 Back
BackPericyclic Reactions: Mechanisms, Stereochemistry, and Applications
Study Guide - Smart Notes

Pericyclic Reactions
Overview and Classification
Pericyclic reactions are a class of organic reactions that proceed via concerted mechanisms, involving cyclic transition states and the redistribution of bonding electrons. These reactions are governed by the Woodward-Hoffmann rules, which predict the stereochemistry and feasibility of pericyclic processes based on orbital symmetry.
Electrocyclic reactions: Ring opening or closing involving π and σ bonds.
Cycloadditions: Two or more π systems combine to form a ring.
Sigmatropic rearrangements: Migration of a σ bond adjacent to one or more π systems.
‘Ene’ reactions: Transfer of an allylic hydrogen with simultaneous bond formation.
Example: The biosynthesis of endiandric acid B by Nicolaou demonstrates the prevalence of pericyclic reactions in both synthetic and natural processes. 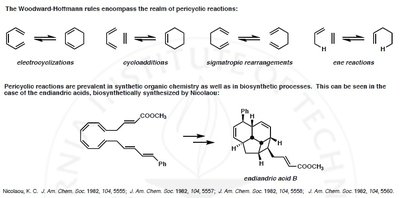
Woodward-Hoffmann Rules
The Woodward-Hoffmann rules use molecular orbital symmetry to predict whether a pericyclic reaction is allowed under thermal or photochemical conditions.
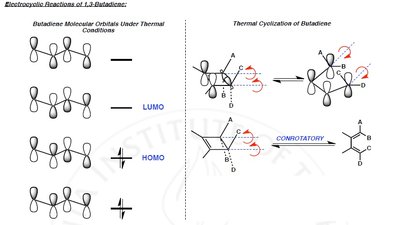
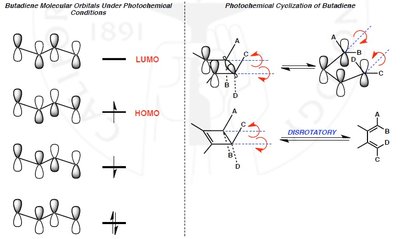
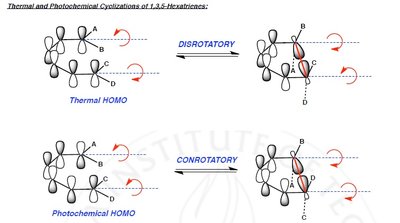
Thermal reactions: Allowed for systems with electrons (aromatic transition state), favoring suprafacial or disrotatory mechanisms.
Photochemical reactions: Allowed for systems with electrons (antiaromatic transition state), favoring antarafacial or conrotatory mechanisms.
Table:
Reaction Type | Transition State Class | Configurational Preference |
|---|---|---|
Thermal (4n+2) | Aromatic | Suprafacial or Disrotatory |
Thermal (4n) | Antiaromatic | Antarafacial or Conrotatory |
Photochemical (4n+2) | Aromatic | Antarafacial or Conrotatory |
Photochemical (4n) | Antiaromatic | Suprafacial or Disrotatory |
Cycloadditions
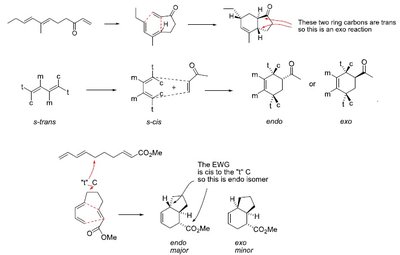
Diels-Alder Reaction: Mechanism and Stereochemistry
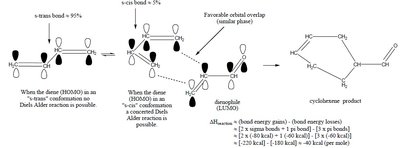
The Diels-Alder reaction is a [4+2] cycloaddition between a conjugated diene and a dienophile, forming a six-membered ring. The reaction is stereospecific, preserving the configuration of substituents.
Diene: Electron-donating groups (EDG) enhance reactivity.
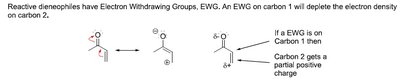
Dienophile: Electron-withdrawing groups (EWG) increase reactivity.
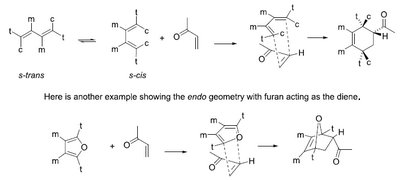
Stereochemistry: The s-cis conformation of the diene is required for effective orbital overlap.
Example: Stereochemistry must match at the reacting carbons; s-trans and s-cis conformations influence product formation. 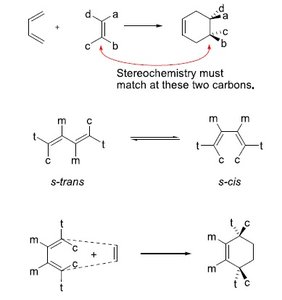
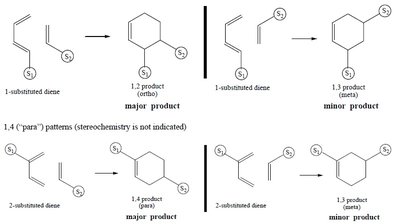
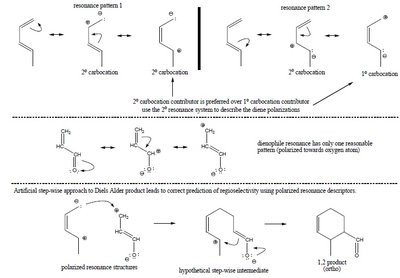
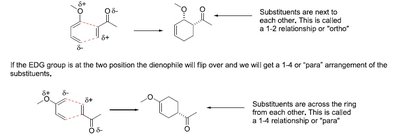
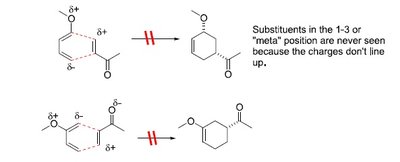
Regioselectivity in Cycloadditions
Regioselectivity is determined by the position of substituents on the diene and dienophile.
