 Back
BackSolutions: Properties, Types, and Quantitative Aspects
Study Guide - Smart Notes

Solutions and Their Components
Definition and Nature of Solutions
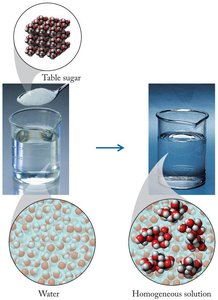
Solutions are homogeneous mixtures composed of two or more substances. The substance present in the greatest amount is called the solvent, while the other components are termed solutes. Solutions can contain one or more solutes dissolved in a solvent, forming a single phase at the molecular level.
Solvent: The component present in the largest amount.
Solute: Any component other than the solvent; present in a lesser amount.
Homogeneous mixture: Uniform composition throughout the sample.
Example: Salt dissolved in water forms a homogeneous solution.
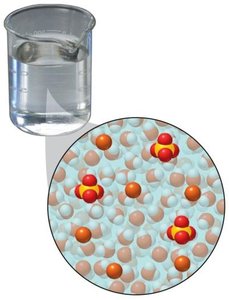
Solubility and Factors Affecting It
Solubility Concepts
Solubility refers to the ability of a solute to dissolve in a solvent. If a solute dissolves, it is termed soluble; if not, it is insoluble. The phrase "like dissolves like" summarizes the principle that polar solvents dissolve polar or ionic solutes, while nonpolar solvents dissolve nonpolar solutes.
Polar solvents (e.g., water) dissolve polar or ionic solutes (e.g., NaCl).
Nonpolar solvents (e.g., hexane) dissolve nonpolar solutes (e.g., oil).
Hydrophilic groups: OH, CHO, C=O, COOH, NH2, Cl (promote solubility in water).
Hydrophobic groups: C-H, C-C (promote solubility in nonpolar solvents).
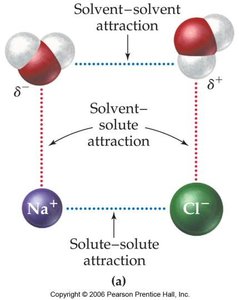
Attractive Forces and Dissolution
The process of dissolution depends on the balance of attractive forces between solute and solvent molecules. For ionic compounds, the solvent molecules (such as water) surround and stabilize the ions, leading to dissolution.
Solvent-solvent attraction: Forces between solvent molecules.
Solute-solute attraction: Forces holding solute particles together.
Solvent-solute attraction: Forces between solvent and solute particles; must be strong enough to overcome solute-solute and solvent-solvent attractions for dissolution to occur.
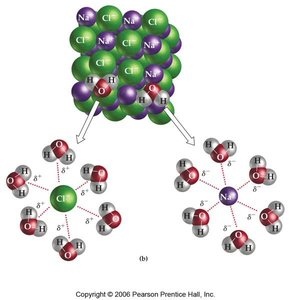
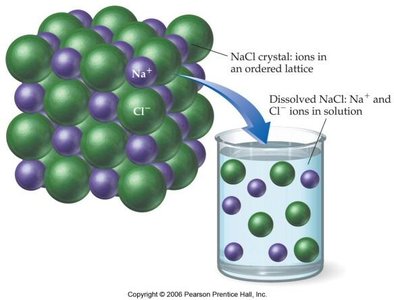
Types of Solutions
Classification by Physical State
Solutions can be classified based on the physical states of their solute and solvent:
Solution Phase | Solute Phase | Solvent Phase | Example |
|---|---|---|---|
Gas | Gas | Gas | Air (N2 & O2) |
Liquid | Gas | Liquid | Soda (CO2 in H2O) |
Liquid | Liquid | Liquid | Vodka (C2H5OH in H2O) |
Liquid | Solid | Liquid | Seawater (NaCl in H2O) |
Solid | Solid | Solid | Brass (Zn in Cu) |
Solubility of Gases in Water
Temperature Dependence
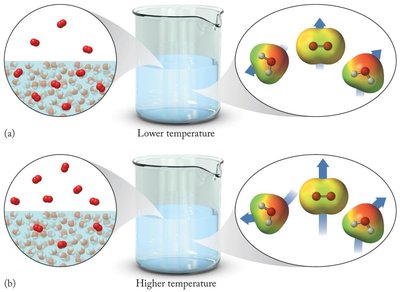
The solubility of gases in water decreases as temperature increases. At higher temperatures, gas molecules have more kinetic energy and are more likely to escape from the solution.
Lower temperature: More gas molecules remain dissolved.
Higher temperature: Fewer gas molecules remain dissolved.
Immiscible Liquids
Definition and Example
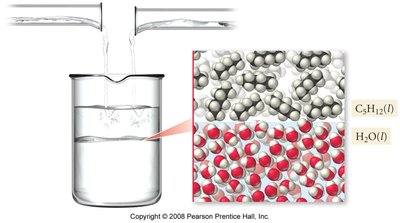
Immiscible liquids are liquids that do not mix to form a homogeneous solution. For example, oil and water are immiscible due to differences in polarity.
Electrolytes and Nonelectrolytes
Definitions and Properties
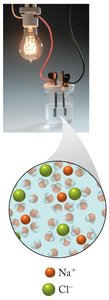
Electrolytes are substances that dissociate into ions when dissolved in water, allowing the solution to conduct electricity. Nonelectrolytes do not produce ions in solution and do not conduct electricity.
Strong electrolytes: Nearly completely dissociate into ions (e.g., NaCl, HCl).
Weak electrolytes: Partially dissociate into ions (e.g., CH3COOH).
Nonelectrolytes: Do not dissociate into ions (e.g., sugar, ethanol).
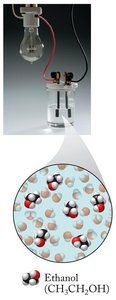
Solubility Rules and Precipitation Reactions
Solubility Rules for Ionic Compounds
Solubility rules help predict whether an ionic compound will dissolve in water. Compounds containing certain ions are generally soluble, while others are insoluble and may form precipitates.
Positive Ions | Negative Ions |
|---|---|
Li+, Na+, K+, Rb+, Cs+, NH4+ | NO3-, C2H3O2- |
Cl-, Br-, I- (except with Ag+, Pb2+, Hg22+) | |
SO42- (except with Ba2+, Pb2+, Ca2+, Sr2+, Hg22+) |

