 Back
BackStep-by-Step Guidance for Organic Chemistry Practice Exam Questions
Study Guide - Smart Notes

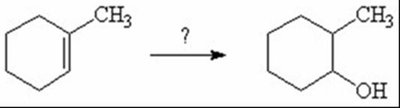
Q1. What reagents are needed to convert methylcyclohexene to methylcyclohexanol?
Background
Topic: Alkene Hydroboration-Oxidation
This question tests your understanding of how to convert an alkene to an alcohol, specifically focusing on the regioselectivity (Markovnikov vs. anti-Markovnikov) and stereochemistry (syn vs. anti addition) of the reaction.
Key Terms and Formulas
Hydroboration-Oxidation: A two-step reaction that adds water across a double bond in an anti-Markovnikov fashion.
Markovnikov Rule: The hydrogen adds to the carbon with more hydrogens, the other group adds to the more substituted carbon.
Anti-Markovnikov: The group (OH) adds to the less substituted carbon.
Syn Addition: Both groups add to the same side of the double bond.
Typical reagents: 1) 2) , ,
Step-by-Step Guidance
Identify the starting material: methylcyclohexene (an alkene with a methyl group attached).
Determine the desired product: methylcyclohexanol (an alcohol at the less substituted carbon adjacent to the methyl group).
Recall that hydroboration-oxidation adds water across the double bond in an anti-Markovnikov manner, placing the OH group on the less substituted carbon.
Write the sequence of reagents: First, for hydroboration, then , , for oxidation.
Try solving on your own before revealing the answer!
Final Answer: E) 1) BH3/THF 2) HO-, H2O2, H2O
This reagent sequence accomplishes anti-Markovnikov hydration of the alkene, resulting in the alcohol at the less substituted carbon.
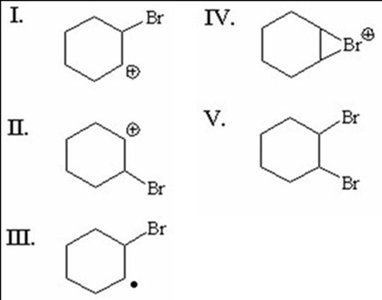
Q2. Which reaction intermediate and product are formed when Br2/CCl4 reacts with cyclohexene?
Background
Topic: Electrophilic Addition to Alkenes (Halogenation)
This question tests your knowledge of the mechanism for bromine addition to alkenes, including the formation of intermediates and the stereochemistry of the product.
Key Terms and Formulas
Bromonium ion: A three-membered ring intermediate formed when Br2 reacts with an alkene.
Anti addition: The two bromine atoms add to opposite sides of the double bond.
Final product: Vicinal dibromide (two bromines on adjacent carbons).
Step-by-Step Guidance
Recognize that Br2 reacts with cyclohexene via electrophilic addition.
The alkene attacks Br2, forming a bromonium ion intermediate (a three-membered ring with Br+).
The second Br- attacks the more substituted carbon, opening the ring and resulting in anti addition.
Identify the intermediate (bromonium ion) and the product (vicinal dibromide).
Try solving on your own before revealing the answer!
Final Answer: D) IV & V
IV is the bromonium ion intermediate, and V is the vicinal dibromide product.
Q8. What reagent is needed to convert 2-butyne to cis-2-butene?
Background
Topic: Alkyne Reduction (Partial Hydrogenation)
This question tests your understanding of how to selectively reduce an alkyne to a cis-alkene using specific catalysts.

Key Terms and Formulas
Lindlar's catalyst: A poisoned palladium catalyst used for partial hydrogenation of alkynes to cis-alkenes.
cis-alkene: Both substituents are on the same side of the double bond.
Partial hydrogenation: Addition of hydrogen to an alkyne, stopping at the alkene stage.
Step-by-Step Guidance
Identify the starting material: 2-butyne (an internal alkyne).
Determine the desired product: cis-2-butene (an alkene with both groups on the same side).
Recall that Lindlar's catalyst is used to selectively hydrogenate alkynes to cis-alkenes.
Write the reagent: with Lindlar's catalyst.
Try solving on your own before revealing the answer!
Final Answer: B) H2/Lindlar's catalyst
Lindlar's catalyst allows for partial hydrogenation, producing the cis-alkene.
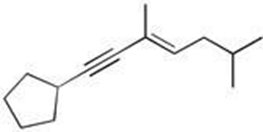
Q17. What is the correct IUPAC name for the given structure?
Background
Topic: IUPAC Nomenclature of Alkenes and Alkynes
This question tests your ability to apply IUPAC rules to name a molecule containing both alkene and alkyne functional groups, as well as substituents.
Key Terms and Formulas
IUPAC nomenclature: Systematic method for naming organic compounds.
Alkene: Double bond, indicated by "-ene" in the name.
Alkyne: Triple bond, indicated by "-yne" in the name.
Substituents: Groups attached to the main chain, named and numbered according to IUPAC rules.
Step-by-Step Guidance
Identify the longest carbon chain containing both the double and triple bonds.
Number the chain to give the lowest possible numbers to the functional groups (alkene and alkyne).
Identify and name the substituents (cyclopentyl, methyl groups).
Determine the E/Z configuration of the double bond if applicable.
Try solving on your own before revealing the answer!
Final Answer: D) (E) 1-cyclopentyl-3,6-dimethylhept-3-en-1-yne
The name reflects the correct numbering, substituents, and E configuration.
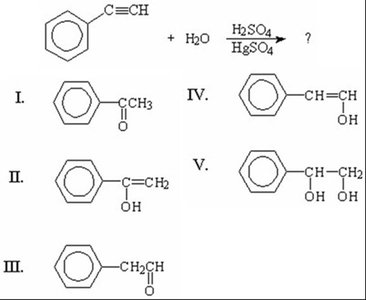
Q20. What is the final and major product of the reaction between phenylacetylene and H2O/H2SO4/HgSO4?
Background
Topic: Alkyne Hydration (Keto-Enol Tautomerism)
This question tests your understanding of the hydration of terminal alkynes, the formation of enol intermediates, and their tautomerization to ketones.
Key Terms and Formulas
Hydration: Addition of water to a triple bond.
Keto-enol tautomerism: The enol intermediate rapidly converts to the more stable ketone.
Terminal alkyne: Alkyne at the end of a carbon chain.
Step-by-Step Guidance
Identify the starting material: phenylacetylene (a terminal alkyne).
Recall that hydration of terminal alkynes with , , and produces a ketone via keto-enol tautomerism.
Draw the enol intermediate and then convert it to the ketone product.
Match the product to the options provided.
Try solving on your own before revealing the answer!
Final Answer: I) Acetophenone
The major product is the ketone, acetophenone, formed after tautomerization of the enol intermediate.
