 Back
BackStep-by-Step Guidance for Organic Chemistry Reactions: Carboxylic Acid Derivatives, Condensations & Alpha Carbon Reactions
Study Guide - Smart Notes

Q1. Predict the product for the following sequence: ethyl cyanide reacts with (1) OH/H2O, (2) acetyl chloride, (3) phthalic acid/AlCl3.
Background
Topic: Carboxylic Acid Derivatives & Friedel-Crafts Acylation
This question tests your understanding of multi-step organic synthesis, including hydrolysis of nitriles, acylation reactions, and aromatic substitution.
Key Terms and Formulas
Nitrile hydrolysis: Conversion of a nitrile to a carboxylic acid using aqueous base.
Acyl chloride: Used for acylation reactions, often in Friedel-Crafts reactions.
Friedel-Crafts acylation: Aromatic substitution using AlCl3 as a catalyst.
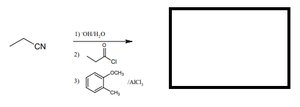
Step-by-Step Guidance
Try solving on your own before revealing the answer!
Q2. Predict the product for the following sequence: cyclopentane-1,3-dione reacts with (1) NaOEt/EtOH, (2) ethyl bromide, (3) H2O/180°C.
Background
Topic: Alpha Carbon Reactions & Alkylation
This question tests your ability to perform enolate formation, alkylation, and subsequent hydrolysis/decarboxylation.
Key Terms and Formulas
Enolate formation: Deprotonation of an alpha hydrogen using a base.
Alkylation: Reaction of an enolate with an alkyl halide.
Decarboxylation: Loss of CO2 from a carboxylic acid under heat.
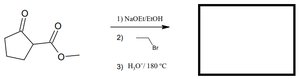
Step-by-Step Guidance
Try solving on your own before revealing the answer!
Q3. Predict the product for the following sequence: benzamide reacts with (1) POCl3, (2) CH3MgBr then H2O, (3) NaOEt/EtOH/Heat.
Background
Topic: Carbonyl Condensations & Alpha Carbon Reactions
This question tests your understanding of amide dehydration, Grignard addition, and enolate chemistry.
Key Terms and Formulas
Amide dehydration: Conversion of an amide to a nitrile using POCl3.
Grignard addition: Nucleophilic addition of a Grignard reagent to a nitrile.
Enolate formation and condensation: Use of base to form enolate and subsequent condensation.
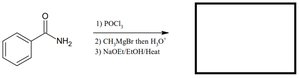
Step-by-Step Guidance
Try solving on your own before revealing the answer!
Q4. Predict the product for the following sequence: glutaric acid reacts with (1) NaBH4/EtOH, (2) H+
Background
Topic: Reduction of Carboxylic Acids
This question tests your understanding of reduction reactions, specifically the use of NaBH4 to reduce carboxylic acids or esters.
Key Terms and Formulas
NaBH4: Sodium borohydride, a reducing agent.
Reduction: Conversion of carboxylic acids to alcohols.

Step-by-Step Guidance
Try solving on your own before revealing the answer!
