 Back
BackStereochemistry and Chirality: Chiral Molecules and Isomerism
Study Guide - Smart Notes

Chapter 5: Stereochemistry
Introduction to Stereochemistry
Stereochemistry is the study of the three-dimensional arrangement of atoms within molecules and how this arrangement affects their chemical properties and behavior. Understanding stereochemistry is essential for identifying, naming, and predicting the properties of organic molecules, especially those with chiral centers.
Isomerism in Organic Molecules
Types of Isomers
Isomers: Compounds with the same molecular formula but different structures.
Constitutional Isomers: Isomers with different connectivity of atoms.
Stereoisomers: Isomers with the same connectivity but different spatial arrangement of atoms.
Enantiomers: Stereoisomers that are nonsuperimposable mirror images of each other.
Diastereomers: Stereoisomers that are not mirror images of each other.
Geometrical isomers (cis/trans) are a type of diastereomer.
Chirality and Stereochemistry
Chirality and Achirality
An object or molecule is achiral if it is identical to its mirror image. In contrast, a chiral object or molecule cannot be superimposed on its mirror image. Chirality is a key concept in stereochemistry, as it leads to the existence of enantiomers.

Example: A glass and its mirror image are superimposable, making the glass achiral.
Chiral Objects and Molecules
A chiral object or molecule cannot be superimposed on its mirror image. This property is fundamental to the existence of enantiomers in organic chemistry.


Example: Human hands are chiral—they are mirror images but cannot be superimposed.
Enantiomers and Chiral Molecules
Enantiomers occur only in compounds with chiral molecules.
A chiral molecule is not superimposable on its mirror image.
The relationship between a chiral molecule and its mirror image is called enantiomeric.
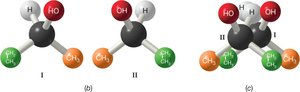
Example: Molecules I and II are nonsuperimposable mirror images (enantiomers).
Chirality Centers
A chirality center (or stereocenter) is a tetrahedral carbon atom bonded to four different groups. A molecule with one chirality center is always chiral and can exist as a pair of enantiomers. If a tetrahedral atom has two or more identical groups, it is not a chirality center, and the molecule is achiral.
Interchanging any two groups at the chirality center converts one enantiomer into the other.
Testing for Chirality: Planes of Symmetry
A molecule is achiral if it possesses a plane of symmetry—an imaginary plane that divides the molecule into two mirror-image halves. All molecules with a plane of symmetry in their most symmetric conformation are achiral.
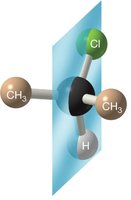
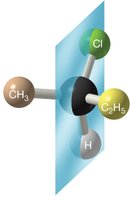
Example: The first molecule has a plane of symmetry and is achiral; the second lacks a plane of symmetry and is chiral.
Naming Enantiomers: The R,S System
Assigning R and S Configurations
The R,S system is used to distinguish between enantiomers by assigning a unique configuration to each stereocenter:
Assign priorities to the four groups attached to the stereocenter based on atomic number (higher atomic number = higher priority).
If two groups have the same atom directly attached, move outward along the chain until a difference is found.
Orient the molecule so the lowest priority group is directed away from you. Trace a path from highest to lowest priority:
Clockwise path: (R) configuration
Counterclockwise path: (S) configuration
For groups with double or triple bonds, treat the bonded atoms as if they are duplicated or triplicated for priority assignment.
Example: (R)-2-Butanol and (S)-2-Butanol are distinguished by the direction of the priority path.
Properties of Enantiomers: Optical Activity
Physical and Chemical Properties
Enantiomers have identical physical properties (melting point, boiling point, refractive index, solubility, etc.).
They have the same chemical properties except in reactions with other chiral substances.
They rotate plane-polarized light in opposite directions (optical activity).
Optical Activity and Polarimetry
Optical activity is the ability of a chiral substance to rotate the plane of polarization of plane-polarized light. This property is measured using a device called a polarimeter.
The observed rotation () depends on:
Concentration of the sample
Length of the cell ()
Wavelength of light (commonly the D-line of Na lamp, nm)
Temperature
The specific rotation is calculated as:
where is the observed rotation, is the concentration in g/mL, and is the path length in dm.
Two enantiomers have equal but opposite specific rotations.
Racemic Mixtures and Enantiomeric Excess
A racemic mixture contains equal amounts of both enantiomers and shows no net optical rotation.
A sample with only one enantiomer is enantiomerically pure (enantiomeric excess = 100%).
Enantiomeric excess (ee) (also called optical purity) can be calculated from optical rotations:
Example: A mixture of 2-butanol enantiomers with a specific rotation of +6.76 (pure enantiomer = +13.52) has an enantiomeric excess of 50%.
Molecules with More Than One Chirality Center
Number of Stereoisomers
For a molecule with tetrahedral stereocenters, the maximum number of stereoisomers is .
Draw all possible stereoisomers by interchanging groups at each chirality center.
Identify pairs of enantiomers and diastereomers.
Diastereomers are stereoisomers that are not enantiomers and usually have different physical and chemical properties.
Meso Compounds
Compounds with two or more stereocenters may have fewer than stereoisomers if some forms are meso compounds—achiral molecules with stereocenters due to an internal plane of symmetry. Meso compounds are optically inactive ().
Example: 2,3-dichlorobutane has two stereocenters but only three stereoisomers (including a meso form).
Biological Significance of Chirality
Chirality is crucial in biological systems. Many drugs and biomolecules are chiral, and often only one enantiomer is biologically active or safe. For example, one enantiomer of a drug may be therapeutic, while the other may cause harmful side effects. The pharmaceutical industry invests heavily in developing single-enantiomer drugs due to their specificity and safety.
66% of drugs in development are chiral; 51% are studied as single enantiomers.
In 2008, $205 billion of $475 billion in pharmaceutical sales were from single-enantiomer drugs.
