 Back
BackStructure and Preparation of Alkenes: Elimination Reactions
Study Guide - Smart Notes

Structure and Preparation of Alkenes: Elimination Reactions
Introduction to Alkenes
Alkenes are hydrocarbons characterized by the presence of at least one carbon–carbon double bond. They are also known as olefins, a term derived from their historical oil-forming properties. The double bond imparts unique chemical and physical properties to alkenes, distinguishing them from alkanes and alkynes.
Structure and Bonding in Alkenes
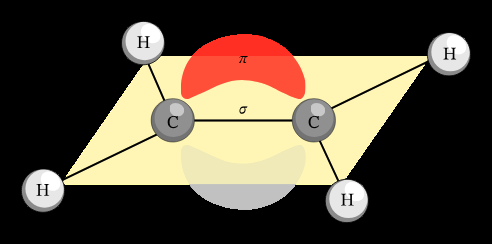
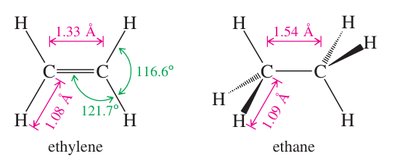
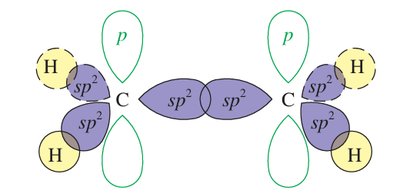
Hybridization: Each carbon in an alkene double bond is sp2 hybridized, resulting in a planar structure with bond angles close to 120°.
Sigma (σ) Bond: Formed by the overlap of sp2 orbitals between the two carbons and between carbon and hydrogen atoms.
Pi (π) Bond: Formed by the side-to-side overlap of unhybridized p orbitals on each carbon, above and below the plane of the molecule. This π bond restricts rotation around the double bond.
Physical Properties of Alkenes
Alkenes generally resemble alkanes in their physical properties, such as boiling and melting points. However, the presence of the double bond introduces a dipole moment in some cases and affects molecular geometry.
Lower alkenes (up to C4H8) are gases at room temperature.
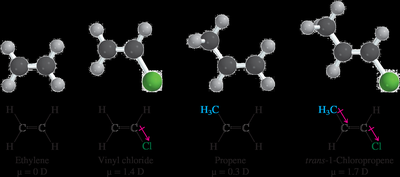
Alkenes can exhibit dipole moments depending on their substituents.
Elements of Unsaturation
Alkenes are considered unsaturated hydrocarbons because the double bond reduces the number of hydrogen atoms compared to alkanes. Each element of unsaturation (double bond, triple bond, or ring) corresponds to two fewer hydrogens than the saturated formula (CnH2n+2).
To calculate the number of elements of unsaturation, subtract the number of hydrogens in the molecular formula from the saturated formula and divide by two.
Halogens are counted as hydrogens; oxygen is ignored; nitrogen counts as half a carbon.

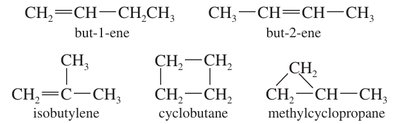
Nomenclature of Alkenes
Alkenes are named by replacing the -ane ending of the corresponding alkane with -ene. The parent chain is the longest continuous chain containing the double bond, and numbering gives the double bond the lowest possible locant. Double bonds take precedence over alkyl and halo substituents, but hydroxyl groups outrank double bonds.
Common alkenyl groups: vinyl, allyl, isopropenyl.
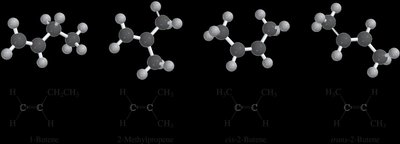
Isomerism in Alkenes
Alkenes can exhibit structural isomerism and geometric (cis-trans) isomerism. Geometric isomerism arises when each carbon of the double bond has two different substituents, leading to cis (same side) and trans (opposite side) isomers.
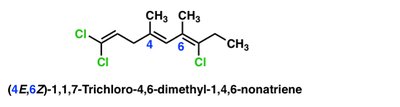
E/Z Notational System
The E/Z system (from the German Entgegen and Zusammen) is used when cis/trans notation is ambiguous. The Cahn-Ingold-Prelog priority rules assign priorities to substituents on each carbon of the double bond. Z (zusammen) means higher priority groups are on the same side; E (entgegen) means they are on opposite sides.

