 Back
BackSolid State Materials: Phases, Microstructure, and Synthesis Methods
Study Guide - Smart Notes

Solid Materials and Microstructure
Phases and Microstructure in Solids
Solid materials are composed of one or more phases, each with a distinct composition and atomic structure. These phases often combine to form a polycrystalline material, which consists of multiple grains—regions with different crystallographic orientations. The arrangement and size of these grains, as well as the presence of different phases, define the material's microstructure. Microstructure plays a critical role in determining the physical properties of materials, such as strength, toughness, and conductivity.
Phase: A region within a material that has uniform physical and chemical properties.
Grain: A single crystal within a polycrystalline material, separated from others by grain boundaries.
Microstructure: The arrangement of phases and grains within a material, observable at the microscopic scale.
Both the phases present and the microstructure can be controlled by manipulating the thermodynamics and kinetics during synthesis and processing.
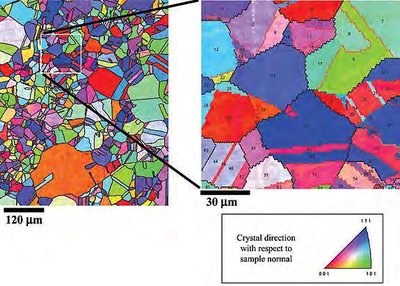
Examples of Microstructure
Different classes of materials exhibit characteristic microstructures:
Ceramics (e.g., Mullite): Often display complex grain structures and multiphase regions.
Metals (e.g., Al-Cu Aerospace Alloy): Exhibit grains and precipitates, with microstructure controlled by composition and processing.
Polymers (e.g., SBS Rubber): Show self-assembled nanostructures encoded during synthesis.

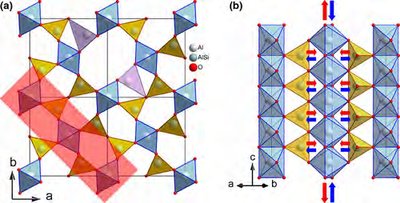
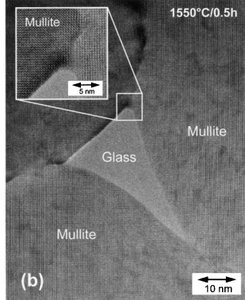
Phase Diagrams
Equilibrium Phase Diagrams
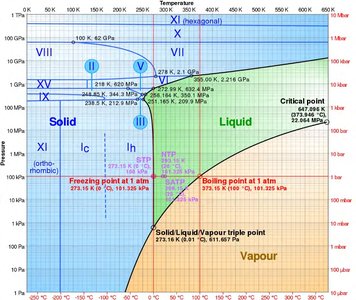
Phase diagrams are graphical representations showing which phases are thermodynamically stable under different conditions (e.g., temperature, pressure, composition). They are essential tools for predicting material behavior and guiding synthesis.
Lines on the diagram indicate boundaries where two or more phases coexist (phase transitions).
Critical point: The end point of a phase equilibrium curve.
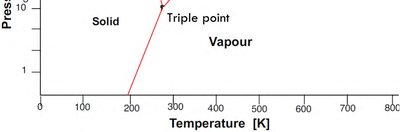
Triple point: The unique set of conditions where three phases coexist in equilibrium.
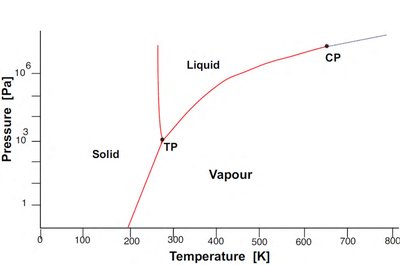
Allotropy and Phase Transitions
Many elements exhibit allotropy, meaning they can exist in different crystal structures depending on temperature and pressure. Each allotrope has distinct physical properties.
