 Back
BackProtecting Groups in Organic Chemistry: Principles, Applications, and Orthogonality
Study Guide - Smart Notes

Protecting Groups in Organic Chemistry
Introduction to Protecting Groups
Protecting groups (PGs) are essential tools in organic synthesis, allowing chemists to temporarily mask reactive functional groups (FGs) so that selective transformations can be performed on multifunctional molecules. Their use is critical in complex synthetic sequences, such as the total synthesis of natural products, where chemoselectivity and functional group selectivity are required to achieve the desired product.
Functional group selectivity: The ability to selectively react with one functional group in the presence of others.
Chemoselectivity: The preferential reaction of a reagent with one of several different functional groups.
Protection/Deprotection: The process of adding (protection) and removing (deprotection) a PG to enable selective reactions.
Example: In the total synthesis of Taxol, 10 different PGs were used in 7 orthogonal or semi-orthogonal sets, demonstrating the complexity and necessity of PG strategies in advanced organic synthesis.
Characteristics of an Ideal Protecting Group
An ideal protecting group should:
Be introduced and removed under mild conditions with high yield (>99%).
Be inert to a wide range of reaction conditions.
Be small and readily available (good atom economy).
Act as an “innocent observer” – not altering the molecule’s chemistry beyond protection.
Note: There are no perfect PGs; the choice depends on the molecule and the planned reactions.
Common Protecting Groups and Their Applications
Protecting Carboxylic Acids
Carboxylic acids are commonly protected as esters to prevent unwanted reactions such as proton donation, nucleophilic attack, or enolization.
Protection: Conversion to esters (e.g., t-butyl, benzyl, or 4-methoxybenzyl esters) using acid or DCC (dicyclohexylcarbodiimide).
Deprotection: Removal by concentrated acid (e.g., >50% trifluoroacetic acid, TFA) or by reduction (e.g., H2, Pd/C for benzyl esters).
Why protect? To avoid side reactions and enable selective transformations.
How? Remove the acidic proton and introduce steric hindrance.
Protecting Amines
Amines are often protected as carbamates to prevent them from acting as nucleophiles.
Protection: Common PGs include Boc (tert-butyloxycarbonyl), Fmoc (9-fluorenylmethoxycarbonyl), and Cbz (carboxybenzyl). For example, Boc protection uses Boc2O and DMAP (4-dimethylaminopyridine).
Deprotection: Boc is removed with concentrated acid (e.g., 95% TFA or HCl), Fmoc with base (e.g., 20% piperidine), and Cbz with hydrogenation (H2, Pd/C).
Why protect? To prevent nucleophilic activity and enable selective reactions.
How? Introduce steric hindrance or conjugation; decarboxylation is a common strategy.
Protecting Alcohols
Alcohols are protected to prevent them from acting as nucleophiles, being oxidized, or donating protons.
Protection: Conversion to acetyl esters (using Ac2O and base), or other groups such as trityl (Trt) ethers.
Deprotection: Removal by strong base (e.g., NaOEt), acid, or hydrogenation depending on the PG.
Why protect? To prevent unwanted side reactions during synthesis.
How? Introduce steric hindrance or conjugation.
Orthogonality and Semi-Orthogonality of Protecting Groups
Orthogonal Protecting Groups
Orthogonal PGs are sets of protecting groups that can be removed independently under chemically distinct conditions, allowing selective access to different functional groups in a molecule.
Example: Fmoc and PMB are orthogonal PGs for amines and acids, removed by base (20% piperidine) and acid (95% TFA), respectively.
Three orthogonal amine PGs: Boc (removed by acid), Fmoc (removed by base), and Cbz (removed by hydrogenation).
Orthogonality is crucial for complex syntheses, enabling stepwise deprotection and functionalization.
Semi-Orthogonality
Semi-orthogonal PGs are those that can be selectively removed under conditions of varying strength (e.g., dilute vs. concentrated acid). The lability of PGs depends on factors such as carbocation stability, aromaticity, and inductive effects.
Example: Trityl (Trt) ethers can be removed with dilute acid, while t-butyl (tBu) ethers require concentrated acid.
Relative carbocation stability: Many acid-labile PGs exploit carbocation stability to drive deprotection.
Understanding the context and reactivity of each PG is essential for successful synthetic planning.
Summary Table: Common Protecting Groups and Their Removal Conditions
Functional Group | Protecting Group | Protection Conditions | Deprotection Conditions |
|---|---|---|---|
Carboxylic Acid | t-Butyl Ester (tBu) | tBuOH, H+ or DCC | Concentrated acid (e.g., >50% TFA) |
Carboxylic Acid | Benzyl Ester (Bn) | BnOH, DCC | H2, Pd/C |
Amine | Boc | Boc2O, DMAP | Concentrated acid (e.g., 95% TFA) |
Amine | Fmoc | Fmoc-Cl, base | 20% piperidine |
Amine | Cbz | Benzyl chloroformate, base | H2, Pd/C |
Alcohol | Acetyl Ester | Ac2O, base | Strong base or acid |
Alcohol | Trityl (Trt) | Trt-Cl, pyridine | Dilute acid |
Key Takeaways
Protecting groups are vital for enabling selective reactions in complex organic syntheses.
The choice of PG depends on the functional group, the desired transformations, and the required orthogonality.
Understanding the mechanisms and conditions for protection and deprotection is essential for successful synthetic planning.
Orthogonality and semi-orthogonality allow for stepwise and selective deprotection, critical in multi-step syntheses.
Relevant Structure Example: Trityl (Trt) Protecting Group
The trityl group (triphenylmethyl) is a common protecting group for alcohols, removable under mild acidic conditions due to the stability of the resulting trityl carbocation.
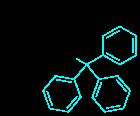
Additional info: The trityl group is particularly useful for protecting alcohols in the presence of other acid-sensitive groups, as it can be selectively removed with dilute acid, demonstrating semi-orthogonality.