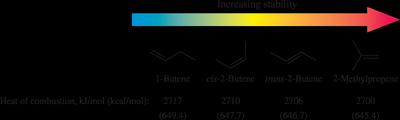
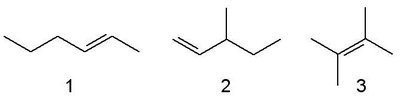
Relative Stabilities of Alkenes
The stability of alkenes depends on several factors:
Degree of substitution: More substituted alkenes are more stable (tetra > tri > di > mono-substituted).
Steric effects: Trans isomers are generally more stable than cis due to less steric strain.
Chain branching: Branched alkenes are more stable than unbranched isomers.
Heat of hydrogenation: Lower heat of hydrogenation indicates greater stability.
Elimination Reactions: Preparation of Alkenes
Alkenes are commonly prepared by elimination reactions, where atoms are removed from adjacent carbons, forming a double bond. The two main mechanisms are E1 (unimolecular elimination) and E2 (bimolecular elimination).
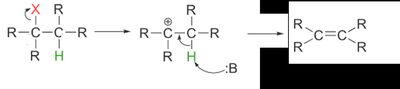
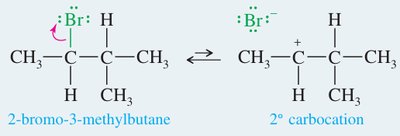
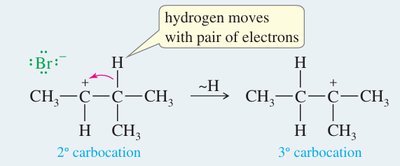
E1 Mechanism
Two-step process: formation of a carbocation intermediate, followed by loss of a proton.
First-order kinetics: rate depends only on the substrate.
Common with tertiary and some secondary alkyl halides in polar solvents.
Often competes with the SN1 substitution mechanism.
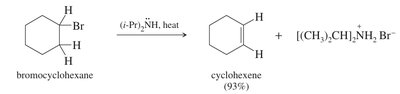
E2 Mechanism
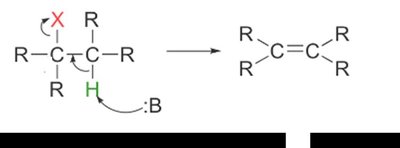
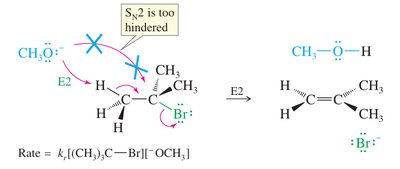
One-step, concerted process: base abstracts a proton as the leaving group departs.
Second-order kinetics: rate depends on both substrate and base.
Favored by strong bases and hindered substrates (e.g., tertiary alkyl halides).
Preferred for synthetic purposes due to fewer side reactions.
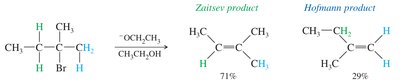
Regioselectivity: Zaitsev's Rule
Elimination reactions are often regioselective. Zaitsev's rule states that the most substituted (and thus most stable) alkene is usually the major product.

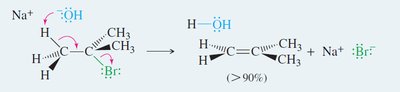
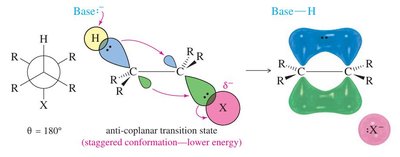
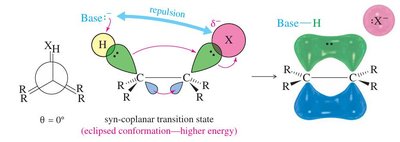
Stereochemistry of E2 Mechanism
E2 eliminations are stereospecific: the base and leaving group must be anti-coplanar (180° apart) for the lowest energy transition state.
Syn-coplanar eliminations are rare due to higher energy (eclipsed conformation).
Comparison of E1 and E2 Mechanisms
Feature | E1 | E2 |
|---|---|---|
Base | Weak base sufficient | Strong base required |
Solvent | Good ionizing solvent | Wide variety of solvents |
Substrate | 3° > 2° | 3° > 2° > 1° |
Leaving group | Good one required | Good one required |
Kinetics | First order | Second order |
Orientation | Zaitsev product | Zaitsev product |
Stereochemistry | No special geometry | Coplanar transition state required |
Rearrangements | Common | Impossible |

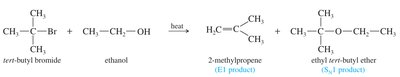
Preparation of Alkenes from Alcohols
Alkenes can also be prepared by acid-catalyzed dehydration of alcohols. This reaction typically follows an E1 mechanism for secondary and tertiary alcohols, and an E2 mechanism for primary alcohols. The major product is usually the most substituted alkene (Zaitsev's rule).
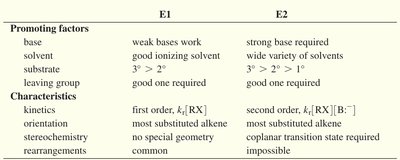
Summary Table: E1 vs E2 Mechanisms
Promoting Factors | E1 | E2 |
|---|---|---|
Base | Weak bases work | Strong base required |
Solvent | Good ionizing solvent | Wide variety of solvents |
Substrate | 3° > 2° | 3° > 2° > 1° |
Leaving group | Good one required | Good one required |
Characteristics | ||
Kinetics | First order, | Second order, |
Orientation | Most substituted alkene | Most substituted alkene |
Stereochemistry | No special geometry | Coplanar transition state required |
Rearrangements | Common | Impossible |
Key Equations
General formula for alkenes:
Degree of unsaturation:
Heat of hydrogenation: (used to compare alkene stabilities)
Additional info:
For more complex elimination reactions, consider possible carbocation rearrangements (hydride or alkyl shifts) in E1 mechanisms.
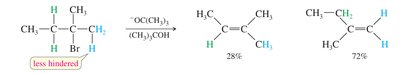
Bulky bases (e.g., tert-butoxide) favor the formation of the less substituted (Hofmann) alkene due to steric hindrance.
Alcohol dehydration is an important laboratory method for alkene synthesis, especially for tertiary alcohols.
