 Back
BackStereochemistry: Structure, Chirality, and Isomerism in Organic Molecules
Study Guide - Smart Notes

Chapter 5: Stereochemistry
Introduction to Stereochemistry
Stereochemistry is the study of the three-dimensional arrangement of atoms in molecules and its impact on their physical, chemical, and biological properties. Understanding stereochemistry is essential for organic chemistry, biochemistry, and biology, as biological systems often distinguish between molecules with subtle stereochemical differences.
Stereoisomers: Molecules with the same connectivity but different spatial arrangements.
Structural Isomers: Molecules with different connectivity.
Geometric Isomers: Isomers with restricted rotation, such as cis and trans forms.
Optical Isomers: Non-superimposable mirror images, known as enantiomers.
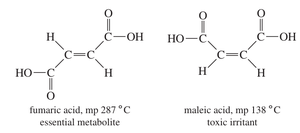
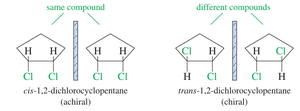
Geometric Isomerism: Cis and Trans Isomers
Geometric isomers differ in the arrangement of substituents around a bond with restricted rotation, such as double bonds or rings. The cis and trans prefixes describe the relative configuration of substituents.
Cis Isomer: Substituents on the same side.
Trans Isomer: Substituents on opposite sides.
Example: Maleic acid (cis) and fumaric acid (trans) are geometric isomers with distinct properties.
Chirality: The Concept of Handedness
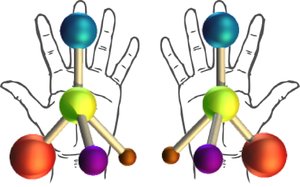
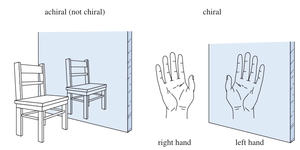
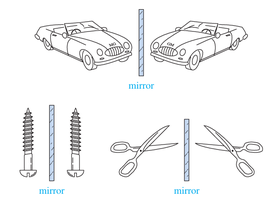
Chirality refers to the property of a molecule that makes it non-superimposable on its mirror image, much like left and right hands. Chiral molecules have mirror images that are distinct from the original, while achiral molecules are identical to their mirror images.
Chiral Objects: Have non-superimposable mirror images.
Achiral Objects: Have superimposable mirror images.
Chirality and Enantiomerism in Organic Molecules
Molecules can be classified as chiral or achiral based on their symmetry and the presence of chirality centers. Enantiomers are pairs of molecules that are nonsuperimposable mirror images of each other.
Enantiomers: Mirror-image isomers; pairs of compounds that are nonsuperimposable mirror images.
Chiral: Different from its mirror image; having an enantiomer.
Achiral: Identical with its mirror image; not chiral.

