 Back
BackPolymer Chemistry: Chain Length, Molar Mass, and Characterization Techniques
Study Guide - Smart Notes

From Monomers to Polymers: Influence of Chain Length
Effect of Chain Length on Material Properties
The chain length of polymers, often expressed as the degree of polymerization (DPn), significantly influences their physical properties. As the molar mass increases, polymers transition from waxy or oily substances to tough, high-strength materials.
Low molar mass (oligomers): Used as lubricants or waxes, with limited mechanical strength.
Medium molar mass: Used in films and packaging, offering flexibility and moderate strength.
High molar mass: Used in specialized applications, such as medical implants, due to their exceptional mechanical properties.
Ultra-high molar mass: Used in demanding engineering applications.



Mechanical Strength and Glass Transition Temperature (Tg)
Mechanical properties such as strength and flexibility, as well as the glass transition temperature (Tg), are strongly dependent on chain length. The Flory-Fox equation describes the relationship between Tg and the number-average molar mass (Mn):
Tg,∞: Glass transition temperature at infinite molar mass.
K: Empirical thermodynamic factor.
Mn: Number-average molar mass.
Physical Chain Entanglement
Above a critical chain length, polymer chains can physically entangle, leading to dramatic changes in properties such as mechanical strength, viscosity, and thermal behavior. This entanglement is analogous to cooked spaghetti or macaroni, where chains interlock and resist flow.
Entanglement: Interlocking of polymer chains, enhancing strength and viscosity.


Chain Length Distribution and Average Molar Masses
Polymers as Mixtures of Macromolecules
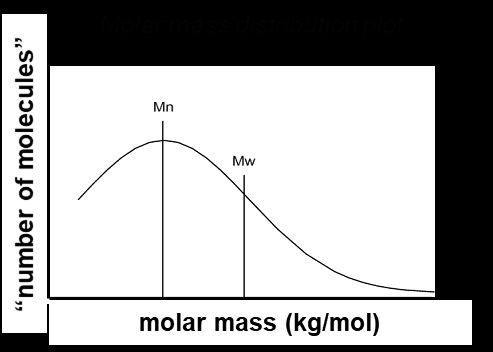
Polymers are typically mixtures of chains with varying lengths. To describe these mixtures, several average molar masses are used:
Number-average molar mass (Mn): Arithmetic mean, sensitive to the number of small chains.
Weight-average molar mass (Mw): Weighted mean, more influenced by larger chains.
Formulas:
Ni: Number of chains with molar mass Mi.
Mi: Molar mass of a chain with degree of polymerization i.
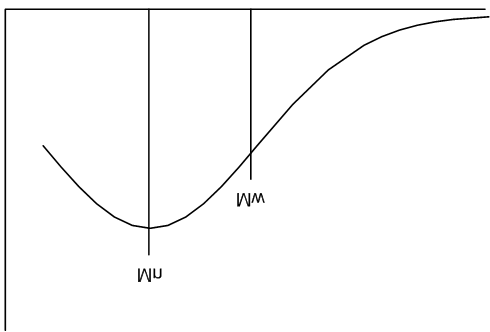
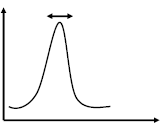
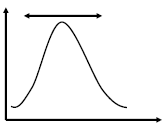
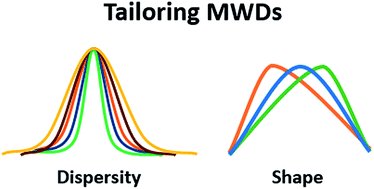
Molar Mass Distribution and Dispersity (Đ)
The molar mass distribution describes how the number of molecules varies with molar mass. Dispersity (Đ) quantifies the breadth of this distribution:
Đ = 1: Monodisperse polymer (all chains same length).
Đ > 1: Polydisperse polymer (mixture of chain lengths).
Effect of Chain Length on Dispersity
Mn is sensitive to the number of small chains, while Mw is dominated by the presence of large chains. Dispersity increases with the presence of more small chains, even if the total mass remains constant.
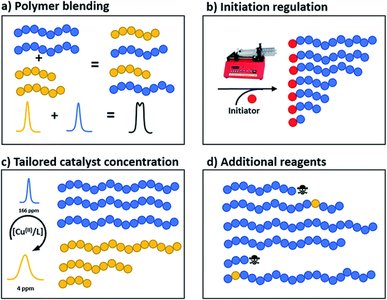
Tailoring Dispersity and Molar Mass Distribution (MMD)
Controlling dispersity and the shape of the MMD is crucial for material properties, processability, and self-assembly. Strategies include polymer blending, initiation regulation, catalyst concentration, and use of additional reagents.
Determination of Polymer Molar Masses
Colligative Properties
Colligative properties such as boiling point elevation, osmotic pressure, and vapor pressure lowering depend on the number of polymer chains in solution and can be used to determine Mn.
Light Scattering
Light scattering techniques (e.g., Rayleigh scattering) provide access to Mw by relating the intensity of scattered light to molecular weight and concentration.
NMR Spectroscopy
NMR can determine the degree of polymerization (DPn) and Mn by integrating signals from end-groups and repeating units.
Viscometry
Viscometry measures the intrinsic viscosity of polymer solutions, which relates to the viscosity-average molar mass (Mv) via the Mark-Houwink equation:
K, α: Mark-Houwink parameters, specific to polymer-solvent system.
Size-Exclusion Chromatography (SEC) / Gel-Permeation Chromatography (GPC)
SEC (or GPC) separates polymer chains based on their hydrodynamic volume, not directly by mass. Larger chains elute first, smaller chains last. This technique provides Mn, Mw, and the full molar mass distribution.




