 Back
BackStereochemistry and Organic Reaction Mechanisms: Mini-Textbook Study Notes
Study Guide - Smart Notes

Stereochemistry
Absolute and Relative Configuration
Stereochemistry is the study of the three-dimensional arrangement of atoms in molecules and its impact on their properties. The configuration of chiral molecules can be described as either relative or absolute:
Relative configuration: The experimentally determined relationship between the configurations of two molecules, even if the absolute arrangement is unknown.
Absolute configuration: The precise spatial arrangement of atoms in a molecule, often determined by X-ray crystallography.
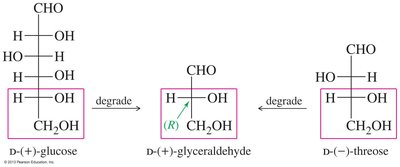
Historically, the D-L system (Fischer-Rosanoff Convention) was used to assign relative configurations to sugars and amino acids based on their similarity to (+)-glyceraldehyde (D) or (–)-glyceraldehyde (L).
With modern techniques, the absolute configuration of glyceraldehyde enantiomers is known: (+) is (R) and (–) is (S).
Physical Properties of Stereoisomers
Stereoisomers are compounds with the same molecular formula and connectivity but different spatial arrangements. They are classified as enantiomers or diastereomers:
Diastereomers: Have different physical properties (melting point, boiling point, solubility) and can be separated by conventional methods such as distillation or recrystallization.
Enantiomers: Differ only in their interaction with other chiral molecules and the direction in which they rotate plane-polarized light. They are difficult to separate.
Resolution of Enantiomers
Pure enantiomers are often isolated from biological sources, but chemical synthesis from achiral reagents typically yields racemic mixtures. The process of separating enantiomers is called resolution:
Reacting a racemic mixture with a chiral compound (resolving agent) forms diastereomers, which can be separated.
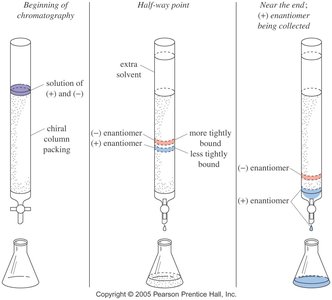
Chromatographic resolution involves passing the racemic mixture through a column coated with chiral molecules, allowing differential binding and separation of enantiomers.
Organic Reaction Mechanisms
Writing Equations for Organic Reactions
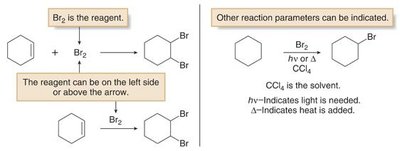
Organic reactions are typically represented with a single reaction arrow between starting materials and products. Reagents may be shown on the left or above the arrow, and solvents or conditions (such as heat or light) are indicated above or below the arrow.
Sequential reactions are numbered above the arrow to indicate the order of reagent addition.
Kinds of Organic Reactions
Organic reactions are classified based on the changes occurring in the molecules:
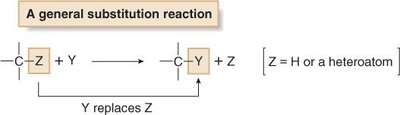
Substitution: An atom or group is replaced by another atom or group. Involves breaking and forming σ bonds at the same carbon atom.
Elimination: Elements are removed from the starting material, typically forming a π bond. Two σ bonds are broken.
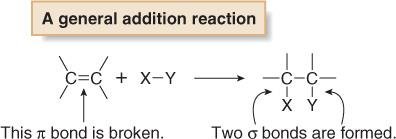
Addition: Elements are added to the starting material, breaking a π bond and forming two σ bonds.

Bond Making and Bond Breaking
Bonds in organic molecules can be broken in two ways:
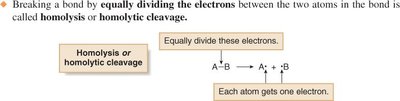
Homolysis (homolytic cleavage): Electrons are divided equally between the two atoms, generating uncharged reactive intermediates (radicals).
Heterolysis (heterolytic cleavage): Electrons are divided unequally, generating charged intermediates (ions).
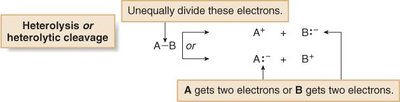
Arrow conventions are used to illustrate electron movement:
Half-headed curved arrow (fishhook): Shows movement of a single electron.
Full-headed curved arrow: Shows movement of an electron pair.


