 Back
BackPolar Covalent Bonds, Acids, and Bases: Foundations of Organic Chemistry
Study Guide - Smart Notes

Chapter 2: Polar Covalent Bonds; Acids and Bases
Polar Covalent Bonds: Electronegativity
Organic molecules often contain covalent bonds with varying degrees of polarity, determined by the electronegativity (EN) of the atoms involved. Electronegativity is the intrinsic ability of an atom to attract shared electrons in a covalent bond. Differences in EN between atoms lead to bond polarity, which influences molecular properties and reactivity.
Covalent bonds have equal electron sharing; polar covalent bonds have unequal sharing; ionic bonds involve complete electron transfer.
Fluorine (F) is the most electronegative element (EN = 4.0), while cesium (Cs) is the least (EN = 0.7).
Metals (left of the periodic table) attract electrons weakly; halogens and reactive nonmetals (right) attract electrons strongly.
Bond polarity increases with greater EN difference between bonded atoms.

Bond Polarity and Inductive Effect
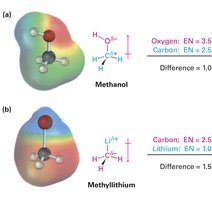
Bond polarity arises when the difference in EN between two atoms is less than 2 (polar covalent) or greater than 2 (ionic). The inductive effect describes the shifting of electrons in a sigma (σ) bond in response to the EN of nearby atoms, resulting in partial charges (δ+ and δ−).
C–H bonds are relatively nonpolar; C–O and C–X bonds are polar.
Electrons are drawn toward the more electronegative atom, giving it a partial negative charge (δ−) and the other atom a partial positive charge (δ+).
Dipole Moments
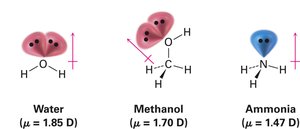
The dipole moment (μ) quantifies the net molecular polarity, resulting from the vector sum of individual bond polarities and lone-pair contributions. Strongly polar substances dissolve in polar solvents like water, while nonpolar substances do not.
Dipole moment formula: (in debyes, D)
1 D = coulomb meter
Examples: Water (μ = 1.85 D), Methanol (μ = 1.70 D), Ammonia (μ = 1.47 D)
Compound | Dipole moment (D) |
|---|---|
NaCl | 9.00 |
CH2O | 2.33 |
CH3Cl | 1.87 |
H2O | 1.85 |
CH3OH | 1.70 |
CH3CO2H | 1.70 |
CH3SH | 1.52 |
NH3 | 1.47 |
CH3NH2 | 1.31 |
CO2, CH4, CH3CH3, Benzene | 0 |

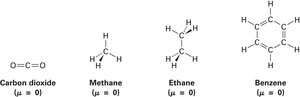
Absence of Dipole Moments
Symmetrical molecules have dipole moments that cancel out, resulting in no net dipole moment. Examples include carbon dioxide, methane, ethane, and benzene.
Formal Charges
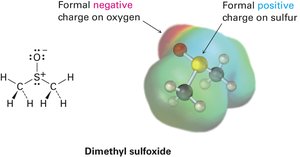
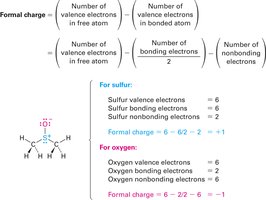
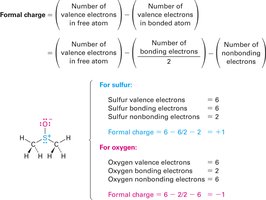
Formal charge is used to keep track of electron distribution in molecules, especially when resonance or unusual bonding occurs. It is calculated by comparing the number of valence electrons in the free atom to those assigned in the molecule.
Formula:
Example: In dimethyl sulfoxide, sulfur has a formal positive charge, and oxygen has a formal negative charge.
Atom | Valence electrons | Number of bonds | Number of nonbonding electrons | Formal charge |
|---|---|---|---|---|
C | 4 | 4 | 0 | 0 |
N | 5 | 3 | 2 | 0 |
O | 6 | 2 | 4 | 0 |
S | 6 | 2 | 4 | 0 |
P | 5 | 5 | 0 | 0 |

