.
Terminal alkynes can be deprotonated to form acetylide ions, which are strong nucleophiles.
Example: Markovnikov and anti-Markovnikov hydration of alkynes.
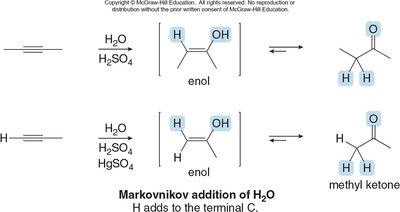
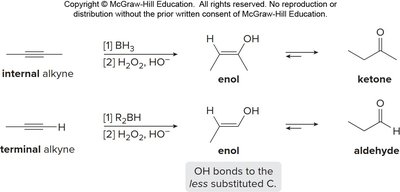
Oxidation of Alkenes and Alkynes
Oxidative cleavage breaks both sigma and pi bonds, forming carbonyl compounds. Alkenes yield aldehydes and ketones, while alkynes yield carboxylic acids and CO2 (for terminal alkynes).
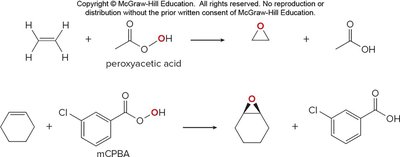
Epoxidation of alkenes is achieved with mCPBA or peroxyacids.
Ozonolysis cleaves double or triple bonds, producing carbonyl compounds.
Example: Ozonolysis of alkenes and alkynes.
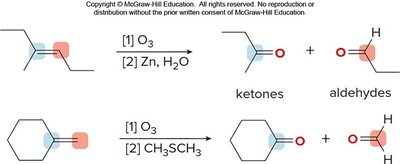
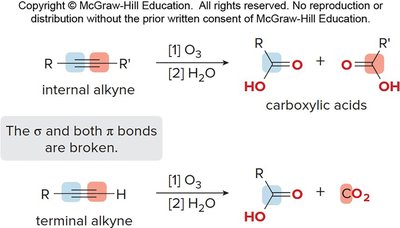
 Additional info: These notes cover essential topics from Orgo 1, including resonance, functional groups, spectroscopy, nucleophilic substitution, elimination, alcohol chemistry, and reactions of alkenes and alkynes. All tables and images included are directly relevant to the explanations and reinforce key concepts.
Additional info: These notes cover essential topics from Orgo 1, including resonance, functional groups, spectroscopy, nucleophilic substitution, elimination, alcohol chemistry, and reactions of alkenes and alkynes. All tables and images included are directly relevant to the explanations and reinforce key concepts.