Testing for Chirality: Superimposability
To determine if a molecule is chiral, compare its mirror image. If the mirror image cannot be superimposed, the molecule is chiral and the two forms are enantiomers.
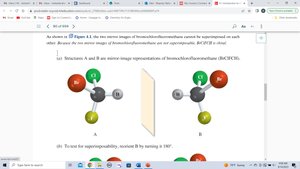
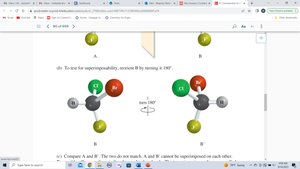
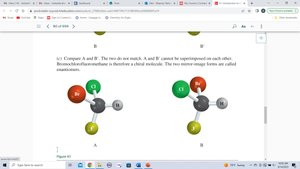
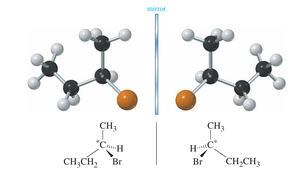
Chirality Centers and Stereocenters
A chirality center (often a carbon atom) is bonded to four different groups. The presence of a chirality center is a strong indicator of chirality, but molecules with multiple chirality centers may or may not be chiral.
Chirality Center: Tetrahedral carbon attached to four different atoms or groups.
Stereocenter: Any atom at which the interchange of two groups gives a stereoisomer.
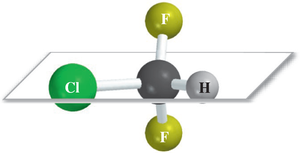
Symmetry and Chirality
Molecules with a plane or center of symmetry are achiral. A plane of symmetry divides the molecule into two mirror-image halves, while a center of symmetry means any line drawn from the center encounters identical elements at equal distances.
Absolute Configuration: Cahn-Ingold-Prelog (R,S) Notation
The absolute configuration of a chirality center is determined by ranking substituents according to atomic number and observing their spatial arrangement. The Cahn-Ingold-Prelog rules are used to assign R or S configuration.
Rule 1: Higher atomic number takes precedence.
Rule 2: Compare atoms attached to the chirality center at the first point of difference.
Rule 3: Work outward, comparing all atoms attached to a particular atom.
Rule 4: Evaluate substituent atoms one by one.
Rule 5: Multiply bonded atoms are considered as replicated substituents.
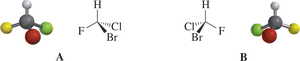
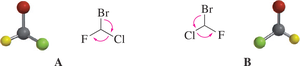
Physical Properties of Enantiomers
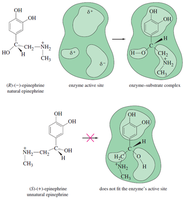
Enantiomers have identical physical properties (melting point, boiling point, density) except for optical activity. Their interactions with other chiral molecules, such as enzymes, can differ dramatically.
Optical Activity and Polarimetry
Optical activity is the ability of a chiral substance to rotate plane-polarized light. This property is measured using a polarimeter. Enantiomers rotate light in equal amounts but opposite directions, while racemic mixtures are optically inactive.
Specific Rotation [α]: Calculated using the formula , where is the observed rotation, is concentration (g/mL), and is path length (dm).
Enantiomeric Excess (ee):
Enantiomeric Ratio (er):

Chirality in Conformationally Mobile Systems
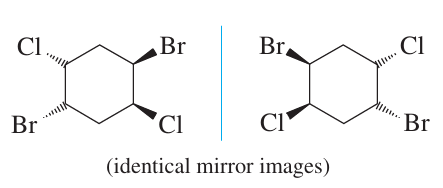
Some molecules, such as cyclohexane derivatives, can interconvert rapidly between mirror-image conformations, resulting in achirality despite having chiral forms.
Fischer Projections
Fischer projections are a two-dimensional representation of three-dimensional molecules, useful for visualizing stereochemistry in molecules with multiple chirality centers. Vertical bonds point away, horizontal bonds point toward the viewer.
Erythro Diastereomer: Like substituents on the same side.
Threo Diastereomer: Like substituents on opposite sides.
Diastereomers and Meso Compounds
Diastereomers are stereoisomers that are not mirror images. Meso compounds are achiral despite having asymmetric carbon atoms, due to internal symmetry.
Number of Stereoisomers: , where is the number of chirality centers.
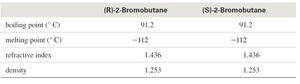
Summary Table: Physical Properties of Enantiomers
Property | (R)-2-Bromobutane | (S)-2-Bromobutane |
|---|---|---|
Boiling Point (°C) | 91.2 | 91.2 |
Melting Point (°C) | -112 | -112 |
Refractive Index | 1.436 | 1.436 |
Density | 1.253 | 1.253 |
Key Definitions
Enantiomers: Mirror-image isomers; pairs of compounds that are nonsuperimposable mirror images.
Chiral: Different from its mirror image; having an enantiomer.
Achiral: Identical with its mirror image; not chiral.
Racemic Mixture: Mixture containing equal quantities of enantiomers; optically inactive.
Meso Compound: Achiral compound with asymmetric carbon atoms due to internal symmetry.