1,2 (ortho) and 1,4 (para) products: Major products arise from favorable charge alignment.
1,3 (meta) products: Minor or forbidden due to unfavorable charge distribution.
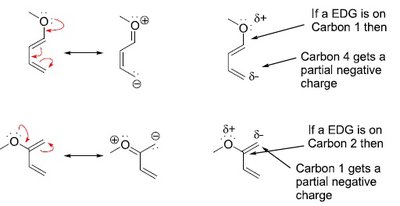
Resonance and Polarization Effects
Resonance patterns in the diene and dienophile influence regioselectivity. Polarized resonance structures help predict the major product.
Electron-donating groups (EDG) on the diene create partial negative charges at specific carbons.
Electron-withdrawing groups (EWG) on the dienophile create partial positive charges.
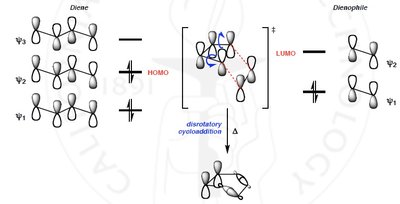
Orbital Interactions and Transition States
The reaction proceeds via the interaction of the diene's HOMO and the dienophile's LUMO. Favorable orbital overlap is essential for product formation.
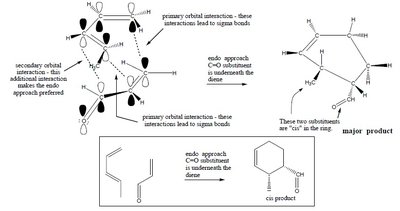
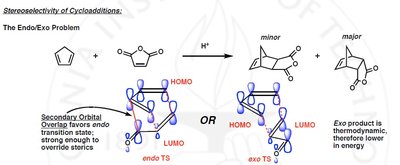
Endo approach: Secondary orbital interactions favor the endo product.
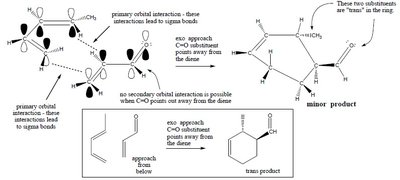
Exo approach: Thermodynamically favored but less common due to weaker secondary interactions.
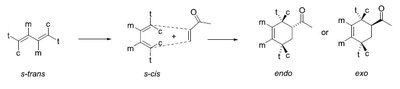
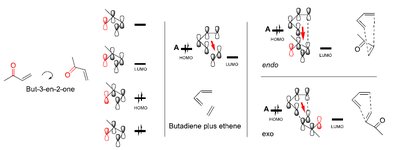
Stereochemistry: Endo vs. Exo Products
The endo rule states that the endo product is favored due to secondary orbital interactions, even if the exo product is thermodynamically more stable.
Endo product: Substituents are oriented under the diene, maximizing orbital overlap.
Exo product: Substituents are oriented away from the diene.
Configurational Outcomes
The configuration of the starting materials (cis or trans) is preserved in the product.
Cis dienophile: Leads to cis product.
Trans dienophile: Leads to trans product.
Thermal vs. Photochemical Cycloadditions
Thermal [2+2] cycloadditions: Forbidden due to orbital symmetry.
Photochemical [2+2] cycloadditions: Allowed, leading to cyclobutane formation.
![Thermal and photochemical [2+2] cycloadditions](https://static.studychannel.pearsonprd.tech/study_guide_files/organic-chemistry/sub_images/2cdca205_image_24.png)
Electrocyclic Reactions
Mechanism and Stereochemistry
Electrocyclic reactions involve the ring opening or closing of conjugated π systems. The stereochemistry is determined by the number of π electrons and whether the reaction is thermal or photochemical.
Disrotatory: Ends of the π system rotate in opposite directions.
Conrotatory: Ends of the π system rotate in the same direction.
Applications in Organic Synthesis
Complex Natural Product Synthesis
Pericyclic reactions are used in the synthesis of complex molecules, such as dihydrocostunolide, as demonstrated by E.J. Corey.
Multiple pericyclic steps can be combined to construct intricate ring systems.

Summary Table: Pericyclic Reaction Types
Reaction Type | Key Features | Example |
|---|---|---|
Electrocyclic | Ring opening/closing, stereochemistry depends on electron count | Thermal ring opening of cyclobutene |
Cycloaddition | Two π systems combine, stereospecific | Diels-Alder reaction |
Sigmatropic | σ bond migration, rearrangement | Cope and Claisen rearrangements |
Ene | Allylic hydrogen transfer | Ene reaction with alkenes |
Key Equations
Electron count for allowed reactions: (aromatic, allowed thermally), (antiaromatic, allowed photochemically)
General Diels-Alder reaction:
Conclusion
Pericyclic reactions are fundamental in organic chemistry, providing powerful tools for constructing complex molecular architectures with precise stereochemical control. Understanding their mechanisms, stereochemistry, and applications is essential for advanced study and practical synthesis. Additional info: The notes expand on the original slides by providing definitions, examples, and tables for clarity and completeness.