Polymer Characterization Techniques
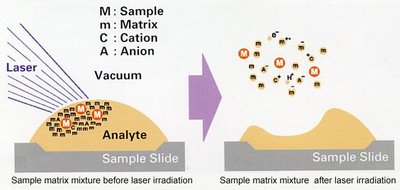
Matrix-Assisted Laser Desorption/Ionisation Time-of-Flight (MALDI-ToF)
MALDI-ToF is a soft ionization mass spectrometry technique that provides absolute molar mass and structural information (e.g., repeating units, end groups) for polymers. It is not a proof of purity and cannot be integrated for quantitative analysis.

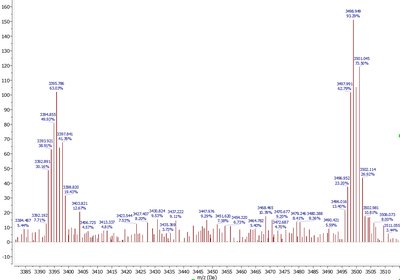
Thermogravimetric Analysis (TGA)
TGA measures the mass of a sample as it is heated, cooled, or held at a fixed temperature under a controlled atmosphere. It provides the decomposition temperature (Td) and information on thermal stability.
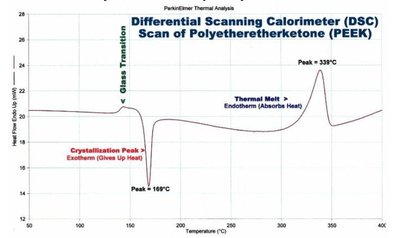
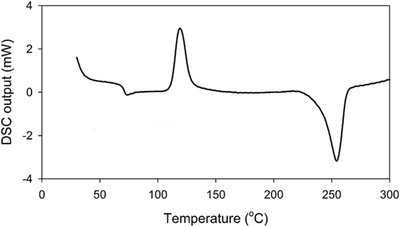
Differential Scanning Calorimetry (DSC)
DSC measures the energy absorbed or released by a sample during heating or cooling. It provides information on glass transition temperature (Tg), melting temperature (Tm), crystallization temperature (Tc), and degree of crystallinity.

Powder X-Ray Diffraction (PXRD)
PXRD is used to determine the crystal structure and degree of crystallinity in polymers.
Polymerization Mechanisms: Step-Growth vs Chain-Growth
Step-Growth Polymerization
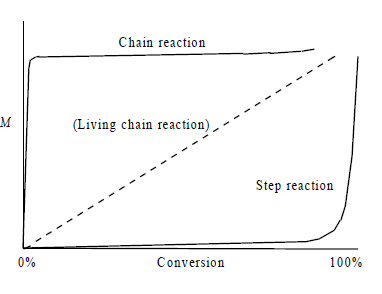
In step-growth polymerization, any two molecular species (monomers, dimers, oligomers) can react. High molecular weights are achieved only at high monomer conversion. Impurities can limit the achievable molecular weight. Typical reactions include polycondensation and polyaddition.
Bi-functional monomers: e.g., diols and di-acids.
Statistical copolymerization: Random incorporation of different monomers.
Chain-Growth Polymerization
In chain-growth polymerization, growth occurs by addition of monomers to active chain ends. Monomer consumption is steady, and molecular weight increases with conversion. Termination reactions can limit molecular weight unless the process is 'living'. Mechanisms include radical, cationic, anionic, and catalyst-mediated reactions.
Unsaturated monomers: Typically contain double bonds.
Block copolymers: Achievable due to end-group fidelity.

Comparison Table: Step-Growth vs Chain-Growth Polymerization
Feature | Step-Growth | Chain-Growth |
|---|---|---|
Reacting Species | Any two species (monomer, oligomer, polymer) | Monomer adds to active chain end |
Monomer Consumption | Rapid, early in reaction | Steady throughout reaction |
Molecular Weight Growth | High MW only at high conversion | MW increases steadily with conversion |
Termination | Not typical | Possible (limits MW) |
Examples | Polyesters, polyamides | Polyethylene, polystyrene |
Additional info: The notes above expand on the original lecture content with definitions, equations, and context for each technique and concept, ensuring a self-contained study guide for polymer chemistry students.