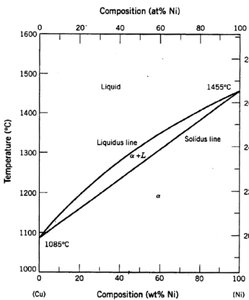
Composition-Temperature Phase Diagrams
These diagrams show the stability regions of phases as a function of both composition and temperature, often for binary systems (two components). They are widely used to understand alloy and ceramic systems.
Miscible systems: Components are completely soluble in each other in all proportions.
Immiscible systems: Components have limited solubility, leading to eutectic or eutectoid reactions.
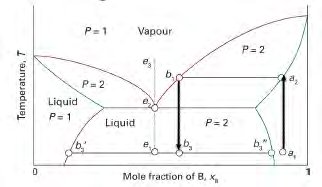
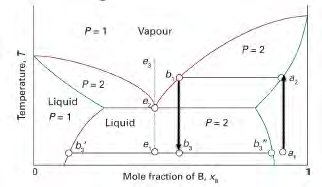
Lever Rule and Phase Proportions
In two-phase regions, the lever rule is used to determine the relative amounts of each phase present at equilibrium. For a system at temperature T and overall composition C:
Fraction of phase α:
Fraction of phase γ:
Where X and Y are the distances on the composition axis from the overall composition to the phase boundaries.
Thermodynamics and Kinetics of Solid-State Reactions
Solid-State Diffusion
Reactions in solids are often slow due to limited atomic mobility. Diffusion in solids occurs via random atomic jumps between lattice sites and is described by Fick's Laws:
Fick's Second Law:
Diffusion coefficient:
Where D is the diffusion coefficient, is the pre-exponential factor, Q is the activation energy, k is Boltzmann's constant, and T is temperature.
Nucleation and Growth
Formation of a new phase requires nucleation, which involves overcoming an energy barrier due to the creation of new interfaces (surface energy). The critical radius and energy barrier for nucleation are given by:
Critical radius:
Critical energy:
Where is the surface energy, v is the atomic volume, and is the Gibbs free energy change per unit volume.
Synthesis of Solids: Methods and Applications
Liquid Phase Syntheses
Melts: Ingredients are melted together and cooled to form the desired phase. Used for metals, glasses, and some ceramics.
Czochralski Crystal Growth: Single crystals are grown from a melt by slowly pulling a seed crystal.
Hydrothermal/Solvothermal Synthesis: Materials are crystallized from high-temperature, high-pressure solutions.
Precipitation: Solids are formed from solution by chemical reaction, often used for purification.
Sol-Gel Synthesis: Involves the transition from a colloidal solution (sol) to a gel, followed by drying and calcination.
Dry Syntheses
Solid State Reactions: Powders are mixed and heated to induce reaction via diffusion.
Mechanosynthesis: Mechanical energy (e.g., ball milling) is used to drive reactions at lower temperatures.
Combustion/Flame Synthesis: Exothermic reactions or flame processes rapidly produce powders.
Vapour Transport/Sealed Tube: Volatile intermediates transport material from one region to another in a sealed tube.
Physical Vapour Deposition (PVD), Chemical Vapour Deposition (CVD): Thin films are deposited from vaporized precursors.
Applications and Examples
Method | Example Material | Application | Key Property |
|---|---|---|---|
Melt | Al alloys | Aircraft | Specific strength |
Czochralski | Silicon | Electronics | Semiconductor |
Solvothermal | Quartz | Watches | Piezoelectric |
Precipitation | TiO2 | Paint | Optical pigment |
Sol-gel | Silica | Chromatography | Surface area |
Solid state reaction | BaTiO3 | Capacitors | Ferroelectric |
Calcination | Cement (CaO) | Construction | Mechanical |
Vapour transport | Nickel | Chemical industry | Corrosion resistance |
Combustion | NiZn ferrite | Transformer | Magnetism |
Flame synthesis | TiO2 | Paint | Optical pigment |
CVD | GaN | LEDs | Semiconductor |
Summary
Understanding the relationship between synthesis, microstructure, and properties is fundamental in materials chemistry. Control over phase formation and microstructure enables the design of materials with tailored properties for a wide range of applications, from structural alloys to electronic devices.
