 Back
BackProteins and Amino Acids: Structure, Function, and Biochemistry
Study Guide - Smart Notes

Proteins and Amino Acids
Introduction to Proteins
Proteins are the most abundant organic molecules in living organisms, essential for structure, function, and regulation of tissues and organs. They are polymers of amino acids, each with unique sequences and structures that determine their biological roles.
Catalytic Proteins (Enzymes): Accelerate biochemical reactions, including synthesis and degradation of biomolecules.
Transport Proteins: Carry molecules or ions across membranes or within the body (e.g., hemoglobin transports O2 and CO2).
Regulatory Proteins: Control cellular processes (e.g., insulin regulates glucose metabolism).
Structural Proteins: Provide support and shape (e.g., collagen in connective tissue, keratin in hair).
Contractile Proteins: Enable movement (e.g., actin and myosin in muscles).
Protective Proteins: Defend against pathogens (e.g., antibodies).
Storage Proteins: Store nutrients (e.g., casein in milk, ovalbumin in egg white).
Proteins are classified as fibrous (insoluble, structural) or globular (soluble, functional).
Amino Acids
Structure and Classification
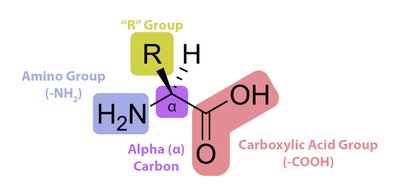
Amino acids are the building blocks of proteins, each containing an amino group, a carboxylic acid group, a hydrogen atom, and a unique side chain (R group) attached to a central alpha (α) carbon. The properties of the R group determine the amino acid's characteristics and its role in proteins.
Nonpolar (hydrophobic): Side chains repel water.
Polar but neutral (hydrophilic): Side chains attract water but are uncharged.
Acidic (hydrophilic): Side chains contain additional carboxyl groups, negatively charged at physiological pH.
Basic (hydrophilic): Side chains contain additional amino groups, positively charged at physiological pH.
All amino acids (except glycine) are chiral and exist in the L-configuration in nature.
Essential and Non-Essential Amino Acids
Mammals require 20 α-amino acids; 10 are essential and must be obtained from the diet because they cannot be synthesized by the body.
Peptides and Peptide Bonds
Formation and Structure
Peptides are formed by the condensation (dehydration) reaction between the amino group of one α-amino acid and the carboxyl group of another, creating a peptide bond (an amide linkage). Peptides are named based on the number of amino acid residues: dipeptides, tripeptides, oligopeptides (<10 residues), and polypeptides (>10 residues).
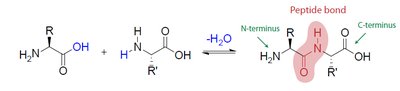
N-terminal: Free amino group at one end (drawn on the left).
C-terminal: Free carboxyl group at the other end (drawn on the right).
Constitutional isomers of peptides arise from different sequences of amino acids. The number of possible isomers for a peptide with n different residues is n! (factorial).
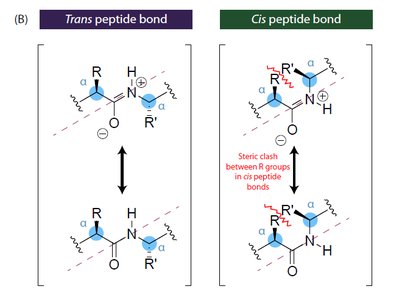
Peptide Bond Properties
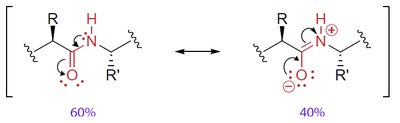
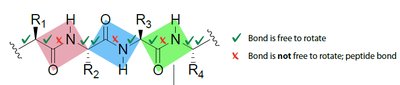
The peptide bond has partial double-bond character due to resonance, restricting rotation and resulting in a planar structure. Most peptide bonds are in the trans configuration to minimize steric clashes between R groups.
Zwitterions and Acid-Base Properties of Amino Acids
Zwitterionic Nature
Amino acids exist as zwitterions at physiological pH, with both a positively charged amino group and a negatively charged carboxyl group. This ionic nature explains their high melting points and solubility in water.
Effect of pH
Acidic conditions (low pH): The carboxylate group is protonated, resulting in a net positive charge.
Basic conditions (high pH): The ammonium group loses a proton, resulting in a net negative charge.
Isoelectric point (pI): The pH at which the amino acid has no net charge (exists as a zwitterion). Each amino acid has a characteristic pI value.
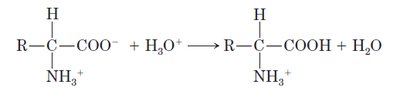
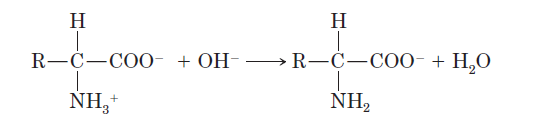
Protein Structure
Primary Structure
The primary structure is the unique sequence of amino acids in a polypeptide chain, determined by genetic information. This sequence dictates the higher levels of protein structure and ultimately its function.