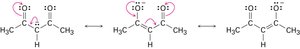
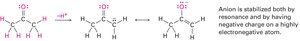
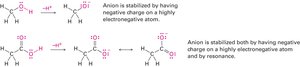
Resonance
Some molecules cannot be represented by a single Lewis structure. Instead, they are described by resonance forms, which differ only in the placement of π or nonbonding electrons. The true structure is a resonance hybrid, which is more stable than any individual form.
Resonance forms are imaginary; the real structure is a hybrid.
Curved arrows indicate electron movement, not atom movement.
Resonance increases molecular stability.
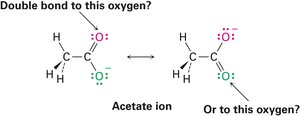
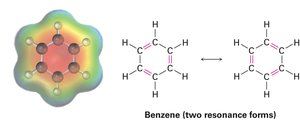
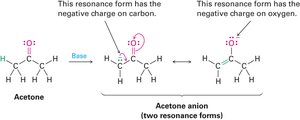
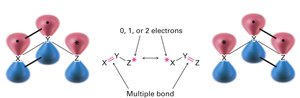
Drawing Resonance Forms
Any three-atom grouping with a p orbital on each atom can have two resonance forms. Recognizing these groupings within larger structures helps generate resonance forms symmetrically.
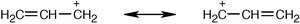
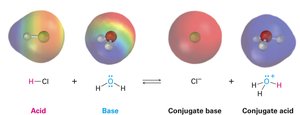
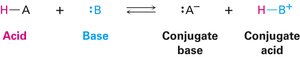
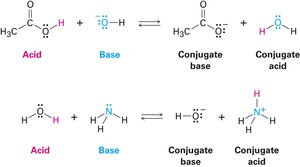
Acids and Bases: The Brønsted–Lowry Definition
The Brønsted–Lowry theory defines acids as proton (H+) donors and bases as proton acceptors. This definition is more useful in organic chemistry than the traditional view of acids and bases as sources of H+ and OH− ions in solution.
Conjugate base: Formed by deprotonation of an acid.
Conjugate acid: Formed by protonation of a base.
Acid and Base Strength
Acid strength is measured by the acidity constant (Ka) and commonly expressed as pKa, the negative logarithm of Ka. Stronger acids have smaller pKa values, while weaker acids have larger pKa values.
Formula:
Water acts as both an acid and a base.
Ion product of water:
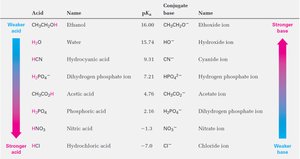
Predicting Acid–Base Reactions from pKa Values
pKa values can be used to predict the direction and extent of acid–base reactions. The difference in pKa values relates to the equilibrium constant for proton transfer.
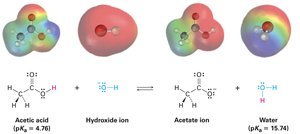
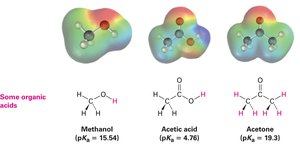
Organic Acids and Bases
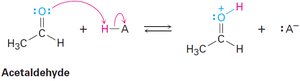
Organic acids are characterized by the presence of a positively polarized hydrogen atom, typically bonded to an electronegative oxygen atom (O–H) or to a carbon atom adjacent to a carbonyl group (O=C–C–H). Organic bases contain atoms with lone pairs that can bond to H+, most commonly nitrogen or oxygen.
Nitrogen-containing compounds derived from ammonia are common organic bases.
Oxygen-containing compounds can act as acids or bases depending on the reaction conditions.
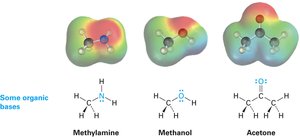
Acids and Bases: The Lewis Definition
The Lewis definition broadens the concept of acids and bases: a Lewis acid is an electron pair acceptor, and a Lewis base is an electron pair donor. This definition includes metal cations and compounds with vacant orbitals as Lewis acids.
Group 3A elements (e.g., BF3, AlCl3) and transition-metal compounds (e.g., TiCl4, FeCl3) are Lewis acids.
Curved arrows indicate electron pair movement from base to acid.
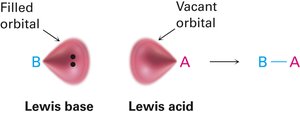
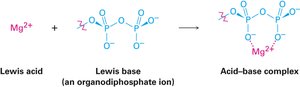
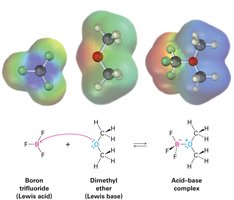
Lewis Bases
Lewis bases are compounds with nonbonding electron pairs available for bonding to Lewis acids. Many organic compounds containing oxygen or nitrogen are Lewis bases and can also act as Brønsted bases.
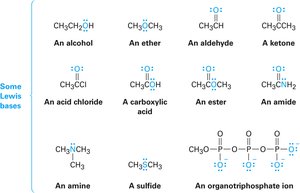
Noncovalent Interactions Between Molecules
Noncovalent interactions are essential for molecular recognition and stability in organic chemistry. The main types are dipole–dipole forces, dispersion forces, and hydrogen bonds.
Dipole–dipole forces: Electrostatic interactions between polar molecules.
Dispersion forces: Present in all molecules due to temporary changes in electron distribution.
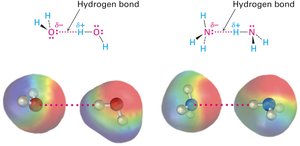
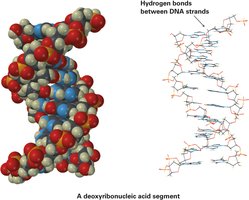
Hydrogen bonds: Attractive interactions between a hydrogen atom bonded to O or N and an unshared electron pair on another O or N atom.

Hydrophilic and Hydrophobic Molecules
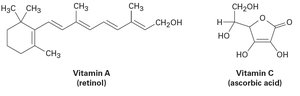
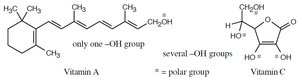
Solubility in water depends on the ability of molecules to form hydrogen bonds. Hydrophilic molecules (e.g., vitamin C) have polar groups that interact with water, while hydrophobic molecules (e.g., vitamin A) lack such groups and are fat-soluble.
Summary
Organic molecules often have polar covalent bonds due to differences in electronegativity.
Molecular polarity is measured by dipole moment (μ).
Formal charges help track electron distribution in molecules.
Resonance hybrids represent delocalized electron structures, increasing stability.
Brønsted–Lowry acids donate protons; bases accept protons. Strength is related to pKa.
Lewis acids accept electron pairs; Lewis bases donate electron pairs.
Noncovalent interactions include dipole–dipole, dispersion, and hydrogen bond forces.
