 Back
BackRadical Reactions in Organic Chemistry: Structure, Stability, and Halogenation Mechanisms
Study Guide - Smart Notes

Radical Reactions
Definition and Properties of Radicals
Radicals are highly reactive intermediates in organic chemistry, characterized by a single unpaired electron. They play a crucial role in various organic reactions, especially those involving the formation and breaking of covalent bonds.
Radical: An atom or molecule with a single unpaired electron, denoted by a dot (•).
Octet Deficiency: Radicals lack a complete octet, making them extremely reactive.
No Formal Charge: Radicals are neutral species.
Hydrogen Atom: The hydrogen atom (H•) is technically a radical.
Types of Bond Cleavage
Homolytic vs. Heterolytic Cleavage
Bond cleavage can occur in two distinct ways, each leading to different products and mechanisms.
Homolytic Cleavage: The bond breaks evenly, with each atom taking one electron, resulting in two radicals. This process is depicted using half-headed curved arrows (fishhooks).
Heterolytic Cleavage: Both electrons from the bond are transferred to one atom, generating two charged species (a cation and an anion). Full-headed curved arrows are used to show electron movement.
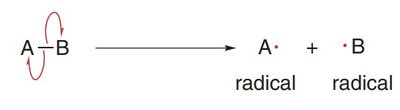
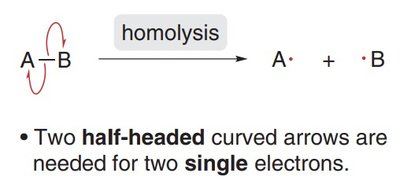
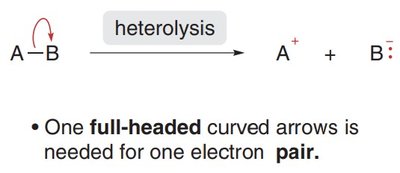
Radical Structure
Hybridization and Geometry
Carbon radicals are typically sp2-hybridized and exhibit trigonal planar geometry. The unpaired electron resides in an unhybridized p orbital.
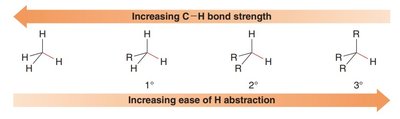
Primary (1°), Secondary (2°), Tertiary (3°): Classification depends on the number of alkyl groups attached to the radical center.
Geometry: Trigonal planar with bond angles of approximately 120°.
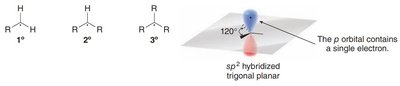
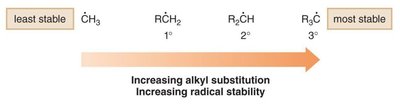
Radical Stability
Effect of Alkyl Substitution
The stability of carbon radicals increases with greater alkyl substitution due to hyperconjugation and inductive effects.
Order of Stability: Tertiary (3°) > Secondary (2°) > Primary (1°) > Methyl (Me).
Rearrangement: 2° and 1° radicals do not undergo rearrangement.
Formation of Radicals
Methods of Radical Generation
Radicals are typically formed by the application of energy, such as light or heat, or by using radical initiators.
Light (hν): Photochemical initiation.
Heat (Δ): Thermal initiation.
Radical Initiators: Peroxides (RO–OR) with weak O–O bonds that homolyze easily.
Radical Reactions
General Reactivity and Mechanisms
Radicals seek to achieve a stable octet and can react in several ways:
React with σ-bonds: Halogenation of alkanes and allylic carbons.
Add to π-bonds: Addition reactions (e.g., HBr addition).
Polymerization: Initiation of chain reactions in polymers.
React with other radicals: Termination steps in radical mechanisms.
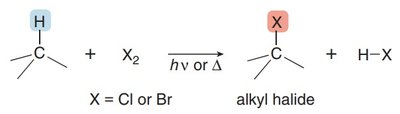
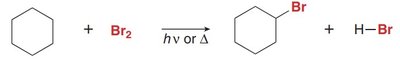
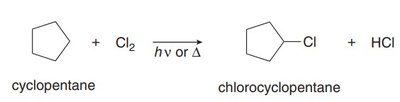
Radical Halogenation of Alkanes
Mechanism and Conditions
Radical halogenation converts alkanes to alkyl halides using chlorine (Cl2) or bromine (Br2) in the presence of light or heat. The process involves a radical substitution mechanism.
Halogen X: Replaces a hydrogen atom via a radical mechanism.
Monohalogenation: Can produce a mixture of alkyl halide products.
Mechanism of Radical Halogenation
Stepwise Process
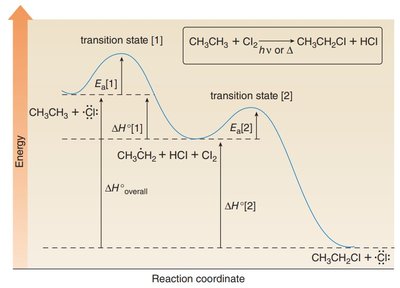
The radical halogenation mechanism consists of three distinct steps:
Initiation: Homolysis of a σ-bond (usually in X2) forms two radicals.
Propagation:
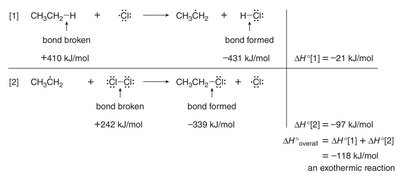
Step 1: Halogen radical abstracts a hydrogen atom, forming HX and a carbon radical.
Step 2: Carbon radical abstracts a halogen atom from X2, forming the alkyl halide and regenerating the halogen radical.
Termination: Two radicals combine to form a stable bond, often leading to unwanted side-products.
Energetics and Rate-Determining Step
Bond Energies and Reaction Profile
The rate-determining step (RDS) in radical halogenation is the abstraction of the hydrogen atom to form the carbon radical. This step is exothermic and depends on the strength of the C–H bond.
Weaker C–H Bonds: More readily undergo radical halogenation.
Energetics: The overall reaction is exothermic, as shown by the sum of enthalpy changes for bond breaking and forming.
Summary Table: Radical Halogenation Steps
Step | Description | Key Features |
|---|---|---|
Initiation | Homolytic cleavage of X2 bond | Forms two radicals |
Propagation | Radical abstracts H, then X | Chain mechanism, forms product |
Termination | Two radicals combine | Forms stable bond, side-products |
Key Equations
Homolytic Cleavage:
Heterolytic Cleavage:
Radical Halogenation:
Example: Radical Halogenation of Ethane
Step 1:
Step 2:
Overall:
Additional info: The notes cover the fundamental aspects of radical chemistry, including structure, stability, and the mechanism of radical halogenation, which are essential for understanding advanced organic reactions and synthetic strategies.