Sequence is always written from the N-terminal to the C-terminal.
Even small changes in sequence can affect protein function, as seen in insulin variants among species.
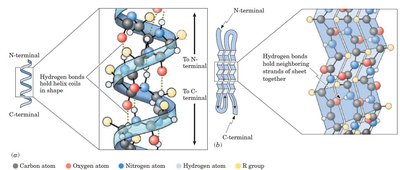
Secondary Structure
Secondary structure refers to regular, repeating patterns in regions of the polypeptide chain, stabilized by hydrogen bonds between backbone atoms. The two main types are:
α-Helix: Right-handed coil stabilized by hydrogen bonds between the C=O of one residue and the N-H of another four residues ahead. Side chains project outward.
β-Pleated Sheet: Extended strands aligned side-by-side, stabilized by hydrogen bonds between strands. Can be parallel or antiparallel.
Random Coil: Regions without regular secondary structure.
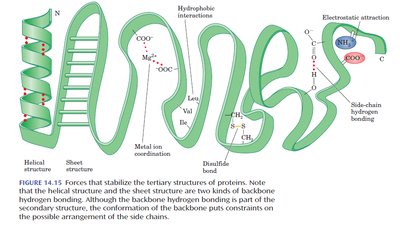
Tertiary Structure
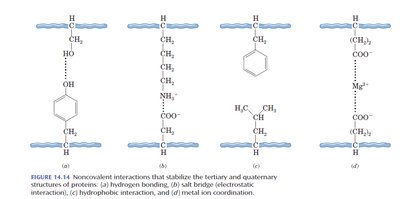
The tertiary structure is the overall three-dimensional shape of a single polypeptide chain, including interactions between side chains (R groups). It is stabilized by several types of interactions:
Covalent Bonds: Disulfide bridges between cysteine residues.
Hydrogen Bonds: Between polar side chains or backbone atoms.
Salt Bridges: Electrostatic attractions between oppositely charged side chains.
Hydrophobic Interactions: Nonpolar side chains cluster away from water.
Metal Ion Coordination: Metal ions stabilize structure by binding to side chains.

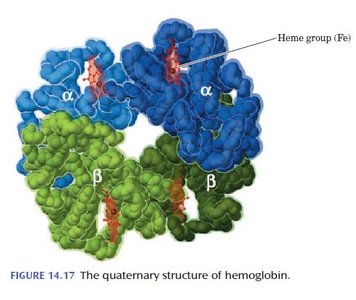
Quaternary Structure
Quaternary structure describes the arrangement and interaction of multiple polypeptide chains (subunits) in a protein. Subunits are held together by the same forces as tertiary structure (hydrogen bonds, salt bridges, hydrophobic interactions, and sometimes covalent bonds).
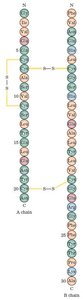
Hemoglobin: Composed of two α and two β chains, each with a heme prosthetic group. Fetal hemoglobin contains γ chains instead of β, giving it higher oxygen affinity.
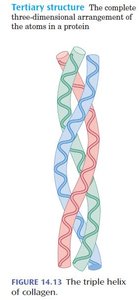
Collagen: Triple helix of polypeptide chains, stabilized by hydrogen bonds and covalent cross-links.
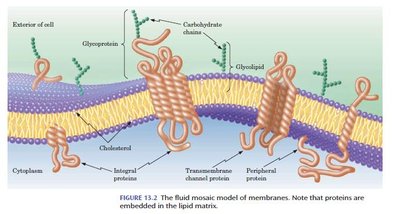
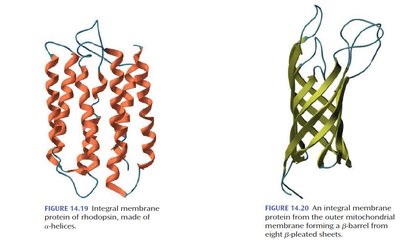
Integral Membrane Proteins: Span the lipid bilayer, often as bundles of α-helices or β-barrels, with nonpolar surfaces interacting with the membrane interior.
Protein Denaturation
Denaturation and Its Effects
Denaturation is the loss of native protein structure due to disruption of secondary, tertiary, or quaternary interactions by physical or chemical agents (e.g., heat, pH changes, detergents). Denatured proteins lose their biological activity and may precipitate or coagulate.
Heat: Breaks hydrogen bonds, disrupting α-helices and β-sheets (e.g., boiling an egg).
Chemicals: Acids, bases, or detergents can disrupt ionic and hydrophobic interactions.
Mechanical agitation: Can also denature proteins (e.g., whipping egg whites).
Denaturation is often irreversible, but in some cases, proteins can refold if the denaturing agent is removed.