Stereochemistry: Chiral and Achiral Molecules
Chirality and Superimposability
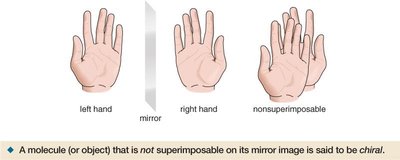
A molecule is chiral if it is not superimposable on its mirror image, much like left and right hands. An achiral molecule is superimposable on its mirror image, like a pair of socks.
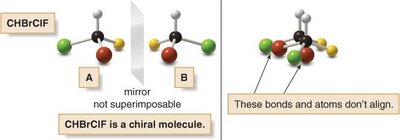
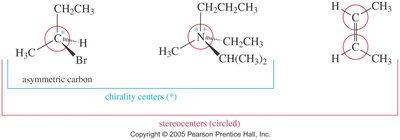
Chiral Centers and Stereogenic Centers
A chiral center (asymmetric carbon) is a tetrahedral carbon atom bonded to four different groups. The presence of a chiral center usually makes the molecule chiral. Stereogenic centers are atoms where the interchange of two groups produces a stereoisomer.
Asymmetric carbons are the most common chiral centers.
Double-bonded carbons in cis-trans isomers are also stereogenic centers.
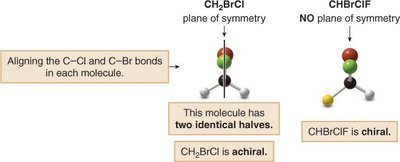
Internal Plane of Symmetry
An internal plane of symmetry divides a molecule into two identical halves. Molecules with such a plane are achiral, even if they contain asymmetric carbons.
Cis-1,2-dichlorocyclopentane is achiral due to its internal plane of symmetry.
Trans-1,2-dichlorocyclopentane is chiral because it lacks a plane of symmetry.
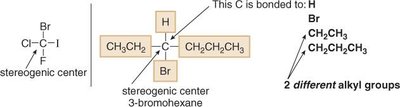
Locating Stereogenic Centers
To identify stereogenic centers, examine each tetrahedral carbon and the four groups attached. Exclude CH2, CH3, and any sp or sp2 hybridized carbons.
Drawing Enantiomers
To draw both enantiomers of a chiral compound, use the tetrahedral convention: two bonds in the plane, one on a wedge (in front), and one on a dash (behind). Place the four groups arbitrarily, then draw the mirror image.
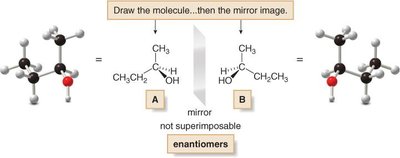
Practice and Application
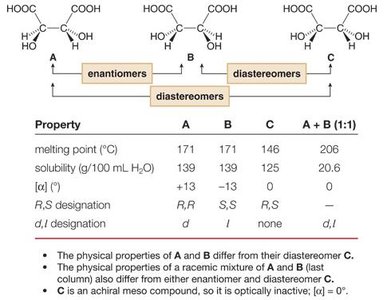
Example Table: Physical Properties of Stereoisomers
The table below compares the physical properties of enantiomers and diastereomers:
Property | A | B | A+B (1:1) | C |
|---|---|---|---|---|
Melting point (°C) | 171 | 171 | 146 | 206 |
Solubility (g/100 mL H2O) | 139 | 139 | 125 | 20.6 |
[α] | +13 | –13 | 0 | – |
R,S designation | R,R | S,S | R,S | – |
d,l designation | d | l | none | d,l |
Key points:
The physical properties of A and B differ from their diastereomer C.
The racemic mixture (A+B) has properties distinct from either enantiomer or diastereomer.
C is an achiral meso compound and is optically inactive ([α] = 0).

Chromatographic Resolution Example
Chromatographic resolution separates enantiomers using a chiral column. The (+) and (–) enantiomers bind differently to the chiral packing, allowing their separation.

Summary Table: Arrow Conventions in Organic Chemistry
Arrow | Name | Use |
|---|---|---|
→ | Reaction arrow | Drawn between starting materials and products |
⇌ | Double reaction arrows | Equilibrium equations |
↔ | Double-headed arrow | Resonance structures |
| Full-headed curved arrow | Movement of an electron pair |
| Half-headed curved arrow (fishhook) | Movement of a single electron |

Additional info: These notes cover key concepts from Chapters 4 and 5: The Study of Chemical Reactions and Stereochemistry, including reaction types, mechanisms, and the identification and properties of chiral molecules and stereoisomers.