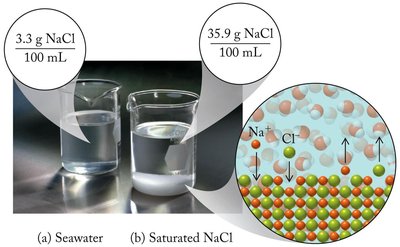
Types of Solutions: Saturated, Unsaturated, and Supersaturated
Definitions
Saturated solution: Contains the maximum concentration of solute possible at a given temperature.
Unsaturated solution: Contains less solute than the maximum amount that can dissolve.
Supersaturated solution: Contains more solute than is predicted to be soluble at a given temperature; unstable and can precipitate excess solute.

Colloids, Suspensions, and Solutions
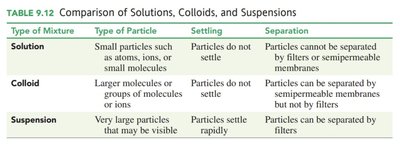
Comparison of Mixtures
Mixtures can be classified as solutions, colloids, or suspensions based on particle size and behavior.
Type of Mixture | Type of Particle | Settling | Separation |
|---|---|---|---|
Solution | Small particles (atoms, ions, small molecules) | Do not settle | Cannot be separated by filters or semipermeable membranes |
Colloid | Larger molecules or groups of molecules/ions | Do not settle | Can be separated by semipermeable membranes, not by filters |
Suspension | Very large particles (may be visible) | Settle rapidly | Can be separated by filters |
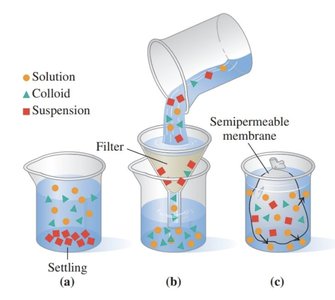
Quantifying Particles in Solution
Concentration Units
Concentration expresses the amount of solute in a given amount of solvent or solution. Common units include molarity, mass percent, and volume percent.
Molarity (M): Moles of solute per liter of solution.
Mass percent (m/m%):
Mass/volume percent (m/v%):
Dilutions
Principle and Calculation
Dilution is the process of decreasing the concentration of a solution by adding more solvent. The number of moles of solute remains constant before and after dilution.
Key equation:
Stock solution: A concentrated solution used to prepare solutions of lower concentration.
Example: To prepare 500.0 mL of 0.145 M HCl from a 12.0 M stock solution, use the dilution equation to solve for the required volume of stock solution.

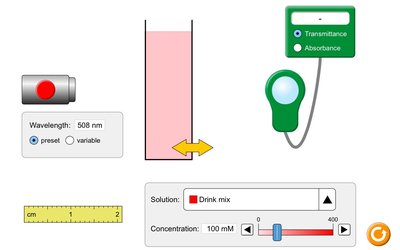
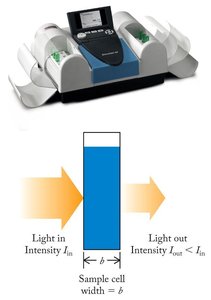
Beer’s Law and Determining Concentration
Beer’s Law
The intensity of color in a solution can be used to measure concentration using Beer’s Law:
Beer’s Law equation:
A: Absorbance (amount of light absorbed by the sample)
\\varepsilon: Molar absorptivity (L mol-1 cm-1)
b: Path length (cm)
c: Concentration of absorbing species (mol/L)
Additional info: These notes cover the fundamental concepts of solutions, solubility, electrolytes, precipitation, colloids, suspensions, concentration units, dilutions, and spectrophotometric analysis, which are essential for understanding solution chemistry in both general and organic chemistry contexts.
